
 |
 |
Published by:
Aardsma Research & Publishing
412 N Mulberry St
Loda, Illinois 60948-9651
www.BiblicalChronologist.org
AgingCauseAndCure.com
Printed in the United States of America
Library of Congress Control Number: 2021909407
ISBN 978-0-9647665-9-4

| List of Tables | 11 |
| List of Figures | 13 |
| Dedication | 17 |
| Acknowledgments | 19 |
| Preface | 21 |
| Part 1: Aging 1 | 27 |
| 1 Beginnings | 29 |
| 1.1 Cleaning the Slate | 29 |
| 1.1.1 "Aging" | 29 |
| 1.1.2 Impact of Modern Medicine on Maximum Life Span | 30 |
| 1.1.3 "Special" Groups and Individuals | 31 |
| 1.1.4 "Normal" Life Span | 31 |
| 1.2 A New Hypothesis | 32 |
| 1.2.1 Malady X | 33 |
| 1.2.2 The Number One Health Problem | 34 |
| 1.3 The Difficulty of the Research Problem | 35 |
| 1.4 Conclusion | 36 |
| 2 The Data | 37 |
| 2.1 Life Spans | 38 |
| 2.2 Birthdates | 40 |
| 2.3 Life Expectancy | 42 |
| 2.4 Conclusion | 44 |
| 3 The Biblical Life Expectancy Graph | 45 |
| 3.1 A Powerful Instrument | 46 |
| 3.1.1 Supernatural | 46 |
| 3.1.2 Vapor Canopy | 48 |
| 3.1.3 Evolution | 49 |
| 3.2 Conclusion | 50 |
| 4 The Central Hypothesis | 51 |
| 4.1 Deficiency Diseases | 51 |
| 4.1.1 The Example of Vitamin C | 52 |
| 4.2 Central Hypothesis | 54 |
| 4.2.1 Complex of Symptoms | 54 |
| 4.2.2 Particular Symptoms | 55 |
| 4.2.3 Apparent Contrast | 56 |
| 4.2.4 Variable Time of Onset | 56 |
| 4.2.5 Why Life Spans Changed | 56 |
| 4.3 Conclusion | 57 |
| 5 Properties of Vitamin X | 59 |
| 5.1 The Eber–Peleg Drop | 59 |
| 5.2 Biological Half-life | 61 |
| 5.3 Biological Half-life of Vitamin X | 61 |
| 5.4 Environmental Half-life of Vitamin X | 63 |
| 5.5 Conclusion | 64 |
| 6 The Environmental Abundance of Vitamin X | 65 |
| 6.1 Noah's Life Span | 65 |
| 6.2 The Environmental Abundance Graph | 66 |
| 6.2.1 The Post-Peleg Decline | 68 |
| 6.2.2 The Moses Drop | 68 |
| 6.3 Conclusion | 70 |
| 7 The Natural Synthesis and Distribution of Vitamin X | 73 |
| 7.1 How Was Vitamin X Distributed by Nature? | 73 |
| 7.2 How Was Vitamin X Synthesized by Nature? | 75 |
| 7.3 How Did Vitamin X Enter the Human Diet? | 76 |
| 7.4 The MSA Example | 76 |
| 7.5 Conclusion | 77 |
| 8 What the Flood Broke | 79 |
| 8.1 The Nature of the Flood | 79 |
| 8.2 The Nature of Sea Floors | 80 |
| 8.3 The Source of the Precursor | 81 |
| 8.4 Conclusion | 81 |
| 9 Vitamin X Revealed | 83 |
| 9.1 A Phosphorus Trace Gas | 84 |
| 9.1.1 Methylated Phosphorus Gases | 87 |
| 9.2 A Theory of Vitamin X | 91 |
| 9.3 Conclusion | 93 |
| 10 Vitamin X Confirmed | 95 |
| 10.1 The Data | 96 |
| 10.2 Is it Real? | 96 |
| 10.3 Discussion | 98 |
| 10.4 Conclusion | 100 |
| 11 Modeling the Biblical Life Expectancy Data | 101 |
| 11.1 The Model | 102 |
| 11.1.1 Details | 102 |
| 11.2 Results | 108 |
| 11.2.1 Environmental Free Parameters | 108 |
| 11.2.2 Physiological Free Parameters | 109 |
| 11.3 Conclusion | 110 |
| 12 The Cure for Aging 1 | 113 |
| 12.1 Dietary Supplementation With MePiA | 113 |
| 12.2 Recommended Daily Intake of MePiA | 114 |
| 12.2.1 Relative Versus Absolute Concentrations | 115 |
| 12.2.2 Maximum Saturation Intake | 116 |
| 12.2.3 Ballpark Estimates | 118 |
| 12.2.4 Overdosing | 119 |
| 12.2.5 Present Recommendation | 121 |
| 12.3 Rejuvenation Once Aging 1 is Cured | 122 |
| 12.4 Conclusion | 122 |
| 13 Conclusion to Part 1 | 125 |
| Part 2: Aging 2 | 129 |
| 14 Human Aging is a Two-Phase Disease | 131 |
| 14.1 Implications of the Pre-Flood Survival Curve | 133 |
| 14.1.1 A Mechanical Example | 135 |
| 14.2 Two-Phase Human Aging | 136 |
| 14.3 Conclusion | 137 |
| 15 The Cause of Aging 2 | 139 |
| 15.1 Derivation of a Theory of Aging 2 | 139 |
| 15.1.1 The mtDNA Replacement Hypothesis | 142 |
| 15.1.2 Length of the Poisson Sequence | 143 |
| 15.1.3 Modern Age of Onset of Aging 2 | 144 |
| 15.1.4 The Poisson Sequence Conundrum | 145 |
| 15.1.5 A Bold Postulate | 147 |
| 15.1.6 A Second Obvious Question and a Second Bold Postulate | 151 |
| 15.2 Conclusion | 151 |
| 16 Testing the Theory of Aging 2 | 153 |
| 16.1 Psephomere Vestiges | 154 |
| 16.1.1 A Brief Review of Aging 2 Diseased Human mtDNA Replication | 155 |
| 16.1.2 Idling and Other Vestiges | 158 |
| 16.2 Conclusion | 162 |
| 17 The Cure for Aging 2 | 163 |
| 17.1 Curing Aging 2 the Right Way | 164 |
| 17.2 Noah's Rejuvenation | 164 |
| 17.2.1 Robustness of Noah's Rejuvenation | 165 |
| 17.3 The Progression and Healing of Aging 2 | 167 |
| 17.4 Check: A Digital Organism Model | 168 |
| 17.4.1 Results | 170 |
| 17.5 Using the Model | 173 |
| 17.5.1 A Second Estimate of the Modern Age of Onset of Aging 2 | 173 |
| 17.5.2 The Healing Rate of Aging 2 | 175 |
| 17.6 Conclusion | 176 |
| 18 Conclusion to Part 2 | 179 |
| Part 3: Aging 0 | 181 |
| 19 A Distinction Between Ancient and Modern Aging | 183 |
| 19.1 Aging 0 Applies Only to Modern Aging | 183 |
| 19.1.1 A Caveat | 185 |
| 19.2 An Important Implication | 186 |
| 19.3 Conclusion | 186 |
| 20 The Cause of Aging 0 | 189 |
| 20.1 MePA Is a Vitamin | 189 |
| 20.1.1 Vitamin Discovery Has Not Ended | 189 |
| 20.1.2 MePA Behaves Similar to Other Vitamins | 190 |
| 20.1.3 MePA is Vitamin-Like, Not Drug-Like | 193 |
| 20.1.4 MePA Satisfies the Criteria for a Vitamin | 194 |
| 20.2 Aging 0 in Ancient Times | 196 |
| 20.3 Conclusion | 197 |
| 21 The Cure for Aging 0 | 199 |
| 21.1 Recommended Daily Intake | 199 |
| 21.2 Conclusion | 201 |
| 22 Conclusion to Part 3 | 203 |
| Part 4: The Eradication of Human Aging | 205 |
| 23 The Present Emergency | 207 |
| 23.1 Defining the Problem | 209 |
| 23.2 The Modern Actuarial Dataset | 210 |
| 23.3 Method | 212 |
| 23.3.1 Step 1: Model the Dominant Aging Disease | 212 |
| 23.3.2 Step 2: Fit the First-Approximation Model to the Modern Life Span Dataset | 213 |
| 23.3.3 Step 3: Upgrade the Model to Include Aging 2 Saturation | 226 |
| 23.3.4 Step 4: Upgrade the Model to Include a Second Aging Disease | 228 |
| 23.3.5 Interim Assessment | 229 |
| 23.3.6 Step 5: Upgrade the Model to Include Aging 1, the Third Aging Disease | 230 |
| 23.4 Discussion | 234 |
| 23.5 Conclusion | 237 |
| 24 Potential Longevity Today | 239 |
| 24.1 Method | 240 |
| 24.1.1 Life Expectancy | 241 |
| 24.1.2 Computer Program | 242 |
| 24.2 Results | 248 |
| 24.3 Discussion | 248 |
| 24.4 Conclusion | 252 |
| 25 How to Maximize Health and Longevity | 255 |
| 25.1 Rule 1 | 256 |
| 25.2 Rule 2 | 258 |
| 25.3 Conclusion | 259 |
| 26 Conclusion to Part 4 | 261 |
| Epilogue | 263 |
| Matthew and Esther's Personal Testimonial | 264 |
| Personal Health | 264 |
| Getting and Staying Motivated | 265 |
| Parenting and the Anti-Aging Vitamins | 267 |
| Esther | 268 |
| Gerald and Helen's Personal Testimonial | 275 |
| Conclusion | 287 |
| Appendix MeP_20170105.F95 | 289 |
| Index | 303 |

| 2.1 Selected biblical life span data. | 39 |
| 2.2 Birthdates of selected biblical males. | 41 |
| 2.3 Point estimates of life expectancies from Adam to Moses. | 43 |
| 14.1 Life span data for pre-Flood males. | 131 |
| 23.1 Numbers of deaths in 2016 resulting from the three aging diseases. | 237 |
| 2.1 Biblical data showing life expectancy at birth for selected males. | 44 |
| 3.1 Measured temperature versus time for a bowl of hot water. | 47 |
| 4.1 Structure of the vitamin C molecule. | 53 |
| 5.1 The Eber–Peleg Drop. | 60 |
| 6.1 Sudden increase and drop in abundance of vitamin X. | 67 |
| 6.2 The post-Peleg decline and the Moses Drop. | 69 |
| 6.3 Pre-Flood, Spike, Decline, and Modern time periods. | 71 |
| 9.1 Oceanic phosphate surface distribution. | 85 |
| 9.2 Oceanic nitrate surface distribution. | 85 |
| 9.3 The MeP, MePiA and MePA molecules. | 90 |
| 10.1 Survival curve datasets for the MePA-vs-MePiA-treated ICR female weanling mice experiment. | 97 |
| 10.2 Survival curve datasets for the MePA-vs-MePiA-treated ICR female weanling mice experiment with three additional batches of the same type of mice. | 99 |
| 11.1 Results of the biblical life span data model. | 103 |
| 11.2 Rate of "aging" versus MePiA body pool size. | 107 |
| 11.3 Pre-Flood, break-even, and saturation body pool sizes. | 109 |
| 11.4 The good fit of the model to the data. | 111 |
| 12.1 Acute oral toxicity for MePiA compared to table salt and caffeine in laboratory rats. | 120 |
| 12.2 Rejuvenation during the Spike. | 123 |
| 14.1 Survival curves for ancient and modern males. | 133 |
| 14.2 Survival curves for ancient and modern males plus modern curve stretched along the time axis by a factor of ten. | 134 |
| 15.1 Survival curves for ancient (orange dots) and modern (blue dots) males. | 140 |
| 15.2 Death rate distributions for modern and ancient human males. | 143 |
| 15.3 Conceptual diagram of human double-stranded mtDNA with "in-line telomere" shown in red. | 147 |
| 16.1 Schematic diagram of human double-stranded mtDNA in its resting state with details of the NCR. | 155 |
| 16.2 Aging 2 diseased human double-stranded mtDNA being replicated. | 157 |
| 16.3 Healthy human double-stranded mtDNA with details of the NCR. | 159 |
| 16.4 Healthy human double-stranded mtDNA being replicated. | 159 |
| 16.5 Hypothesized "queen bee" replication mechanism of mtDNA. | 161 |
| 17.1 Induction of Aging 2 near 800 years of age. | 171 |
| 17.2 Reversal of Noah's progression toward induction of Aging 2 during the Spike. | 171 |
| 17.3 Induction of Aging 2 near 14 years of age. | 172 |
| 17.4 Average cell age (green) for induction of Aging 2 near 14 years of age. | 173 |
| 17.5 Solution giving an average cell age between 7 and 10 years. | 174 |
| 17.6 Example healing curves for Aging 2 disease. | 175 |
| 20.1 Geriatric features in niacin-deficient 6-month-old pig. | 190 |
| 23.1 Survival curve data for U.S. males and females for the year 2016. | 211 |
| 23.2 Survival curve data for U.S. males and females for the year 2016 with least squares fit curves of the Aardsma model. | 211 |
| 23.3 The 2016 actuarial life table data for U.S. males. | 215 |
| 23.4 The 2016 actuarial life table data for U.S. males with the Y axis expanded by a factor of one hundred. | 218 |
| 23.5 The 2016 actuarial life table data for U.S. males with the Y axis expanded by a factor of one thousand. | 219 |
| 23.6 The 2016 actuarial life table data for U.S. males with least squares fit of the single-aging-disease model. | 221 |
| 23.7 Probability of death per year calculated by the digital organism model compared with the 2016 actuarial life table data for U.S. males. | 225 |
| 23.8 The 2016 actuarial life table data for U.S. males with least squares fit of the single-aging-disease model including Aging 2 saturation. | 227 |
| 23.9 The 2016 actuarial life table data for U.S. males showing the least squares fit of the two-aging-diseases model. | 229 |
| 23.10 Putative Aging 0 contribution of the least squares fit of the two-aging-diseases model to the 2016 actuarial life table data for U.S. males. | 231 |
| 23.11 Final least squares fit of the three-aging-diseases model for U.S. males. | 232 |
| 23.12 Final least squares fit of the three-aging-diseases model for U.S. females. | 233 |
| 24.1 Life expectancy first exceeds 1,000 years in 4444 B.C. | 239 |
| 24.2 Life expectancy for modern U.S. males and females not taking the anti-aging vitamins. | 242 |
| 24.3 Example healing curves for Aging 2 disease. | 245 |
| 24.4 Life expectancies for U.S. males and females starting to take the anti-aging vitamins and not starting to take the anti-aging vitamins. | 249 |
| 24.5 Survival curves for U.S. females supplementing with the anti-aging vitamins and not supplementing with the anti-aging vitamins. | 251 |
| 25.1 Life expectancy increase factor for U.S. males and females starting to take the anti-aging vitamins. | 255 |
| 25.2 Life expectancies for U.S. males and females starting to take the anti-aging vitamins and not starting to take the anti-aging vitamins. | 256 |

Helen,
How many times have we looked at one another these past seventeen years and recited those words from David Mamet's film, The Winslow Boy, "Are we crazy, you and I?"
Thank you for choosing to walk this difficult road with me. You have been a friend encouraging me to press on, a secretary running the business, a comrade covering my back, a nurse watching by my bedside, a business partner financing her research scientist, a teammate setting me up for the shot, … and all the while a poverty-line housewife looking after her children and her man. How can I not admire you?
Well, now we have the answer. It seems we were not crazy after all. And I am so looking forward to growing young with you.
Gerald
We wish to express our sincere appreciation to Jennifer Hall, Steve Hall, and Tom Godfrey who assisted in multiple ways with the production of this second edition. All three were involved with proofreading and editing assistance. In addition, Steve Hall supplied graphic design and artwork, and Tom Godfrey crafted the index.

by Gerald Aardsma
So at last Faramir and Éowyn and Meriadoc were laid in beds in the Houses of Healing; and there they were tended well. For though all lore was in these latter days fallen from its fullness of old, the leechcraft of Gondor was still wise, and skilled in the healing of wound and of hurt, and all such sickness as east of the Sea mortal men were subject to. Save old age only. For that they had found no cure…[1]
I am a research scientist. I have spent most of my life researching at the interface of science and the Bible. My science specialty is physical dating methods such as radiocarbon. My earliest full-time Bible/science research effort centered around the question of why nobody had ever been able to pin a functional historical date on Noah's Flood. This led, eventually, to the discovery that exactly 1000 years had been accidentally dropped from traditional biblical chronology due to an inadvertent copy error in a number found in 1 Kings 6:1.[2]
This rapidly led to answers to other Bible/science questions I had not even set out to investigate. For most of the final decade of the twentieth century, I was immersed in research connected to the Exodus of Israel from Egypt, the ensuing Conquest of Canaan, and the much earlier-in-time Flood of Noah. I found that the Exodus was a real historical event, and that the biblical description of it was simply historical.[3] The scholars were all saying the opposite, but that was because they had their biblical chronology wrong by 1000 years. You cannot find an object if you spend all your time looking for it a thousand miles from where it is located, and you cannot find a historical event if you spend all your time looking for it one thousand years from when it happened.
Also severely in conflict with mainstream scholarship, I found that the Flood, too, was a real historical event, and that the biblical record of it, too, was simply historical. Mainstream scholarship has held for some decades that the Flood is mere legend.
The lengths of reign of the earliest kings [of the Sumerian King List] are immense, and clearly belong to purely legendary time, an assumption confirmed by the fact that they are presented as ruling 'before the flood'.[4]In sharp contrast, I found that the Genesis record of the Flood was of such accuracy that it had to have been written by an eyewitness of that event.[5]
Concurrent with all of this research, I pursued a lifelong interest in why, according to Genesis, humans had lived so much longer before the Flood than we do today. I began to tackle that question full time with the advent of the new millennium. The first edition of this book shared what I had found during the first seventeen years of all-out, strenuous research effort on that question. This second edition adds an additional four years of research to the story. Once again, I have found that mainstream scholarship is lost at sea with respect to aging. This is not coincidental. It is inevitable. To understand what aging is and how aging is to be cured, the history of our planet and of our species found in Genesis is indispensable, as the pages of this book will show. You cannot be wrong about the historicity of Genesis and have any hope of being right about aging.
The need for this second edition arises out of both (1) the ethics of research into human aging and (2) the pace of research discovery since publication of the first edition. The ethics of aging research demands unusually rapid publication and application of research findings when those findings promise to save lives with little to no attached risk. Normally, a scientist has the luxury of checking results via duplicated experiments to be certain of his conclusions before moving on to their publication and practical application. Prudence—protection of his reputation and his career—demands that he do so. No such luxury exists in the present case. More than 150,000 people die of aging every day. If it is evil to allow even one person to die who might have been helped by a new potential cure of a disease just to protect one's reputation or career, then it is surely a monstrous evil to allow 150,000 people per day to do so for this reason. This has been recognized by moral philosophers for a very long time:[6]
Deliver those who are being taken away to death,In keeping with this ethical mandate, the first edition of this book was pushed to publication as rapidly as possible. The reduction of age-related morbidity and mortality in real people's lives which resulted from this deliberate action seems undeniable at this stage. Not surprisingly, however, the book was soon seriously in need of correction and revision. Two years following its publication, it was necessary to publish a separate addendum for this purpose. Thus, publication of this second edition, four years following the first, accomplishes two objectives. First, it eliminates the clumsiness and confusion resulting from having to read the first edition and its separately published addendum. Second, it adds a great deal of additional new theoretical and experimental progress made since the first edition was published. As with the first edition, material has been drawn freely from the pages of The Biblical Chronologist newsletter,[7] where research is reported as it happens. Content from these original research reports has been edited, corrected, and updated as necessary to make this book as clear and correct as the present state of knowledge permits. The goal with the production of this second edition has been to provide the reader with a single volume containing as complete an explanation of the cause of aging and its cure as is so far possible.
And those who are staggering to slaughter, O hold them back.
If you say, "See, we did not know this,"
Does He not consider it who weighs the hearts?
And does He not know it who keeps your soul?
And will He not render to man according to his work?
The first edition was necessarily written by myself alone, as I was the sole scientist involved in the research it described up to that point. In 2019, my son Matthew completed his PhD in animal nutrition at Purdue University and kindly consented to lend his considerable talents and energy to this present, urgent, applied research effort. Matthew now oversees the experimental side of the research program at Aardsma Research & Publishing (ARP). While his self-sacrificing devotion to this humanitarian effort is free of personal demands, his original research contributions to this work, of their own accord, demand that he now be included as a coauthor of this second edition. It has thus been my pleasure and good fortune to welcome aboard his considerable help in its creation.
Simultaneous with the past four years of ongoing research, substantial time and money have been invested in making the cure for modern human aging commercially available. The driving motivation behind this commercial effort is humanitarian. Aging has been exacting a terrible toll of sickness, suffering, and death on humanity for thousands of years. The fact that we have grown quite accustomed to the morbidity and mortality of aging does not lessen the ongoing tragedy of aging disease nor make it right. The purpose of the commercial effort is to enable as many people as possible to avail themselves of the new-found cure for aging as quickly as possible. There now exists a website, DrAardsmasVitamins.com, which makes procurement of the cure an easy matter. This commercial effort is seen as an interim measure while we wait on government to come up to speed. Judging from past performance, appropriate government action could easily be decades away.
I am hopeful that this book, with its intensely urgent, practical message, will not come to crowd out the research of the earlier books on which it has been built:[8]
Gerald E. Aardsma
June 30, 2021
Loda, IL

So all the days of Adam were nine hundred and thirty years, and he died. –Genesis 5:5.
So all the days of Methuselah were nine hundred and sixty-nine years, and he died.–Genesis 5:27.
So all the days of Noah were nine hundred and fifty years, and he died. –Genesis 9:29.
According to the record of the ancient past found in the biblical book of Genesis, humans once lived in excess of 900 years. Today, humans rarely live in excess of 90 years. Why were human life spans so very much greater in the distant past than they are today?
Before profitable analysis of the true issues surrounding the ancient mystery of human longevity can begin, there are a number of misconceptions and imprecise definitions in common use which must be cleared up. The meaning of the word "aging" is a good place to begin.
In common use, "aging" can mix together elements of both "maturing" (or "growing up") and "declining" (or "growing old"). Contrary to this common use, biological considerations lead to a natural separation of the concepts of "growing up" and "growing old." "Growing up" is seen biologically as a time of cell proliferation and differentiation. In contrast, "growing old" involves an increasing loss of cell mass and increasing loss of functional ability originating at the cellular level.
Today, humans "grow up" during their first several decades. They then enter a plateau for several decades, during which they are neither maturing physically nor substantially declining. This is followed by another few decades during which physical decline becomes apparent at an ever-accelerating pace, culminating in death.
The phases of a person's life can be likened to the phases in the life of a building. The "growing up" phase corresponds to construction of the building. The plateau phase corresponds to the building's serviceable life. The decline phase corresponds to the building's eventual demise due to loss of structural strength in its materials.
It is natural to separate the concepts of construction and aging when we think about the life of a building. Similarly, the concepts of "growing up" and "growing old" need to be kept separate as we study human aging. Babies mature into adults. The cure for aging will not turn adults back into babies.
In this book, factors affecting the rate of maturation are not of much interest. Factors affecting the length of the plateau and decline phases are the present focus.
Another common misconception is that people are reaching maximum ages today far in excess of the maximum ages people could hope to obtain a thousand years ago. The popular notion here is that modern science and medicine have brought about a remarkable increase in the maximum length of life.
One has simply to recall the "threescore years and ten" of Psalm 90 (KJV) to know that this idea is false. In point of fact, modern science has been able to accomplish next to nothing to increase the maximum age to which people can live. People have been living into their seventies and eighties and beyond for the past several thousand years. Unfortunately, modern science is totally at a loss at present to know how to extend substantially the maximum human life span—which is why this book is necessary.
What modern science has done is to increase the average life span. That is, modern science and medicine have made it possible for a much larger percentage of the population to reach their seventies before dying. For example, in the past, many individuals died in infancy and early childhood as a result of disease. Modern science has found ways to protect children from these diseases, thus enabling many who would have died in infancy in the past to live on into their seventies and beyond in the present. The net effect of this is to increase the average age at death for the overall population.
Modern medicine has become very good at keeping people alive long enough for them to reach the decline phase, and modern medicine has become very good at keeping people alive a little longer during the decline phase. It has so far been able to do almost nothing to alter the age at which the decline phase is reached, and it has so far been able to do nothing to alter the inevitability of death within a few short decades of entering the decline phase.
Still another misconception is that "special" groups or individuals living today have maximum life spans remarkably different from the overall population—either far above or far below the normal life span today. One reflection of this is the notion that "primitive" peoples live only into their thirties.
This is a confusion of average and maximum life spans again. The average life span can be much reduced in primitive living conditions, but this does not alter the maximum possible life span. In primitive living conditions, disease and exposure to a harsh environment can result in the death of many people while they are still relatively young. But one still finds elderly individuals in populations raised in primitive circumstances—individuals who are, in fact, in their eighties and nineties.
Another reflection of this "special groups" idea is the notion that people who live in particular geographical locations (e.g., Tibet) or who hold particular professions (e.g., Tibetan priests) live to extreme ages. In actual fact, no dependence of maximum life span on geographical location or profession is found when authenticated records of individuals of verifiable identity are examined.
Perhaps the most difficult misconception to correct is the belief that 75 years is a normal life span for humans. The Genesis record of human life spans—Noah living to 950, for example—shows immediately that this belief is simply false. If we are to take biblical history seriously—and there are good reasons today why we should do so—then we must conclude that death at 75 is not normal for humans at all.
Imagine an island community, cut off from the rest of the world, where everybody dies before age 40 due to a certain double recessive genetic defect which has come to be found in all individuals in the population. This defect causes them all to be highly susceptible to cancer. As a result, all contract cancer and die in their fourth decade of life.
If this community remained cut off from the rest of the world for many generations, it is easy to see how they could ultimately come to believe that death by age 40 was normal for humans—and not only normal, but indeed proper. The general theory of aging for biological organisms[9] says that this mode of death would, in fact, to them, be "aging." It is probable that many of them would respond with skepticism and disdain if someone were to suggest the idea that many of their distant ancestors, who had discovered and populated the island thousands of years previously, had lived into their eighties and nineties. Certainly many of them would find the suggestion incredible that practical steps (i.e., marriage outside the island population) might be taken to restore an average life span of 75 years to their community. And some, no doubt, would assert that it was the will of God for humans to die before age 40, and that it was impious to meddle in such matters.
But they would be wrong.
The Genesis longevity data teach us that the world in which we live today is like this island community. Seventy-five years has become the average life span. It has been this way for thousands of years. But it is entirely wrong to mistake that to which we have become accustomed for that which should rightfully be.
Genesis teaches us that we must reorient our thinking. We must recognize that the present human life span of 75 years is a very sad state of affairs indeed. Much more dramatic than our imaginary islanders whose life spans were reduced a mere factor of two, our life spans have been reduced by over a factor of ten. Far from 75 years being "normal" for humans, we must acknowledge that the entire human population today is, in fact, subject to a devastating malady.
This idea, that human aging, as we know it, is a malady—a disease—is the fundamental hypothesis underlying this book. You will find that this hypothesis has been corroborated beyond reasonable doubt by the time you have finished reading this book.
We have learned to call this malady "old age," and we have learned to accept it. But Genesis shows us that this is entirely wrong-headed. It shows us that "old age" is a false label, and a highly misleading one. When we come to grips with what Genesis plainly shows and accept it at face value, we see immediately that nobody has ever died of "old age" at 75 or even at 125. The Genesis life span data teach us that 75 is not an old age. It is laughable to call an individual "old" who has lived only 75 years in a population sporting many individuals in excess of 750 years, as was the case in the long-ago days recorded in the early chapters of Genesis.
The biblical life span data make it clear that nobody dies of "old age" at 75 years, for 75 years is not an old age for humans at all. People routinely die within a few decades of the young age of 75 today, but they do not die because of their age. Time is not the killer. They die because they have been afflicted with a devastating malady which tends to kill humans within a few decades of 75 years today. This malady decimates our bodies, causing them to lose functional ability and waste away while we are still very young—before we have achieved even one tenth of our life span potential.
To focus attention on the true essence of the longevity problem, we will, for a few chapters, use "aging" and "old age" in quotes, and we will make use of the new term, "malady X" (read "malady X-bar"). For example: "He died of malady X" rather than "He died of old age." (The logic behind this nomenclature will be explained in a subsequent chapter.)
By substituting "malady X" for "old age," we mean deliberately to part company with the false idea that people die within a few decades of 75 today because they are aged and replace it with the true idea that people die within a few decades of 75 today because they are afflicted. By putting "aging" and "old age" in quotes, we mean to make it perfectly clear that time is not the essence of the problem. We mean to emphasize that the essence of the problem is what medicine routinely calls disease.
If we are to think accurately about longevity in light of what Genesis shows us, then we must begin to see what we presently call "old age" as simply another human disease. We have introduced the new term "malady X" as a temporary name for this disease to facilitate its study until we have learned enough about it to name it something more appropriate.
Malady X is a disease that manifests itself by, among other things, loss of hair color, wrinkled skin, vision impairment, loss of physical strength, and increasing susceptibility to a large number of diseases. Malady X symptoms are universally seen in all individuals over the entire globe today beginning in the fifth decade of life. The sad result is death of most individuals within a few decades of 75 years of age, and of all individuals before 130 years of age—dramatically short of the known life span potential of humans, in excess of 900 years.
From the start of this quest, decades ago, the research problem was to find the physical cause of malady X. The hope and expectation of this research was that once the cause of malady X had been found, a cure could be formulated. The further hope and expectation was that, once a cure for malady X had been formulated and appropriated, the symptoms of malady X would no longer appear in any individual's fifth decade, and people would be able to go on living in the plateau phase of life for hundreds of years, just as they did back in Genesis.
Having clarified the fundamental essence of the longevity problem, it is possible to correct another common misconception. This is the idea that killer diseases such as cancer and cardiovascular disease are mankind's primary health problems today. In actual fact, malady X is the primary health problem.
Cancer and cardiovascular disease are, for the most part, diseases of "old age." That is, they prey on individuals already weakened by malady X. The implication is that the incidence of cancer, cardiovascular disease, and all other "old age"-related diseases will dramatically decline once the cure of malady X has been appropriated.
Note that the converse is not true. Even if total cures for cancer and cardiovascular disease were to be found, people would still continue to die of malady X within a few decades of 75 years, due to diabetes, or Alzheimer's, or pneumonia, or…
A cure for malady X is clearly, by far, the most pressing medical need today. All other diseases combined pale in significance relative to the misery and suffering caused each year by malady X.
The magnitude of the problem of malady X and the urgency of its solution has long been recognized. But finding a cure for malady X has proven to be no easy task. Despite billions of dollars spent on research, modern science is presently at a complete loss regarding how human life spans might ever be significantly increased beyond 100 years. Some well-respected scientists even claim that it is a fundamental impossibility.
Leonard Hayflick, an expert on aging at the University of California, San Francisco, denounced what he called "outrageous claims" by some scientists that humans are capable [of] living well past 100 years."Superlongevity," he said, "is simply not possible."[10]
The apparent intractability of the problem is underscored by consideration of present life span statistics. Despite a current global population of over seven and a half billion people—implying in excess of one hundred fifty thousand deaths per day worldwide (i.e., fifty five million deaths per year)—a life span in excess of 120 years is still a rare and remarkable event, and not one verifiable case of any individual living past 130 years of age has ever been found in modern times.
The difficulty of the problem is further emphasized by the fact that, while the scourge of reduced longevity has been with us for over five thousand years, no one in all that time has been able to discover how to do anything about it.
The biggest difficulty for the modern researcher has been that everybody suffers from this disease today. Normally, a researcher studies a group of diseased individuals relative to a group of healthy individuals. In the case of malady X, there are no healthy individuals to compare to.
If even one individual were to have lived in modern times to, say, 150 years, that individual would surely have been the subject of intense scientific interest. The interest would have focused around the question of what factor or factors had allowed that individual to live so long. Every effort would have been made to isolate factors in that individual's experience which had differed from everybody else, with the expectation that one or more such factors would be found to be responsible for the difference in longevity observed.
But we have no such individual or group of individuals to compare to today. Everybody is afflicted with malady X. The search for differences has no subjects from which even to begin.
Today, that is.
The search for a cure for malady X does have a few subjects to work with from the distant past, if we are willing to believe Genesis: Adam, and Noah, and Arpachshad, and Peleg, and Abraham, for example. Genesis tells us plainly that these men all enjoyed life spans well in excess of 150 years. Is it inappropriate or silly to try to isolate one or more factors in their experience which may have differed from our experience today?
All investigators admit that the problem of how to extend human life spans is one of extreme difficulty. Reliable data from subjects living beyond even 150 years—the sort of data one really needs to have any serious hope of cracking the problem—cannot be obtained today. Many researchers have already spent much time groping about in the dark for some clue to the mystery of human longevity. Unfortunately, they have nothing to show for their efforts. Millions of individuals continue to die each year, most before they have lived even 80 years, as has been the case for thousands of years.
Only one soft ray of light transgresses this blackness. It glimmers unobtrusively but faithfully from a lone window which looks out dimly upon an ancient world where thousands of multicentenarians once worked and played. We suggest the time may have come to take a careful look through this window. It seems to be our only possible hope. And perhaps it was put there for this very purpose.

The present work breaks with other contemporary scientific research on aging in its attitude toward the Genesis life span data. These data are commonly held to be mythological by contemporary researchers. The attitude toward these data underlying the present work is opposite to this. We hold these data to be valid, accurate, historical observations of actual life spans of real individuals.
This attitude is neither arbitrary nor religiously biased. The idea that these data are mythological or otherwise concocted cannot be retained by any scientist who has actually worked with them. These biblical data display certain features which are impossible to explain in any other way than that they are valid historical observations. This property will become increasingly clear as we proceed through this book. For now, we simply point out that the basically historical nature of these life span data is already strongly implied by their intimate association with key biblical chronological data. Many of these life span numbers are recorded together with birthdate numbers which are used directly in the construction of the chronology of biblical history stretching back to Adam. When this biblical chronology is checked using radiocarbon dating, it is found to be remarkably accurate. For example, the biblical chronology date for Noah's Flood is 3520±21 B.C., and the corresponding radiocarbon date is 3525±12.5 B.C.[11] This is probably the most secure and precise date humankind possesses for any historical event of such remote antiquity. The biblical chronological numbers are demonstrably historical. Would it not be odd if the closely associated life span numbers were mythological?
Once the biblical life span data are accepted as historical and reliable, they automatically become the focus of our research interest. They do so because they report on a unique, real-life, natural "experiment" which displays a pronounced life span effect in humans. This is the only experimental evidence we have that maximal human life spans can be meaningfully extended. It is essentially certain that this "experiment" will never be repeated, not least because it covers many generations over thousands of years. Thus, not only are the biblical data the sole experimental data we presently have which show anything of interest regarding human longevity, but also they are almost certainly the sole experimental data of the sort on humans we will ever have. Furthermore, experimental data displaying any evidence for extension of the present human life span are of extreme interest. But the Genesis longevity data go far beyond this, giving clear evidence of more than a tenfold increase in human life spans. Obviously, the biblical data make easy claim to our entire attention.
The biblical life span data of interest to the present study are shown in Table 2.1. The ages at death are taken from the Bible, from the verses shown in the "Bible Reference" column.
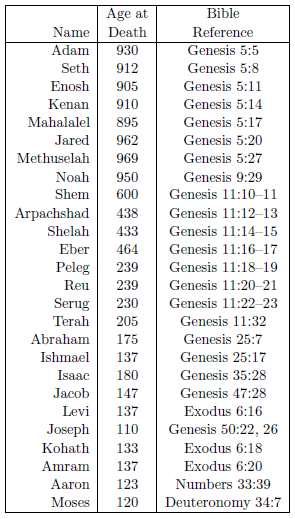 |
Though this list is comprehensive, it is not exhaustive. To begin with, biblical individuals with anomalously low life spans, such as Enoch (Genesis 5:24), Lamech (Genesis 5:31), and Nahor (Genesis 11:24–25) have been excluded. Secondly, biblical females have also been excluded. Males and females have different average life expectancies, so they need to be treated separately. There are significantly more life span data points for biblical males than there are for biblical females, so the present study focuses much of its attention on male life spans, both ancient and modern. Finally, no attempt has been made to add data to the list after Moses. Such data are of limited interest in the present study. They show mainly a continuation of the 75-year average life span which, on the basis of Psalm 90 ("A Prayer of Moses the man of God"), was already operative by the time Moses died.
Despite these exclusions, the list presents a remarkable dataset. Its 26 data items clearly capture the past decline in human longevity.
In the first part of this present study, the Table 2.1 life span data are everything. Understanding why they declined as they did is the goal. It is not sufficient simply to observe that life spans were longer in the past. The goal is to discover why they were longer—what physical, material agent(s) caused human life spans to shorten. The goal is to elucidate a cause and effect relationship. To accomplish this, knowledge of how life spans changed with time is needed. Thus, to make full use of these life span data, it is necessary to attach a unique time to each data item, specifying when in history that particular life span applied. This can be accomplished by assigning proper calendrical birthdates to each of the individuals listed in the table. If it is then assumed that each of the individuals shown in the table died of malady X, then the life spans of these individuals can be used as an estimate of the malady X-specific life expectancy when they were born. This will allow a graph to be constructed of life expectancy versus time for the ancient past, which is what is needed to attack the problem of the cause of reduced human longevity quantitatively.
Table 2.2 shows the needed birthdates. These dates have been computed from a combination of both biblical and extra-biblical chronological data according to the principles of the modern discipline of biblical chronology. The "Chronology Numbers" column shows numbers which are needed in the computations of the birthdates, and the "Bible Reference" column shows where these numbers were obtained.
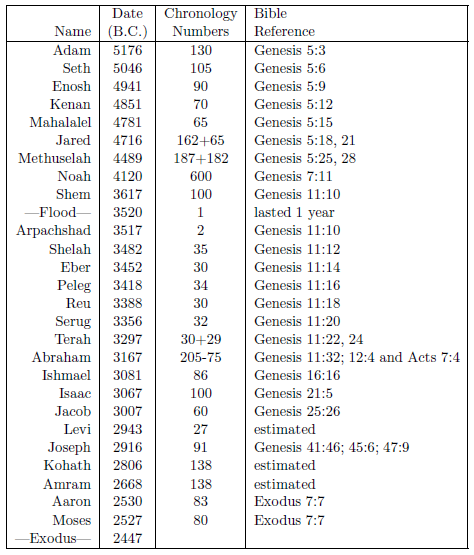 |
The table includes two chronological reference points: Noah's Flood at 3520 B.C.[12] and the Israelite Exodus from Egypt at 2447 B.C.[13] The date of the Flood is used as the reference point for most of the dates in the table, but chronological continuity, provided by recording the age of the father at the birth of the son, is lost following Jacob, during the 450 years Israel was in Egypt, making the final six birthdates of the table somewhat more complicated to compute.
The birthdate of Joseph can be calculated using the three Bible references provided for Joseph in the table. These reveal that Joseph was 39 years old when Jacob was 130 years old. Thus Jacob was 91 years old at the birth of Joseph.
Levi's date of birth has been estimated from Joseph's date of birth by making use of the fact that Joseph was the ninth son after Levi. Natural child spacing tends to be about three years (compare Moses and Aaron, for example) yielding 27 years between Levi and Joseph. This simple means of estimation is called into question in this instance by the fact that four separate wives were involved. This potential complication has been ignored since, from the primary record found in Genesis 29 and 30, the births seem to have been consecutive rather than overlapping, and because even a crude estimate will suffice for the present purpose.
For calculating the dates of birth of Moses and his brother Aaron, the date of the Exodus must be used. Exodus 7:7 reveals that Moses and Aaron were 80 and 83 years old respectively when they confronted Pharaoh. This places the birth of Moses in 2527 B.C.
The birthdates of Kohath and Amram can only be estimated. Even a crude estimate will suffice here once again, because life spans were not changing very rapidly when Kohath and Amram lived. Kohath and Amram have simply been placed equally apart in the 413-year interval spanned by the father-son lineage, Levi–Kohath–Amram–Aaron.
Life expectancy is the length of time a person may be expected to live. Since the current study is concerned with human longevity, it is the life expectancy at birth which is of interest. For the current study, life expectancy at birth data are obtained by combining the age at death data from Table 2.1 with the birthdate data from Table 2.2. The result is shown in Table 2.3.
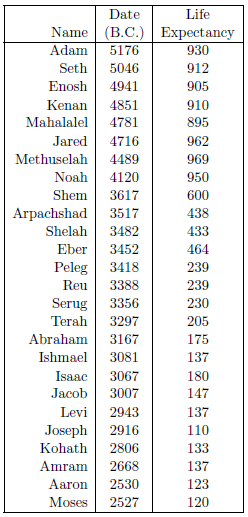 |
Normally, life expectancies are calculated by averaging ages at death from a large population. The biblical life span data provide only individual data points, not averages over many individuals. This is the same as taking the age at death of a single modern male to estimate the life expectancy at birth of modern males worldwide. While the average life expectancy is likely to be near 75 years at present, not all males die at age 75 years. Instead, ages at death of, for example, 62 years or 87 years are quite common. Individual ages at death today can easily differ from the average by plus or minus twenty years. The same is true of the biblical life expectancy data points. In fact, the average life expectancy at birth of the first seven individuals in the table, taken from a time interval during which life expectancies are thought to have been more or less stable, is 926 years, with a standard deviation of plus or minus 28.9 years.
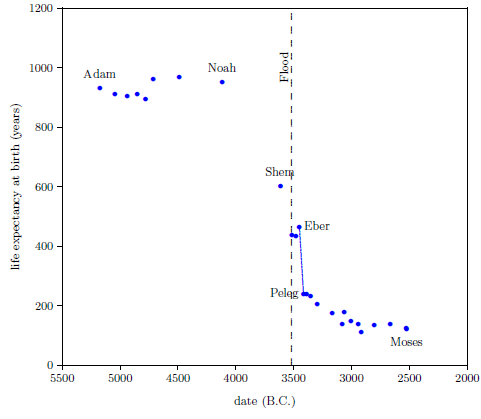
Figure 2.1 shows a graph of these biblical data. Take a good look at it. There is no graph in the whole world of greater practical humanitarian importance than this one. It is the key which unlocks the mystery of the cause and cure of "aging," as subsequent chapters will show.
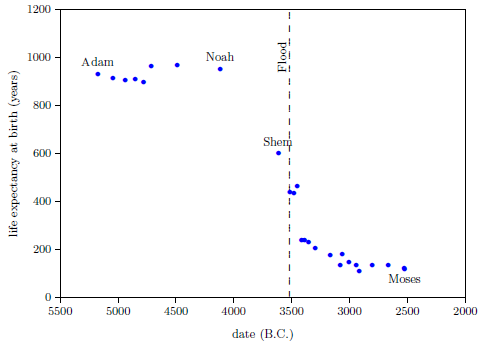 |

Before beginning to apply the data displayed in Figure 2.1 to solving the mystery of aging, it seems appropriate to point out two unique advantages from which the present research has benefited.
The first unique advantage has been the ability even to construct this graph.
To construct this graph, one has to have one's biblical chronology right all the way back to Adam. Most importantly, as will become increasingly clear, one has to have the date of the Flood right. The key to these prerequisites is the recognition that traditional biblical chronology has dropped out a full millennium in 1 Kings 6:1.[14] This discovery was made only in 1990. Extension back to Adam of the new biblical chronology resulting from that discovery was not completed until 1999.[15] Thus, though the biblical life span data of concern to this study are of great antiquity, ability to plot them accurately on the graph of Figure 2.1 is barely two decades old.
In addition to having biblical chronology right, the research reported in this book enjoyed a second important advantage. Figure 2.1 shows plainly that Noah's Flood is the dividing line between the short life span regime of the present day and the long life span regime of the ancient past. The reduced life expectancy data point corresponding to Shem, just before the Flood, may seem to be an exception to this, but Shem's reduced life span results from the fact that he lived most of his life in the post-Flood period. He was born 97 years prior to the Flood, and he died 503 years after the Flood.
The observation that Noah's Flood is the dividing line between the ancient and modern longevity regimes implicates Noah's Flood as the fundamental cause of reduced life spans today. Noah's Flood appears to have done something which subsequently shortened human life spans.
What exactly did it do? This is the fundamental question which must be answered in seeking to solve the cause of malady X.
Clearly, to have any hope of answering this question, an accurate idea of the nature of the Flood is needed. Indeed, the paramount importance of an accurate knowledge of the nature of the Flood to cracking the longevity mystery will become increasingly apparent in subsequent chapters. For now, the point to notice is that the true nature of the Flood was discovered only in 1997.[16]
Thus, both of the ingredients needed to make quantitative, scientific sense of the biblical life span data—a correct biblical chronology and a correct understanding of the nature of the Flood—have become available only since 1998.
These two unique advantages did not guarantee, of course, that this ancient mystery could, at long last, be solved. But they did make it perfectly clear that the time had come for a new, all-out assault on the problem.
The Figure 2.1 graph is a powerful research instrument. Consider its application in the following three cases, each dealing with common theories of "aging."
Perhaps the simplest lay theory of "aging" is that the human life span is determined by God in ways that cannot be understood or ascertained by humankind. This denies any natural cause of "aging," which immediately yields the corollary that scientific investigation into the matter is useless.
The biblical life expectancy data argue strongly against this theory. They show that human life spans declined in a fairly smooth way from roughly 925 years on average before the Flood to roughly 75 years on average following the Flood. This smooth decline took more than a thousand years to settle out to the currently active average life span. If the human life span is fixed by God, then these data require that God performed numerous miracles, continuously readjusting human life spans for more than a thousand years following the Flood. This seems severely contrary to what we learn of the nature of God's supernatural activity elsewhere in the Bible. Based on the miracles we read about in the Bible, such as the conversion of water into wine (John 2:1–11), or the calming of the sea (Luke 8:22–25), or the iron axe head which was made to float on water (2 Kings 6:1–7), we expect miracles to be generally evidenced as point-in-time suspensions of the natural order, not as innumerable slight adjustments of the natural order.
Meanwhile, we expect natural processes to change smoothly with time. For example, the temperature of a bowl of hot water naturally changes in a smooth progression from hot to cold with time (Figure 3.1).
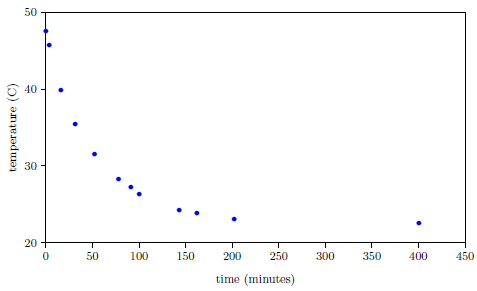 |
Furthermore, the change in temperature of the hot water is more rapid at first, and slows as it nears room temperature. This property is seen in the biblical life span data as well; the rate of change of life spans is rapid immediately following the Flood, and slows as the present value near 75 years is approached.
The biblical life span data reveal that life spans declined in a natural way following the Flood, implying that some natural cause was responsible for this decline.
A second lay theory is that pre-Flood longevity was due to a water vapor canopy which enveloped the earth prior to the Flood. This canopy was supposedly suspended above the atmosphere before the Flood, but it condensed and fell to the earth as rain at the time of the Flood, thereby contributing to the Flood's forty days and nights of rain.
This vapor canopy theory was popular a few decades ago with some people who regard Genesis as historical. Today, it is generally discredited. A scientific explanation of how such a canopy of water vapor might be kept in place is lacking. What would keep the water molecules from mixing with the rest of the atmosphere? Notice that the atmosphere today does a very good job of mixing all of its constituents together. We do not find separate layers of oxygen, nitrogen, carbon dioxide, water vapor, or any other gas. Other serious scientific problems a vapor canopy introduces include greenhouse heating of the surface of the earth and inordinate heating of the atmosphere at the time of the Flood due to the heat of condensation of water vapor and the conversion of gravitational potential energy to heat energy which would result from collapse of such a canopy. The only reason for bringing it up here is to show the power of the biblical life expectancy data for falsifying mistaken theories of aging.
The canopy is credited by its adherents with prolonging life prior to the Flood, usually in one of two ways. The first is through attenuation of hypothetically harmful radiation from space. Some versions of the theory cite ultraviolet rays from the sun, others cite cosmic radiation. The second is through enhanced atmospheric pressure due to the weight of the vapor canopy on the atmosphere.
Both of these versions of the canopy/longevity theory are immediately falsified by the biblical life expectancy data. To see this, notice that any attenuation of harmful radiation would immediately have ceased upon collapse of the canopy at the time of the Flood. Similarly, atmospheric pressure would have changed suddenly and completely upon condensation of the canopy at the time of the Flood. Thus, human life spans should have changed to their post-Flood value suddenly and completely at the time of the Flood. But the biblical data show us that life spans did not change suddenly and completely at the time of the Flood. Rather, they took more than one thousand years to complete their change from the pre-Flood value of 925 years to the present value of roughly 75 years.
One does not need an extensive background in science to see that the idea that a water vapor canopy was responsible for pre-Flood human longevity cannot be correct. The biblical life expectancy data alone are sufficient to yield this conclusion.
A broad spectrum of scientific theories about "aging" falls under the general umbrella of "evolutionary." The central idea in these theories is that "aging" is a by-product of evolution.
One of these theories suggests, for example, that all evolution needs is propagation of the species, and once this function has been fulfilled, an organism is best gotten rid of so it doesn't use up valuable resources. Thus, evolution has arranged for organisms to be discarded once their reproductive task has been completed.
Notice that the entire category of such theories is falsified by the biblical life expectancy data. The idea that a species' longevity is somehow determined by its evolutionary history—specifically, that humans live to 75 years on average because they have been somehow programmed by evolution to do so—cannot be true, because the biblical life expectancy graph shows that humans lived in excess of 900 years only a few thousand years ago. The biblical life expectancy data, in fact, falsify all theories of human longevity which hold death within a few decades of 75 years to be a pre-programmed biological necessity.
Evidently, what we call "aging" today really has nothing to do with evolution at all. The biblical life expectancy graph implies instead the radically new idea that what we call "aging" today has everything to do with catastrophe-occasioned disease.
Very many theories have been advanced in an effort to explain why humans "age" and die the way we do. Theories multiply in science whenever none of them really works. The biblical life expectancy graph easily falsifies many existing theories, clearing the fog, while pointing in a totally new direction.

Years of contemplating the nature of human "aging" in light of the biblical life expectancy data led ultimately to the conjecture that malady X must be a nutritional deficiency disease. This conjecture rapidly became the central hypothesis.
The human body is made up of trillions of microscopic cells. Each cell can be thought of as a very complex and busy city, part of a vast empire (the body). Each moment, raw materials flow into these busy cities, and, together with some waste, many finished products necessary to the overall growth, function, and maintenance of the empire flow out.
Among the raw materials flowing into these cities each moment are some which can be obtained only from outside the boundaries of the empire. For example, the human body cannot manufacture oxygen. The human body gets oxygen by breathing it in from the atmosphere. Many of these raw materials are absolutely vital to the cities—the cities cannot produce necessary finished products without them. If the supply of any one of these vital raw materials is halted for any reason (for example, lack of oxygen due to asphyxiation), then production of one or more vital finished products ceases. The health of the empire then suffers and, if the lack of this vital raw material persists long enough, the empire eventually disintegrates (i.e., the body dies).
On the list of vital raw materials needed by our bodies are such things as oxygen, water, fats, amino acids, certain minerals—calcium, phosphorus, sodium, potassium, chloride, magnesium, iron, copper, iodine, and many others in minute amounts—and a curious assortment of just over a dozen organic substances we call vitamins. If for any reason the cells of the body are unable to obtain one of these vital substances, then a deficiency disease results.
The most common cause of deficiency diseases in humans is inadequate diet. The essential raw material is simply not being taken into the body. But there are other possible causes, such as a faulty digestive system resulting in inadequate absorption of an essential raw material once it has been ingested, or combination of the essential raw material after ingestion with some other chemical and subsequent elimination of the compound from the body.
Of the list of essential raw materials needed by the body, the vitamins are of particular interest in the present context. Malady X appears to be a vitamin deficiency disease.
Scurvy is an example of a vitamin deficiency disease. It results from a diet deficient in vitamin C.
Before it was understood that scurvy is a deficiency disease, scurvy was a common disease of mariners. Vitamin C is abundant in fresh fruits and vegetables, so most of us get plenty of it in our normal diet each day. Vitamin C is easily subject to destruction by oxidation, however, so vitamin C levels decline in fruits and vegetables following harvest. Upon prolonged storage, vitamin C levels in fruits and vegetables become inadequate to meet human dietary requirements for this substance. The difficulty of providing mariners with fresh produce on long sea voyages inevitably resulted in many cases of scurvy.
Long before vitamin C was discovered, a number of individuals began to understand that scurvy could be prevented by a diet containing adequate fresh fruits and vegetables. Ways were sought, and eventually found, to protect the anti-scurvy property of lemons by concentrating and preserving the juice. Early in the 1800's, the British navy adopted regulations requiring daily consumption of lemon juice, bringing the scurvy plague to an end in the British navy. Eventually, this simple remedy was adopted by commercial vessels as well. The substitution of cheaper lime juice for the original lemon juice led eventually to the slang designation of British sailors as "limeys."
The actual anti-scurvy factor in fresh fruits and vegetables—the vitamin C molecule—was isolated, and its structure determined (Figure 4.1), only about ninety years ago.
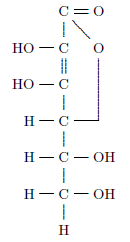 |
Vitamin C is a relatively simple organic molecule, and it is chemically similar to the physiologically ubiquitous molecule glucose, but the human body is unable to synthesize it. This molecule is vital to human health. Without it, connective tissues between cells degenerate. This results in a complex of symptoms at the whole-organism level. Most conspicuously, blood vessels become so weak that hemorrhage results, and teeth lose their strength and become diseased.
Adult patients suffering from scurvy complain of weakness, pains in their legs, swollen and bloody gums and hemorrhages. Examination discloses petechiae, chiefly about the hair follicles of the lower extremities and sometimes brawny, tender thighs. All of these features are due to hemorrhage…Weakness is usually the first thing complained of by persons suffering from vitamin C depletion. Fatigue, palpitation and breathlessness are also common. The patients dislike to stand or walk and often affect a rather characteristic standing position with their legs slightly flexed. The complexion is pallid and dirty looking. Gingivitis occurs, followed by loosening of the teeth, a consequence of resorption of the alveolar bones and infections about the teeth and is accompanied by a foul breath. Other signs of scurvy are hematuria, bloody diarrhea, nasal hemorrhage or hematomas about the jaw or bones of the lower extremities.[17]
Vitamin C is needed only in minute amounts—about one ten-thousandth of our daily food intake on a dry weight-per-weight basis. This miniscule daily requirement relative to the bulk diet is characteristic of all the vitamins. In the case of vitamin D, the amount needed is roughly one five-millionth of our daily food intake. But though so little is needed, this small amount is absolutely essential. Without it, our cells lose their ability to carry out their jobs, and, eventually, a complex of whole-body symptoms—a deficiency disease—develops.
Our central hypothesis is that human "aging" is a vitamin deficiency disease resulting from dietary insufficiency of a previously unknown vitamin which was made globally deficient by the impact of Noah's Flood on earth's environment.
For many years, before the identity of this vitamin was known, we dubbed it simply "vitamin X." It was then logical to call the disease one gets when one is deficient in vitamin X, "malady X" (malady X-bar). The "bar" signifies negation. "Malady X" means "the disease due to not X" or "the disease due to lack of X." In this nomenclature, scurvy is "malady C," beriberi is "malady B1," rickets is "malady D," and "aging" is "malady X."
Early support for this hypothesis was easily gleaned by comparing and contrasting malady X with scurvy.
Notice, first of all, that like scurvy, malady X ("old age") exhibits itself as a complex of whole-body symptoms: skin loses its elasticity, muscles weaken and decrease in size, hair loses its color and thins out, bones become brittle, eye lenses stiffen…
These are very diverse symptoms, yet they show up together in "old age." One could suppose that they are all caused by independent physiological malfunctions of one sort or another, and that these independent malfunctions are all synchronized by some sort of master biological time-clock. But much simpler is the idea that these diverse symptoms are simply varied macroscopic manifestations of a single missing essential molecular component at the microscopic, cellular level—just as is the case with scurvy.
Not only is there a complex of whole-body symptoms in both cases, but some of the particular symptoms of "old age" also show striking similarities to symptoms of scurvy.
Aschoff and Koch were greatly impressed with the similarity of the scorbutic [scurvy] lesions to those in senility. The changes in cortical bone are difficult, if not impossible to distinguish. … In both conditions the bones are notably thin and rarefied, susceptible to fracture and defective in the ability to form a callus once fracture has occurred. …Westin interpreted the tooth lesions as similar to the atrophy of old age and said scurvy may be considered to hasten involution. In his cases the teeth showed the same resistance to caries that is seen in senility as well as the rarefaction common to advanced years.[18]
This demonstrates that vitamin C deficiency disease can produce precisely the same sorts of abnormal changes and injury to body tissues as those which are characteristic of "old age." Evidently then, at least some of the specific symptoms accompanying "old age" fall naturally within the vitamin deficiency disease category.
An apparent contrast between "old age" and scurvy is that only a small percentage of individuals in a normal population ever contract scurvy, while all individuals, if they live long enough, contract malady X.
This apparent difference is easily explained. Normal diets of most individuals supply them amply with vitamin C. Only the few individuals on deficient diets ever contract scurvy. In contrast, normal diets of all individuals have become seriously deficient in vitamin X as a result of the Flood. How this came about will be explained in detail in subsequent chapters.
Another similarity between "old age" and scurvy is that the time of onset can be varied. Prior to the Flood, men lived in excess of 900 years before they succumbed to "old age" and died. After the Flood, men contracted "old age" at younger and younger ages, until the present, much-diminished life span near 75 years was reached.
The time of onset of scurvy can be similarly varied:[19]
They found that less than 50 cc. of milk daily resulted in scurvy within thirty days, that 50 cc. delayed the onset of the disease until the seventy-fifth day and that 100–150 cc. of milk postponed evidence of scurvy for four months.
Milk is a poor source of vitamin C. Thus all of the animals (guinea pigs) referred to in the previous quote were subject to a vitamin C deficient diet. Those getting less milk got less vitamin C. Thus the time of onset of scurvy is seen to be directly related to the daily intake level of vitamin C in the diet.
This intake-dependent time-of-onset characteristic of vitamin deficiency diseases provides an explanation of the change in human life spans following the Flood which Genesis records. It implies that human life spans changed because the availability of dietary vitamin X changed. This could happen if vitamin X was somehow produced and metered out by earth's environment to all individuals globally in a rigidly fixed intake from the time of birth on. This leads to the conjecture that life spans diminished following the Flood because vitamin X, which was already somewhat deficient before the Flood (resulting in death due to "old age" at roughly 925 years), became increasingly scarce globally after the Flood so that, by 2500 B.C. (one thousand years after the Flood), vitamin X had dwindled to the seriously deficient level which characterizes it today. Subsequent chapters will demonstrate the success of this conjecture, explaining why vitamin X availability dwindled the way it did.
We are aware of nothing about "old age" which is inexplicable in terms of it being due to a nutritional deficiency disease resulting from lack of a previously unknown vitamin. This category of disease seems to provide a complete explanation of the facts in regard to "old age" and human longevity. To the best of our knowledge, this is true of no other category of known diseases.
Just as scurvy results from a diet deficient in vitamin C, so "old age" results from a diet deficient in vitamin X. Vitamin C is the anti-scurvy vitamin. Vitamin X is the anti-aging vitamin.
The Flood somehow broke the natural supply of vitamin X. This caused human life spans to be reduced ultimately by more than a factor of ten relative to what they had been prior to the Flood.

Having come to understand that "aging" results from inadequate dietary vitamin X, the research goal becomes to identify the chemical compound—the molecule—corresponding to vitamin X. Once vitamin X has been identified, the expectation is that restoring it to human diets will cure "aging."
But identifying the compound corresponding to vitamin X is no easy task. There are nearly an infinite number of chemical compounds to choose from. It will clearly not do to try random guesses. Rather, it is necessary, like good detectives, to gather every bit of information we can about vitamin X, and then to use these clues to try to deduce the molecular identity of vitamin X. The current chapter begins this process by revealing two important properties of vitamin X.
The biblical life expectancy data display a fairly uniform, natural decline from Noah to Moses. The only real irregularity in this uniform decline, allowing for normal scatter in the data points, is a sudden drop in life spans between Eber and Peleg. This irregularity is highlighted in Figure 5.1 by a line connecting the Eber and Peleg data points.
 |
This sudden drop has been used by some to argue for gaps in the Genesis chapters 5 and 11 genealogies from which many of these life expectancy data points have been taken. The argument, in this case, is that it is unlikely that life spans would have changed so dramatically in a single generation—from 464 years for Eber to just 239 years for Peleg—when they changed little in both the preceding two generations and in the following two generations.
In point of fact, there are no generations missing between Eber and Peleg. The sudden drop in life spans between Eber and Peleg is real history. That is, it really was the case that Peleg was Eber's direct, first-generation son. That this is the case will become increasingly clear in subsequent chapters.
From a modern perspective, this yields a curious result. It means that the son, Peleg, died of "old age" nearly two centuries before his father, Eber, died of "old age." Said another way, the father was yet relatively youthful when the son died of "old age."
How is such a thing possible?
Such a thing is possible if and only if vitamin X has a long biological half-life.
Biological half-life is a measure of how long a substance tends to remain in the body before it is eliminated. The biological half-life can be broken down into half-lives for individual organs, like the heart, or the kidneys. Different organs can have very different half-lives. For example, it is possible for a compound to clear from the kidneys very rapidly, but to be retained in heart muscle for a long time. It would then have a short half-life in the kidneys and a long half-life in the heart.
The overall biological half-life of a substance in the human body can be measured by giving a person a small amount of that substance and measuring the amount remaining in the body (all organs and tissues) after an elapsed time. The biological half-life is the time it takes for just one half of the original amount to be remaining.
Sodium, which we get from common table salt, sodium chloride, is used in tissues throughout the body. It has a relatively short biological half-life of just 29 days. Calcium, on the other hand, tends to get tied up in bone, and has a relatively long biological half-life of 49 years.[20]
Imagine two modern individuals, Bob and Tom, of the same age—say ten years. If neither is given supplemental intakes of vitamin X, then both will die of vitamin X deficiency disease ("old age") within a few decades of seventy-five years. We know this with fair certainty because billions of individuals have corroborated it since the the time of Moses.
Now imagine that Bob is given supplemental intakes of synthetic vitamin X so that he gets all the vitamin X his body needs from age ten on, while Tom remains at a natural, present-day (deficient) level of vitamin X. Tom will die of "old age" within a few decades of 75 years. But not so Bob. Bob's body will be healed of "old age" and protected against "old age" by vitamin X, just as a person receiving an adequate diet of vitamin C will be healed of and protected against scurvy.
We see by this simple example that two individuals of the same age, living at the same time, can experience very different rates of "aging" depending on their respective intake rates of vitamin X.
Now imagine that Bob is given synthetic vitamin X for one year only, after which he, like Tom, receives only a natural present-day level of vitamin X. What will be the result?
If vitamin X has a biological half-life measured in days, like sodium, then the benefit to Bob of his year-long supplemental intake of vitamin X will be merely to increase his life expectancy by one or two years. Extra vitamin X can benefit Bob only while he has it in his body, and he will have it in his body for only a short while after he stops taking it, if it has a short biological half-life. But if vitamin X has a relatively long biological half-life, say 49 years, like calcium, then Bob would be expected to outlive Tom by a century or more. The reason for this is that, in the case of a long biological half-life, vitamin X continues to be maintained at high levels in Bob's body long after supplemental intake has stopped.
This is what happened in the case of Eber and Peleg. Since no means of artificially synthesizing vitamin X was available back at that time, Eber and Peleg were limited to only that intake of vitamin X which the environment naturally provided. Eber was born at a time when the amount of vitamin X in the environment was much higher than it is today. Sometime during the thirty-four years between the birth of Eber and the birth of Eber's son, Peleg, the amount of vitamin X in the environment declined dramatically. There is a very good reason why the amount of vitamin X in the environment declined this way, which will be discussed in a subsequent chapter, but, for now, notice merely that, as a result of this decline, the natural intake of vitamin X received by Peleg was always much less than that which his father Eber had initially received.
From Peleg's birth on, both Eber and Peleg were receiving the same, relatively low, natural intake of vitamin X from the environment. But even though they were both limited to the same natural intake rate of vitamin X from Peleg's birth on, Eber did not die of "old age" at the same time Peleg did. Eber lived on for several centuries after Peleg, his son, had died of "old age." Eber outlived his son Peleg by over two hundred years because Eber carried a higher level of vitamin X with him, in his body, long after vitamin X had declined dramatically in the natural environment.[21] This shows that the biological half-life of vitamin X must be relatively long—on the order of a century.
The long biological half-life of vitamin X is apparent in the Genesis life span data in many other instances than just Eber and Peleg. Notice, as a single additional example, that Shem, who accompanied his father Noah on the ark, outlived not only Peleg, his great-great-grandson, but also even Terah, Peleg's great-great-grandson (and father of Abraham). Shem, born before the Flood, died of "old age" only 25 years before Abraham—born 350 years after the Flood—died of "old age."
Clearly, vitamin X has a long biological half-life.
The environmental half-life is the time it takes for just one half of an original amount of a substance to be remaining in the environment after it has been added to the environment. As with biological half-life, the environmental half-life can be broken down into various compartments. For example, the environment might be broken up into atmosphere, hydrosphere, and biosphere compartments. Each of these compartments would have its own characteristic half-life for a given compound, and these half-lives could differ substantially from one another.
Vitamin X displays two environmental half-lives, a short one and a long one, implying its presence in two environmental compartments. The long half-life compartment is apparent from the slow decline in life expectancies between Peleg and Moses (Figure 5.1). This slow decline compartment will be discussed further in subsequent chapters. The short half-life compartment is apparent from the rapidity of the drop in life expectancy between Eber and Peleg. This drop in life expectancy requires that the environmental abundance of vitamin X dropped dramatically between Eber's birth and Peleg's birth. The time between these two births was just 34 years. Thus we learn that vitamin X in the natural environment can decline dramatically in a matter of a few decades or less. This is just another way of saying that vitamin X has an environmental compartment with a relatively short half-life—on the order of a decade or less.
Vitamin X has an unusually long biological half-life. It has at least two environmental compartments, one with a relatively short half-life and the other with a considerably longer half-life.

We are now able to begin to piece together how the environmental abundance of vitamin X changed as a result of the Flood. By focusing on the Eber–Peleg drop in life expectancies, we have already learned that the environmental abundance of vitamin X declined dramatically between Eber and Peleg. Noah's life span reveals another important change in the environmental abundance of vitamin X which was brought about by the Flood.
Noah's life span shows that the environmental abundance of vitamin X was initially caused to increase by the Flood. This may be seen as follows.
The dashed blue line in the top graph of Figure 6.1 represents Noah's life from birth until death. Notice that Noah lived the final centuries of his life in the post-Flood, post-Peleg regime, when the environmental abundance of vitamin X had dropped to low levels compared to the pre-Flood level. Noah lived his first 600 years before the Flood, and his final 350 years after the Flood. Roughly 250 years of Noah's life were lived in the post-Peleg regime. This seems to imply that Noah's life span should have been shorter than the average pre-Flood life span, when, in fact, it was longer.
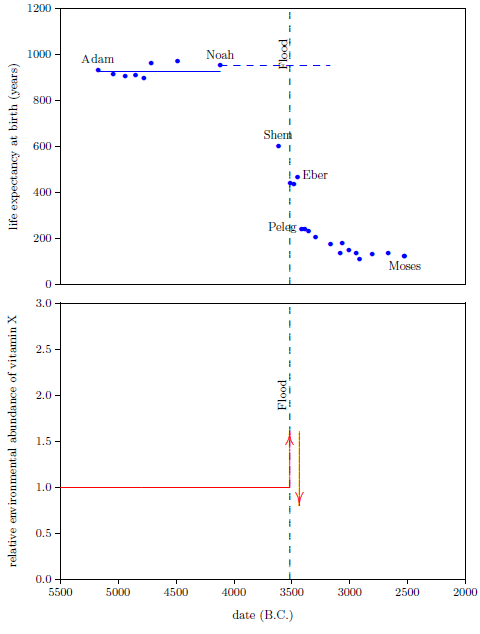 |
The average life span of the seven pre-Flood men preceding Noah in the biblical life expectancy data set is 926 years. This average is shown in the top graph of Figure 6.1 by the solid, blue line extending from Adam to Noah. Notice that Noah's life expectancy at birth data point lies above this line. This shows that Noah's life span was longer than average for a pre-Flood male.
How is this possible, given that Noah lived the final two and a half centuries of his life in an environment which was seriously deficient in vitamin X?
The long biological half-life of vitamin X is not a sufficient answer to this question. From the birth of Peleg on, life expectancies were dramatically shorter than they had been prior to the Flood. As just mentioned, Noah spent the last 250 years of his life in this post-Peleg regime. Even if the biological half-life of vitamin X were 200 years (and it is not that large, as will be shown in Chapter 11), by the time Noah died, his body concentration of vitamin X would have dropped by more than a factor of two relative to his pre-Flood body concentration. Thus his average body concentration of vitamin X during these post-Peleg years would have been less than 75% of what it had been pre-Flood. Said simply, even given a long biological half-life for vitamin X, Noah still would have suffered serious vitamin X deficiency during the post-Peleg years (though not nearly as serious as Peleg's vitamin X deficiency), and this should have shortened his post-Flood life span significantly. Notice that Noah's son, Shem, had his life span reduced from the pre-Flood average of 926 years to just 600 years as a result of living roughly 400 years in the post-Peleg regime. This yields a loss of 326 years of average pre-Flood life span per roughly 400 post-Peleg years. At the same rate, Noah should have lost roughly 200 years relative to the pre-Flood average life span. Instead, Noah lived 24 years longer than the average pre-Flood life span. Clearly, the long biological half-life of vitamin X is not sufficient to explain Noah's longer-than-average life span.
Noah's above average life span requires that Noah entered the post-Peleg years with a concentration of vitamin X in his body which was higher than the pre-Flood concentration. This higher concentration could have been obtained only after the Flood and before Peleg. Thus, the Flood must have caused the environmental abundance of vitamin X initially to increase.
The bottom graph of Figure 6.1 shows both the increase caused by the Flood (vertical arrow pointing up), and the sudden decline between Eber and Peleg (vertical arrow pointing down). The slope of the decline between Eber and Peleg is unknown, other than that it was steep. Keeping things simple, we have assumed that the drop was instantaneous, as the arrow depicts. The arrow is plotted midway between the births of Eber and Peleg.
From Peleg to Moses, life expectancies show a relatively gentle decline (Figure 6.2, top graph). This decline in life spans implies an underlying decline in the environmental availability of vitamin X, which we have illustrated in the bottom graph of Figure 6.2.
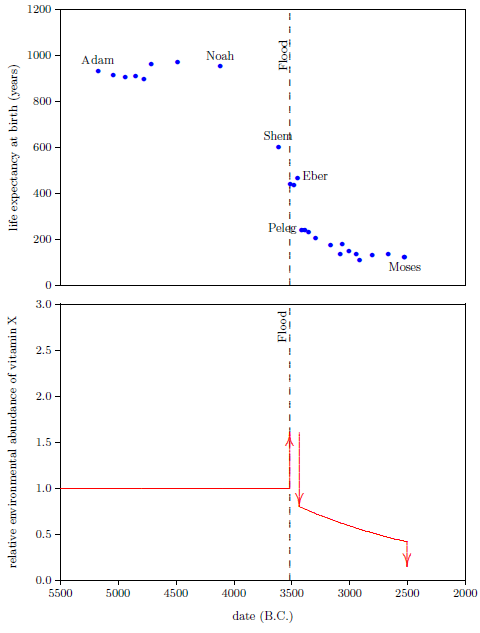 |
The decline in life spans is fastest initially, so we have chosen to represent the decline by a natural exponential decay. At this point, we don't know the initial height of the exponential, and we don't know how fast it should drop off. We have made initial guesses at these two parameters to get the representative exponential decline which is plotted in the bottom graph of Figure 6.2. These guesses will be mathematically refined in a subsequent chapter.
Notice that, as mentioned previously, this adds a second environmental half-life to vitamin X, one which is quite long, implying two environmental compartments.
Life spans appear to have rapidly dropped once again in the lifetime of Moses. In the first half of the tenth verse of Psalm 90, Moses notes that life spans had dwindled to roughly 75 years.
As for the days of our life, they contain seventy years,
Or if due to strength, eighty years,…
Moses himself lived 120 years, and his brother Aaron lived 123 years. Thus, there was another Eber–Peleg type of drop in life spans during Moses' lifetime. This drop appears to have resulted in the modern life span for humans, which has persisted now for more than 4,500 years. This final drop is indicated by the final red arrow. For simplicity, we have chosen to make this drop instantaneous once again. In real life, this final drop would almost certainly have been more gradual.
A date for this drop can be estimated as follows. Because Moses had to observe and record the drop, the latest time that persons could have begun dying of "old age" at 75 years on average is the same time Moses himself died of "old age" at 120 years. These persons would have been born when Moses was 45 years old. This places the latest date for the drop in 2482 B.C. The earliest time this drop could have happened, and Moses and Aaron still enjoy their recorded, significantly longer life spans due to the biological half-life of vitamin X, is the year following the birth of Moses. This is 2526 B.C. The best estimate, in this case, is the midpoint between earliest and latest possibilities, which is 2504 B.C.
We do not know, at present, how far the environmental abundance of vitamin X dropped at this point. This will clarify in subsequent chapters.
The environmental abundance of vitamin X breaks naturally into four distinct time periods. These are depicted in the bottom graph of Figure 6.3.
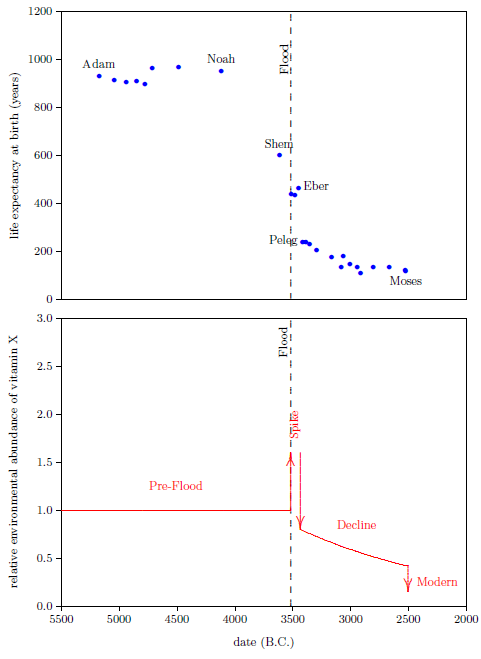 |
The first is the "Pre-Flood" time period. This is a steady state region, in which the environmental availability of vitamin X was constant and relatively high, though still somewhat short of dietary sufficiency. This region ended with the coming of the Flood.
The second is the "Spike" time period, initiated by the Flood. This was a relatively brief period during which the environmental abundance of vitamin X was higher than in any other time period. The Spike ended with a sharp reduction in environmental availability of vitamin X sometime between the births of Eber and Peleg.
The third is the "Decline" time period. Environmental availability of vitamin X, which was already seriously deficient at the beginning of this time period, slowly declined, becoming ever more seriously deficient during this time period.
The fourth is the "Modern" time period. It was ushered in roughly 2504 B.C. by a final rapid drop in life spans to the modern value near 75 years, which took place during the lifetime of Moses.

It is now possible to advance reasonable answers to three key questions.
The biblical life span data seem to imply that, while longevity varied widely in different periods of history, it was globally uniform at a given point in time. The biblical longevity data do not encompass a global population, of course, but they do extend over a large enough geographical area in the Middle East—including at least portions of modern Iraq, Turkey, Israel and Egypt—to make global uniformity seem likely.
Is there any way of explaining a point-in-time geographical uniformity of vitamin X given its large temporal variations?
Yes. The atmosphere is present globally, and it mixes rapidly relative to human life spans—just a year or two from pole to pole. If vitamin X were distributed by the atmosphere, then its consequences for longevity would be globally uniform at any point in time.
The atmosphere is made up of gases. The mixture of gases comprising the atmosphere is called air. Nitrogen and oxygen are the main gases in air, but there are many gases present in minor amounts as well. For example, nearly one percent of air is the gas called argon. Well known gases in air include ozone, carbon dioxide, and methane. Dozens more, not so well known, like carbon disulfide and nitrogen dioxide, might easily be mentioned. All of the gases in air, other than nitrogen and oxygen, are called trace gases. Point-in-time global uniformity would be explained if vitamin X were a trace gas or were somehow involved with a trace gas.
The idea that vitamin X is somehow involved with a trace gas provides an explanation of the Decline time period of the vitamin X environmental abundance graph. In the previous chapter (Figure 6.3), the Decline was shown lasting roughly 900 years. The Flood seems to have broken the fundamental source of vitamin X, but vitamin X did not drop immediately to zero following the Flood. Rather, following an initial spike, it slowly declined for 900 years. Involvement of a trace gas provides an explanation of this prolonged decline. Specifically, there is a component of earth's global environmental system which has this characteristic timescale. One thousand years is the order-of-magnitude timescale for the turnover of earth's oceans. Deep ocean water takes roughly a thousand years to move from the bottom of the ocean to the surface. A change lasting 900 years would be expected if the oceans acted as a reservoir for the conjectured atmospheric trace gas. Gases have a water solubility. Gas molecules, present in earth's oceans, are effectively cut off from the atmosphere. They can get out of the ocean and into the atmosphere only by diffusion through the ocean–atmosphere interface at the surface of the ocean. Gas molecules down deep in the ocean will be stuck in the ocean for hundreds of years waiting for the water in which they are trapped to move slowly up to the surface, where they can vent to the atmosphere via diffusion through the surface. The 900 years of the Decline time period can be readily explained as the time it took for the trace gas involved with vitamin X to empty out of the oceans into the atmosphere.
Said succinctly, though the Flood broke (i.e., shut down) the primary source of some atmospheric trace gas, the oceans continued to act as a source of this gas to the atmosphere until their supply had been exhausted roughly 900 years later.
All of this argues forcefully for the idea that vitamin X was distributed globally via the atmosphere, either as a trace gas or as some atmospheric product of a trace gas.
Notice that this also explains why the environmental abundance of vitamin X has a fast environmental half-life compartment and a slow environmental half-life compartment. The fast compartment is the atmosphere, and the slow compartment is the oceans.
The idea that vitamin X is a trace gas, while possible, is less attractive than the idea that vitamin X is a product of a trace gas. The reason for this has to do with the nature of the thirteen traditional vitamins and the nature of the atmospheric chemistry of trace gases.
To begin with, of the thirteen traditional vitamins, none are gases. Vitamin-likeness argues against vitamin X being a gas.
The atmosphere contains a lot of oxygen. Atmospheric chemistry tends either to break a trace gas down into smaller fragments, or to add a few oxygen atoms to a trace gas. Breaking a trace gas down into smaller fragments will yield yet lower molecular weight products, which will tend to be gases. Thus, vitamin-likeness favors the oxygen-addition process over the fragmentation process. Atmospheric chemistry argues for vitamin X being a small molecule. Atmospheric trace gases tend to be small molecules. Large molecules are too massive to have sufficient vapor pressure to be present in the gas phase. There is no sharp mass cutoff for presence of a compound in the atmosphere, but a molecular weight of 250 g/mole seems to be a reasonable dividing line above which few molecular substances are likely to be found in significant abundance in the atmosphere.
This small-molecule dividing line places vitamin X in the water-soluble vitamin category. Of the thirteen traditional vitamins, nine are water soluble and four are fat soluble. None of the fat-soluble vitamins has a molecular weight below 250 g/mole, while five of the water-soluble vitamins have molecular weights below this cutoff. Thus, vitamin-likeness argues for vitamin X to be a small, water-soluble molecule.
Does the atmospheric chemistry, oxygen-addition process tend to produce water-soluble products?
Yes. Oxidation of trace gases tends to yield small acids as stable end products. Small acids are notoriously water soluble.
Are any of the small water-soluble vitamins acids?
Yes. In fact, of the five water-soluble vitamins having molecular weights below 250 g/mole, four are acids.
Thus, the idea that vitamin X is a small acid, an oxidation product of an atmospheric trace gas, is very attractive.
This idea immediately answers the third question. Because small acids are soluble in water, acids produced in the atmosphere are rapidly washed out of the atmosphere by rain once they have been formed. This means that they are naturally present in rainwater, in freshwater ponds and lakes, and in rivers. Clearly, vitamin X would have entered human diets in antiquity via the water that humans drank.
Methanesulfonic acid (MSA) is an example of a small organic acid which is produced in the atmosphere by oxidation of a trace gas.[22] The trace gas in this instance is the extensively studied DMS (dimethyl sulfide).
DMS is naturally produced in ocean surface waters from DMSP (di-methylsulfoniopropionate), which is a metabolite of some marine algae. In the atmosphere, DMS reacts with the hydroxyl radical (OH•) or the nitrate radical (NO3•) resulting in oxidation to a variety of products, including MSA.
MSA concentrations in glacier ice are generally around 10 micrograms per liter. Thus, use of rainwater for drinking today (not recommended due to ubiquitous pollutants in modern air) would naturally supply human diets with 20 micrograms or more of MSA per day. This is not to suggest that MSA is desirable in human diets today, but rather to show that nature is capable of producing and supplying to the human diet compounds in vitamin-like quantities via an atmospheric trace gas.
The idea that vitamin X is a small acid, an oxidation product of an atmospheric trace gas, is compelling. It provides satisfying explanations of the natural synthesis, distribution, and dietary presence of vitamin X.

Having deduced that vitamin X seems likely to be an oxidation product of an atmospheric trace gas, the next strategic goal is to try to identify the trace gas, as an aid to identifying vitamin X. There are, unfortunately, still way too many trace gases (giving rise to a plethora of oxidation products) to make random guessing a feasible research strategy. It is necessary to make use of every clue once again. But this time, it is knowledge of the nature of the Flood, not the properties of the trace gas, which yields the vital breakthrough.
We are looking for that particular trace gas which is the precursor to vitamin X. This precursor gas was being supplied to the atmosphere pre-Flood in much greater abundance than it is at present. The Flood somehow broke the supply to the atmosphere of this precursor gas.
How might the Flood have broken the supply of an atmospheric trace gas? The answer to this question seems likely to provide the all-important clue to learning the identity of vitamin X. Thus it appears that the cure to human "aging" requires "only" a thoroughgoing understanding of the true nature of Noah's Flood. Fortunately, we have such an understanding.
Reword that. In actual fact, good fortune has had little to do with it. One of the authors (GEA) has spent decades of his life uncovering the truth regarding Noah's Flood. He has found that mainstream academia is seriously mistaken about the Flood. Modern academicians seem to regard it as essentially mythological, if they bother to think about it at all. And he has found that individuals holding out against the view that the Flood was a myth (usually for theological reasons) are generally seriously mistaken in their understanding of the true nature of the Flood. In consequence, none are very well positioned to be of much help in regard to the present question. It was, therefore, necessary to solve the mystery of the true nature of Noah's Flood before the mystery of "aging" could hope to be solved.
The important thing to know about the Flood in the present context is that it was hemispherical in extent. It resulted from the waters of the southern hemisphere flowing up into the northern hemisphere and heaping up to great depth there for some months. Why this happened—the geophysics behind it—is explained in detail in the book, "Noah's Flood Happened 3520 B.C.,"[23] so we will not go into detail here. The important thing to notice at present is only this: heaping the planet's oceans up in the northern hemisphere will expose the sea floor in the southern hemisphere. In fact, the sea floor in the high southern latitudes surrounding Antarctica was entirely uncovered for at least 110 days during the Flood.
Sea floors are hidden from our eyes. As a result, they are not items of common knowledge. The important thing to know about them at present is that they are covered with sediments. The average depth of the sediments covering the floors of the world's oceans exceeds a quarter of a mile.
Sea floor sediments are cut off from atmospheric oxygen. In consequence, they are anaerobic. Anaerobic microorganisms in organic-rich sediments produce organic gases as byproducts within those sediments. Methane is a major, well known byproduct gas of anaerobic digestion. Other anaerobic byproduct gases include, for example, carbon dioxide, hydrogen, nitrogen, and hydrogen sulfide.
We suggest that the source of the atmospheric trace gas which is the precursor to vitamin X was anaerobic digestion of organic detritus within the sea floor sediments of the southern oceans. Steady state diffusion of molecules of this trace gas from the sediments to the overlying ocean water is what the Flood broke. The Flood broke this steady state diffusion by removing the great depth of water which normally covers the sediments. This reduced the pressure these sediments are normally under by hundreds of atmospheres. (Each ten meters of water depth adds another atmosphere of pressure, and the average depth of the oceans is 3800 meters.) The inevitable result was rapid release (outgassing) to the atmosphere of the anaerobic byproduct gases contained within these sediments.
The Flood broke the steady state diffusion of anaerobic gases from the sediments to the bottom waters of the southern oceans by emptying southern sediments of their gases during the year of the Flood.
This explains why the Flood was followed by the Spike. The Flood released thousands of years' worth of vitamin X precursor gas from the sediments into the atmosphere all in one year. The result was decades of abnormally high vitamin X production in the atmosphere during the Spike.
When the atmosphere had finally managed to cleanse itself of vitamin X precursor (and the load of other sea floor gases), only precursor diffusing out of the water of the oceans remained to supply vitamin X. This explains the Decline.
When the oceans' supply of precursor ran out, life spans dropped to near 75 years, initiating the Modern time period.
The sediments of the southern oceans were emptied of their store of precursor gas by the Flood. This vital source of precursor has so far not recovered in five and a half thousand years.

It seems clear that the vitamin X precursor gas was predominantly produced in the sediments of the southern oceans. If its production had been spread out over the sea floor of the entire planet, then the supply of precursor gas would have been reduced only roughly in half by the Flood, and it seems unlikely that reduction of the environmental abundance of vitamin X by a factor of two could have resulted in the observed reduction of life spans by more than a factor of ten. Furthermore, the supply of precursor gas should then have shown some recovery over the past five and a half thousand years, with a correspondingly noticeable recovery in life spans, due to a natural recovery of precursor gas within those sediments which had been only slightly depressurized by the Flood. But no such recovery in life spans has been seen. Thus, available data seem to favor the idea that nearly the entire source of precursor was destroyed by the Flood.
This focuses attention on the southern sea floor. What is so special about it? Why did depressurization of just the southern sea floor destroy nearly the entire source of vitamin X precursor gas?
Modern ocean properties suggest answers to these questions. Phosphate and nitrate are broadly important for biological productivity. Lack of one or the other of these nutrients often limits biological productivity within the global marine environment. But the surface waters of the oceans surrounding Antarctica are especially rich in phosphate and nitrate. Figure 9.1 shows a world map of annual mean sea-surface phosphate.[24] Figure 9.2 shows the same thing for nitrate.[25] The existence of both of these nutrients together in high concentrations in the ocean surface waters surrounding Antarctica more or less guarantees accumulation of organic-rich sediments on the sea floor surrounding Antarctica.[26] Indeed, it seems appropriate (if a trifle prosaic) to regard the sea floor surrounding Antarctica as the planet's septic tank.
 |
 |
The concentration of phosphorus in surface waters surrounding Antarctica makes the planet's septic tank a rich depository of phosphorus-laden biomolecules. The presence of this rich accumulation of biophosphorus instigates the hypothesis that the vitamin X precursor was a phosphorus gas byproduct of anaerobic digestion within pre-Flood sea floor sediments.
This hypothesis seems to encounter an immediate difficulty. Phosphorus is well-known not to have any significant trace gases. In fact, phosphorus contrasts with other elemental environmental cycles in not having any significant atmospheric component. For example, the nitrogen cycle has an obvious atmospheric component in the form of N2 gas. Similarly, the oxygen cycle has an obvious atmospheric component in the form of O2 gas. The sulfur cycle has the already-mentioned DMS as a significant atmospheric component. DMS moves 15–25 million metric tons of sulfur from the oceans into the atmosphere each year.[27] The environmental iodine cycle has methyl iodide (MeI) as an atmospheric component. The environmental carbon cycle has carbon dioxide (CO2) as an atmospheric component. … But there is no atmospheric component in the case of phosphorus.
We suggest that this difficulty is apparent only. If one supposes that the phosphorus cycle is presently operating in steady state—that it has been behaving for time out of mind just as it is observed to be behaving at present—then the presently observed lack of an atmospheric phosphorus component leads immediately to the conclusion that it is the nature of the phosphorus cycle to be without any significant atmospheric component. But as soon as one knows the truth about the Flood, one knows better than to suppose that the phosphorus cycle is presently operating in steady state. We suggest, in fact, that phosphorus normally has an atmospheric component, in analogy with other elemental biogeochemical cycles, but that the present time is not normal, at least as far as phosphorus is concerned. We suggest that the atmospheric component of the environmental phosphorus cycle is the fundamental thing which the Flood broke. And we suggest that it was this breakage which resulted in the loss of human longevity following the Flood—that loss of the atmospheric component of the phosphorus cycle (i.e., loss of this phosphorus trace gas) meant loss of the vitamin X precursor, resulting in loss of vitamin X itself. The fact that the environmental phosphorus cycle is not represented by any atmospheric component today does not mean that this was the case before the Flood. Phosphorus gases do exist and are well known in the chemistry laboratory and in industry.
Phosphine (PH3) is the simplest gas containing phosphorus. It is a possible pre-Flood phosphorus trace gas candidate, but it is not a possible vitamin X precursor candidate. Vitamins are organic compounds (i.e., they contain one or more carbon atoms). Phosphine is not organic. Atmospheric chemistry can add oxygen atoms to a precursor gas; it cannot add carbon atoms. Thus, the precursor gas must start out as an organic compound.
Simple organic trace gases of the elements often result from addition of one or more methyl groups (CH3) to the element. An example is methyl iodide (MeI), already mentioned. MeI is sourced to the atmosphere from the oceans. The oceans also source methyl chloride (MeCl) and methyl bromide (MeBr) to the atmosphere. Dimethyl sulfide (DMS or Me-S-Me) is an example of a trace gas containing two methyl groups. It, too, is sourced to the atmosphere from the oceans.
We are looking for a simple organophosphorus trace gas sourced to the atmosphere from the oceans. Methylated phosphorus gases seem to be the obvious choice.
The biological pathway by which methylated phosphorus gases might arise in anaerobic sedimentary environments has yet to be demonstrated by science. It seems, however, that discovery of such a pathway is imminent.
There are strong indications that phosphine and other reduced phosphorus compounds can be produced biogenically.[28]
Two different modes of production of methylated phosphorus gases can be imagined: 1. biomethylation of phosphorus, and 2. catabolism of phosphonates.
Biomethylation in general is well studied and reasonably well understood.
Biomethylation is the process whereby living organisms produce a direct linkage of a methyl group to a metal or a metalloid, thus forming metal-carbon bonds. Methylation has been extensively studied and biomethylation activity has been found in the soil, but mainly occurs in sediments in e.g. estuaries, harbors, rivers, lakes and oceans. … Anaerobic bacteria are believed to be the main agents of biomethylation in sediments and other anoxic environments.[29]
From a purely utilitarian perspective, however, the second possibility, catabolism of phosphonates, seems to us to be somewhat more attractive than biomethylation.
Phosphonates are chemical compounds having a carbon atom (C) bonded to a phosphorus atom (P). Catabolism is the process by which living organisms break down complex molecules into simpler molecules during metabolism.
In the pre-Flood anaerobic, organic-rich, phosphate-rich marine sedimentary environment surrounding Antarctica, microbes might naturally be expected to have been present which possessed an ability to "mine" phosphonates for their oxygen atoms while discarding the less readily catabolized C-P moieties as methylphosphine (MeP) "tailings," since both carbon and phosphorus would be abundantly available in more easily catabolized forms. The great versatility of microbial life makes it seem likely that some anaerobic microbe having ability to exploit this environment in this way should exist.
From chemistry, it is known that phosphorus can add one, two, or three methyl groups, giving methylphosphine (MeP), dimethylphosphine (DMeP), and trimethylphosphine (TMeP), respectively. Of these three, TMeP is the least interesting for the present purpose. It has a boiling point of 38–40°C, which means that, in the laboratory, it is a liquid rather than a gas. TMeP vapor oxidizes in the atmosphere to trimethylphosphine oxide (TMePO). TMePO is not easily oxidized a second time, and it is a severely hygroscopic solid, which means that it will be rapidly washed out of the atmosphere. If vitamin X is a small acid, as expected, then TMePO, which is not an acid, is not vitamin X.
Both DMeP and MeP are true laboratory gases. Both yield small acids upon oxidation in the atmosphere.
DMeP oxidizes first to dimethylphosphine oxide (DMePO). This is a liquid with a boiling point of 220°C. Like TMePO, this is likely to wash out of the atmosphere. However, it can oxidize further to dimethylphosphinic acid (DMePiA).
The final methylated phosphorus compound and potential vitamin X precursor, the gas MeP, oxidizes first to methylphosphine oxide (MePO). MePO is not very stable. It easily oxidizes a second time to methylphosphinic acid (MePiA). This small acid is hygroscopic; it will wash out of the atmosphere. However, it is readily susceptible to further oxidation, either in the gas phase or in aqueous solution, yielding methylphosphonic acid (MePA) as the stable final product.
Thus, two precursor gases (DMeP and MeP) and three vitamin X candidates (DMePiA, MePiA, and MePA) result from methylated phosphorus.
DMePiA is the least interesting of these three vitamin X candidates. It is a phosphinate. Phosphinates have either one carbon atom and one hydrogen atom or two carbon atoms bonded to the phosphorus atom. Phosphinates appear to possess severely limited natural biological utility.
Phosphinothricin (PT), a non-proteinogenic amino acid found in a number of peptide antibiotics, is the only known phosphinic acid natural product.[30]
In sharp contrast, phosphonates (which have just one carbon atom and no hydrogen atom bonded to the phosphorus atom, as with MePA) appear to have ubiquitous biological utility. Phosphonates have an especially large abundance in the marine environment[31] and in some marine organisms.
… eggs of the freshwater snail Helisoma contain 95% of their phosphorus in the form of 2-aminoethylphosphonate-modified phosphonoglycans, whereas the sea amenaea Tealia possesses up to 50% of its phosphorus in a variety of phosphonolipids, phosphonoglycans and phosphonoglycoproteins. Other organisms, such as the protist Tetrahymena, have lower overall levels of phosphonate, but still synthesize as much as 30% of their membrane lipids in the form of phosphonolipids. The prevalence of C-P compounds in nature is perhaps best exemplified by the recent discovery that as much as 20-30% of the available phosphorus in the world's oceans is comprised of phosphonic acids.[32]
The ubiquitous biological utility of the phosphonates within the marine environment implies that a catabolic route to vitamin X precursor formation exists for MeP alone. MeP has only one carbon atom bonded to the phosphorous atom, while DMeP has two carbon atoms bonded to the phosphorous atom. Thus, formation of MeP may reasonably be expected as a byproduct of catabolism of ubiquitous phosphonate substrate in an anaerobic marine environment, but the catabolic production of DMeP would require abundant phosphinate substrate, which, as we have just seen, does not appear to exist in nature.
Thus, the lack of a catabolic route to formation of DMeP as vitamin X precursor renders DMePiA of low interest.
This leaves MeP as the sole interesting vitamin X precursor gas, with both MePiA and MePA as interesting vitamin X candidates arising from methylated phosphorus (Figure 9.3).
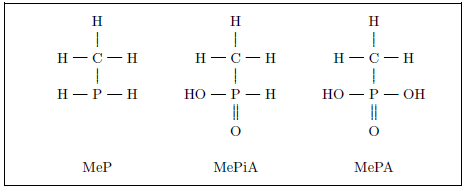 |
MePA is of great interest as a potential vitamin X candidate. It is the simplest member of the phosphonate class of compounds. Phosphonates as a class are biologically versatile compounds with "potent bioactive properties."[33] MePA thus appears appropriately positioned to fill the role of a vitamin having anti-aging potency.
But, in a curious and unexpected fashion, interest in the MePiA candidacy surpasses even the great interest warranted by the MePA candidacy.
It might be thought that the evident lack of biological utility of the phosphinates would play against the MePiA vitamin X candidacy since methylphosphinic acid is a phosphinate. How, for example, could MePiA participate in the sort of biomolecule building activity exemplified by some of the traditional vitamins (e.g., in their use as organic cofactors and prosthetic groups of larger enzymes) given the near-zero biological utility of the phosphinates? The ubiquitous biological utility of the phosphonates opens the door wide for the MePA vitamin X candidacy. Conversely, the near-zero biological utility of the phosphinates seems to all but close the door on the possibility that MePiA might be a vitamin.
However, curiously, the door does not close on the MePiA candidacy if the biological utility of MePiA is merely for it to act as an antioxidant (i.e., a substance which "soaks up" reactive oxygen species [ROS]). In the process of soaking up ROS, MePiA is converted to MePA. Thus, while MePiA, as a phosphinate, might not be expected to participate in the sort of biomolecule building which is characteristic of some of the traditional vitamins, it might still function in a vitamin-like antioxidant capacity, and, in the process, yield a phosphonate product, MePA, which might then very well be expected to participate in biomolecule building and enzymatic processes similar to other vitamins.
Once this much has been understood, a complete theory of vitamin X suddenly emerges. Indeed, MePiA suddenly appears as a vital missing piece of a long-standing theory of aging.
The long-standing theory referred to is Denham Harman's free radical theory of aging in its final, mitochondrial form.[34] Harman postulated that aging is caused by accumulation of biomolecular damage due to free radicals (often called ROS in biological contexts) generated during normal cell metabolism. A strategy to limit damage due to ROS is to make antioxidants (i.e., easily oxidized compounds) available to the cells. The antioxidants will then be on hand to soak up the ROS and thereby keep them from reacting destructively with cellular biomolecules.
Harman found that the average life span of mice could be increased by large daily intakes of select antioxidants. This corroborated the basic idea of free-radical damage as a cause of aging. However, somewhat surprisingly, Harman found that maximum life spans of mice could not be increased in this way.
This led Harman to postulate that the mitochondria (the locus of highest ROS concentration) were the main site of life-limiting free-radical damage and that possibly the mitochondria, which have a double membrane prohibiting entry of most compounds, were inaccessible to his antioxidants.
Harman's theory is all about aging. Vitamin X is all about aging. Harman's theory invokes the use of antioxidants as a cure for aging. Several of the vitamins are antioxidants. More to the point, vitamin-X-candidate MePiA is an antioxidant. The postulate presents itself that MePiA is the missing antioxidant needed to complete Harman's theory. We will call the theory which results from this postulate the theory of vitamin X.
Theory of Vitamin X: MePiA is vitamin X, an antioxidant (produced naturally prior to Noah's Flood) needed to complete Harman's free-radical theory of aging. MePiA has special access to the mitochondria. Dietary intake of MePiA regulates the rate of ROS damage to mitochondria, which regulates the rate of human aging.
This theory of vitamin X is fortified by its immediate ability to explain the previously discussed observation, from the biblical life expectancy data, that vitamin X must have an unusually long biological half-life—on the order of a century. This observation is otherwise difficult to explain. No traditional vitamin has such a long half-life in the body. The biological half-lives of the water-soluble vitamins are notoriously short. Vitamin C has a biological half-life of 10 to 20 days, for example. And even the longer-retained fat-soluble vitamins still tend to have biological half-lives measured in just days or weeks. Vitamin E appears to be exceptional, with a biological half-life of a few years, but no traditional vitamin comes close to a biological half-life of a century.
The idea that MePiA has special access to mitochondria implies that specialized transport proteins exist to take MePiA molecules through the double membrane into the mitochondria, concentrating and keeping MePiA there. This implies genetic support of the protective role played by MePiA in the mitochondria. And this invokes the possibility of genetically programmed enzymes to recycle/reactivate MePiA molecules following their reaction with free radicals in the mitochondria. Genetically programmed sequestering and/or recycling of MePiA in the mitochondria is perhaps the only biological conservation mechanism capable of explaining the extremely long biological half-life of vitamin X.
We propose that MeP is the vitamin X precursor gas and that MePiA is vitamin X.

Once the theory of vitamin X developed in the previous chapter had clarified, a longevity experiment with laboratory mice was launched to test the theory.
MePiA, according to the theory of vitamin X, acts as an antioxidant in the body to mitigate damage due to free radicals, in accordance with Harman's mitochondrial free-radical theory of aging.[35] MePiA is oxidized to MePA by free radicals of the reactive oxygen species (ROS) group. MePA may then operate in a typical vitamin capacity in a variety of biochemical reactions. Ultimately, it is either enzymatically reduced back to MePiA or filtered by the kidneys and excreted from the body. This theory makes MePiA to be essential for life lengthening, and the expectation was that mice receiving MePiA would show life lengthening.
The working hypothesis at the launching of the MePA-vs-MePiA mouse experiment was that MePA and MePiA acted as a pair of anti-aging vitamins in the body. Earlier experiments on mice treated with MePA alone had shown no life lengthening. This implied that mice lacked any enzyme capable of converting dietary MePA to MePiA so that direct administration of MePiA would be necessary to observe life lengthening.
Life span experiments with mice take several years to run to completion. It has previously been described how early results from this experiment seemed contrary to the theory of vitamin X, leaving MePA as the sole anti-aging vitamin, and how it took two years for this mistaken impression to be corrected.[36] The present chapter reports on the final results of this experiment. It shows that the mice receiving MePiA did experience life lengthening, corroborating the theory of vitamin X beyond reasonable doubt.
Figure 10.1 shows the final graph of the MePA-vs-MePiA dataset. Each treatment group began with 4 cages containing 9 mice each, all of the same age. One cage was removed from the MePiA dataset due to a treatment-unrelated, juvenile die-off event.[37] The graph shows that the percentage of survivors in the group of MePiA-treated mice surpassed the percentage of survivors in the group of MePA-treated mice from about 110 weeks of age onward. More importantly from the perspective of aging research,[38] the maximum life span for the MePiA mice was significantly greater (P = 0.02 via a two-sided T-test) than the maximum life span for the MePA mice. Taken at face value, this graph shows life lengthening of the MePiA mice relative to the MePA mice.
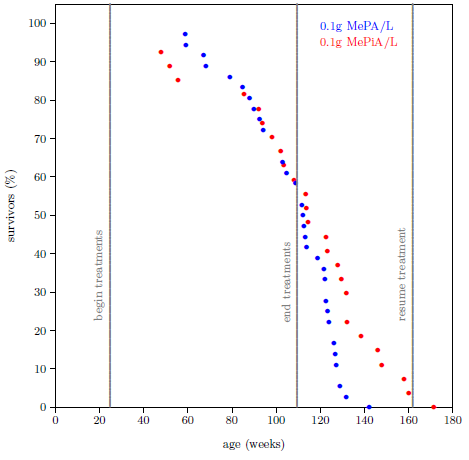 |
Extension of maximum life span is extremely difficult to achieve. On the global research scene, only a very few interventions, such as calorie restricted diets, have succeeded in demonstrating an extension of maximum life span in mice. Though our research with various vitamin X candidate substances has attempted to demonstrate life lengthening for multiple decades with many hundreds of experimental animals—gerbils, fruit flies, vinegar worms, mice, and rats—this is the first time any evidence of increased maximum life span has been seen in our work.
Is this observed life lengthening real, or is it just a statistical fluctuation? To answer this question, we added to the graph all of the data our lab had ever produced using batches of same-age, weanling, female, ICR mice. There had been three additional batches in this category, spanning nearly two decades. These batches had been subjected to a variety of treatments back in their day, intended to induce life lengthening—all of which had uniformly failed in this purpose. Figure 10.2 shows that, for roughly 50% or fewer survivors, the survival curve for the MePA mice of batch 4 (dark blue dots) is similar to the older three batches of ICR mice, while the survival curve of the MePiA mice (red dots) clearly differs from the older three batches. MePiA treatment does indeed appear to have extended the maximum life span for these mice, confirming that MePiA is vitamin X.
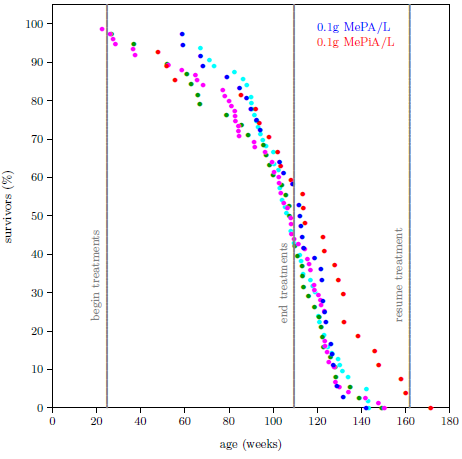 |
The theory of vitamin X provides the following explanation of Figure 10.2.
For the first six months of their lives, before treatment with MePA or MePiA began, all mice aged at a normal rate due to mitochondrial degradation occasioned by free radicals generated within their mitochondria. Following initiation of treatments, MePA mice continued to age at a normal rate, MePA being resistant to oxidation and hence offering no protection against free radicals. Meanwhile, aging in MePiA mice was slowed due to the presence in their mitochondria of easily oxidized MePiA.
When treatment ceased, a normal rate of aging resumed in MePiA mice, but their mitochondria had suffered less damage than the mitochondria of MePA mice throughout the treatment phase. As a result, the MePiA-treated mitochondria were able to sustain post-treatment damage for a longer time than the MePA-treated mitochondria before critical damage levels, resulting in death, were reached. This evidenced as lengthened life spans of the MePiA mice relative to the MePA mice.
Before closing this chapter, one further point requires explanation. Why did the mice yield so little gain from so much treatment? The concentration of MePiA used in the mice experiment of Figure 10.2—100 mg/L—is expected to be on the order of 10,000 times larger than the concentration of MePiA naturally available in rainwater thousands of years ago. Meanwhile, the life spans of the treated mice were increased only about 20% at best, while the life spans of pre-Flood humans exceeded modern human life spans by at least 1,000%.
There are several possibilities. For example, it is possible that, unlike humans, mice do not possess the genetics necessary for active support of the natural antioxidant role played by MePiA in the mitochondria. Possibly, for mice, MePiA is present in mitochondria only because of passive diffusion, not because of active transport, and each MePiA molecule gets to be used only once rather than being recycled and reused repeatedly, resulting in much less efficient protection by MePiA of mouse mitochondria than of human mitochondria. Another possibility is that these mice may have been facing a much higher risk of death from disease than was faced by biblical adult humans. Mice are susceptible to a large number of infectious diseases, some of which can manifest as wasting diseases that look very similar to the physical wasting characteristic of "old age" in mice. Thus, death from diseases shared within the colony of mice could have truncated the life span extension of the MePiA-treated group.
Additional research is being conducted to investigate these and other potential explanations.
A life span experiment with MePiA-treated mice has confirmed that MePiA is the long-sought vitamin X. The life spans of nearly 1,000 years enjoyed by humans before Noah's Flood may be explained by the presence of MePiA in drinking water back at that time. Loss of MePiA from the environment, occasioned by Noah's Flood, explains why modern humans die generally before they have reached even 100 years.

Having developed a qualitative explanation (a theory) of why human life spans plummeted following the Flood, the next task was to apply this theory quantitatively to the biblical life expectancy data. This was done by first putting the theory into mathematical form. To solve the resulting equations required use of numerical methods on a computer. The resulting mathematical computer model (detailed in the appendix) was then applied to the biblical life expectancy data.
It was necessary to model the data quantitatively for two reasons. The first reason was to test the theory against the life expectancy data. It is one thing to draft a qualitative theory of a complex process such as changing life spans. It is quite another thing to get the qualitative theory to agree quantitatively with real-life data. This will not happen by chance. Would the theory succeed with the biblical life expectancy data or be falsified by it?
The second reason, assuming success, was to be able to learn as much as possible about the nature of vitamin MePiA from these ancient data. For example, it was clear, as previously discussed, that MePiA must have a long biological half-life. The need was to move beyond this qualitative observation to an objective quantitative appraisal of the biological half-life—to get the best objective, quantitative estimate possible of this and other parameters from the available data.
A second component of this second reason was the desire to be able to follow some processes in greater detail. An example in this category is the "aging" process experienced by Noah. Noah lived 600 years in the Pre-Flood, then he lived through the Spike, and then he lived several centuries in the Decline. His natural daily intake of MePiA went from high, to very high, to low. How did all of this affect his "agedness" along the way?
The biblical life expectancy data are the only data that presently exist on the effects of MePiA on human longevity. To exploit these data fully to the benefit of the planet's modern human population, a rigorous mathematical model was mandatory.
The mathematical computer model was constructed using Fortran. The source listing can be found in the appendix. The result of the model is shown in Figure 11.1. The model clearly succeeds in explaining the data. This demonstrates both that the theory is sound and that the biblical life span data are valid historical observations.
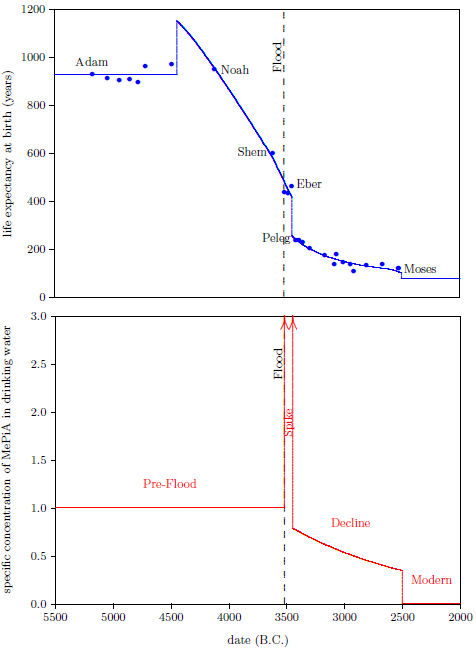 |
The sudden jump upwards in life expectancies between Adam and Noah may look like a mathematical glitch, but it is a real jump in life expectancies. It results from the Spike. People born immediately to the left of the jump have a normal pre-Flood life expectancy of 926 years. These people die of "aging" just as the Spike is beginning. As a result, they just miss its beneficial effects. People born immediately to the right of the jump live long enough to make it into the Spike and benefit from it. In the Spike, their exposure to very high levels of MePiA extends their life expectancies by several centuries.
The Eber–Peleg Drop is at the opposite end of this benefit to life expectancies caused by the Spike, as previously discussed.
The model involves two components: environmental and physiological. The environmental component yields the concentration of MePiA in drinking water. This is shown by the red line in the bottom graph in Figure 11.1. This is the driving function for the physiological component, which calculates the theoretical life expectancies shown as the solid blue line in the top graph.
For the sake of the model, the biblical longevity data were treated as point estimates of mean life expectancy at birth, as previously discussed.
The first seven data points were simply averaged, resulting in the height of the initial horizontal blue line. These seven points were otherwise excluded from the model. The line through the remaining data points results from a least squares fit of four free (i.e., adjustable) parameters. Two free parameters were needed to describe the changing environmental abundance of MePiA, and another two were needed to describe the physiological response to varying intakes of MePiA.
The environmental component of the model was divided into four distinct time periods, as previously discussed. The flux of MeP from the oceans to the atmosphere was assumed to be constant pre-Flood, resulting in a constant supply of MePiA in drinking water. All drinking water concentrations of MePiA in all time periods were scaled with respect to this (unknown) pre-Flood concentration, yielding the "specific concentration of MePiA in drinking water" plotted in the bottom graph of Figure 11.1 shown previously.
The atmospheric concentration of MeP was very large during the Spike for reasons discussed previously.Just how large is unknown, but we can get a rough idea as follows. Prior to the Flood, assuming steady state conditions, the rain of phosphorus-rich detritus onto the sea floor surrounding Antarctica supplied a flux of MeP to the atmosphere, resulting in unit specific concentration of MePiA in drinking water. This MeP flux was halted as nearly the entire sedimentary supply of MeP was vented to the atmosphere by the Flood. The rain of phosphorus-rich detritus onto the ocean floor around Antarctica resumed after the Flood. At present, five and a half thousand years after the Flood, human longevity indicates that the specific concentration of MePiA is still effectively zero. Subtracting 1,000 years to allow for transport of MeP from the ocean floor to the atmosphere at present yields a minimum of 4,500 years' worth of pre-Flood MeP vented to the atmosphere during the Flood. Thus, the flux of MeP to the atmosphere during the year of the Flood increased by at least a factor of 4,500.
For such a large Spike, the actual amplitude turns out to be irrelevant. The physiological part of the model sets a fairly low cap on the body's reserve capacity of MePiA, and this cap would have been met throughout this interval.
The end of the Spike interval coincides with the Eber–Peleg drop in life expectancies. This drop may have happened anywhere between the third (Eber) and fourth (Peleg) data points following the Flood. The model gives a best fit when the drop occurs close to the third data point, with a total duration of 69 years for this interval.
The atmosphere was being provided with MeP from the oceanic reservoir during the Decline interval. The Decline interval ends with the Moses Drop in 2504 B.C., as previously calculated. This drop signals the exhaustion of the oceanic MeP reservoir.
The existence of the Moses Drop means that the oceanic supply of MeP came to an end suddenly rather than gradually. The whole ocean slowly circulates vertically, dense (cold, saline) water formed in the north during winter sinking rapidly each year to the bottom, pushing the overlying water of the entire ocean slowly upward. The Moses Drop implies that, in the (3520 - 2504 =) 1,016 years it took Flood ocean bottom water to rise to the surface, this Flood ocean bottom water did not mix significantly with the underlying post-Flood ocean water. This, in turn, implies strong stratification between these two bodies of water, which implies that they had significantly different densities. Thus, the existence of the Moses Drop informs us that the density of Flood ocean bottom water at the end of the Flood was significantly less than the density of newly formed post-Flood ocean bottom water.
This is expected. It seems inevitable that the Flood would have effectively homogenized the pre-Flood oceanic water mass. Normally, bottom water is more dense than any other ocean water because it is colder and more saline than any other ocean water. Surface waters are least dense, being warmed by sunlight. Homogenization of the pre-Flood ocean by the Flood would have produced a body of Flood ocean water having a temperature and a density intermediate between normal bottom waters and normal surface waters. Thus the density of this well-mixed body of waters from before the Flood would have differed significantly from the density of newly formed, post-Flood bottom water.
The idea that the Flood homogenized the oceans is supported by simple geometrical considerations. The average depth of the oceans is 3.8 kilometers. The average travel distance for ocean water during the Flood was roughly one quarter of earth's circumference during the waxing of the Flood, then another one quarter of a circumference back again at the waning of the Flood. This is a total distance of roughly 20,000 km. Thus the ratio of the depth to the distance traveled is less than one five-thousandth. It seems nearly impossible to maintain density stratification in such a geometry, and it seems especially so when one considers that the flow was over uneven ocean floor and continental terrain.
The two environmental free parameters mentioned above were needed to describe the specific concentration of MePiA in drinking water during the Decline, which is modeled as a decaying exponential. Decay is due to internal oceanic chemical and/or biological processes consuming MeP precursor. The first free parameter sets the amplitude of the exponential, and the second free parameter sets its rate of decay.
In the Modern time period, the flux of MeP to the atmosphere is taken to be zero by the model. What we know about the nature of the Flood implies that MeP must have been nearly emptied from its phosphorus-rich, anaerobic sedimentary source beds surrounding Antarctica. In addition, the global constancy of life expectancies due to "aging" since the Moses Drop strongly argues for this choice. For example, well water might be expected to be depleted of MePiA due to such processes as sorption of small acids by soils or its utilization by microbes as rainwater moved from the surface through the soil into the well, yet modern populations drinking water from rivers or lakes show no difference in life spans compared with populations drinking water from wells.
The physiological component of the model is most easily constructed by analogy with a small gasoline engine. Wear of engine components results from inadequate lubrication. Wear corresponds to "aging" and the lubricant corresponds to MePiA in this analogy. Lubricant is slowly lost, by being vaporized from the cylinder wall with each ignition cycle, for example. Lubricant is added into the machine's lubricant reservoir at varying rates from an external source not subject to control by the machine. A low rate of supply of lubricant results in a deficient amount of lubricant in the machine's lubricant reservoir. A deficient amount of lubricant results in inferior lubrication and an increased rate of wear of parts. A high rate of supply of lubricant may fill the machine's lubricant reservoir. Once the reservoir is full, the machine can receive no more lubricant regardless of potential supply rate.
The lubricant reservoir corresponds to the body's reservoir for storage of MePiA in this analogy. The size of this reservoir is the first free parameter of the physiological part of the computer model. The second free parameter is the lifetime of MePiA in the body (corresponding to the lifetime of lubricant in the machine). Here again, exponential decay was assumed.
A new machine has no wear (i.e., agedness = 0). The machine runs (lives) until it reaches a critical level of wear (i.e., until agedness = 1), at which point it is worn out and no longer works (i.e., it dies).
The biblical longevity data provide two steady state regimes, allowing a linear relationship between rate of wear and lubricant level to be derived for use in the model. Letting SC be the specific concentration of MePiA in drinking water, the first is the Pre-Flood period in which SC = 1 and the average life span for males is 926 years, and the second is the Modern period in which SC = 0 and the average life span for males is 76.8 years.[39]
This simple mechanical analogy to biological "aging" works well, but it is deficient in one respect. While self-repair (i.e., healing) is not possible mechanically, it is possible biologically. The model allows for this possibility by allowing the rate of wear to become negative for large enough lubricant levels (Figure 11.2).
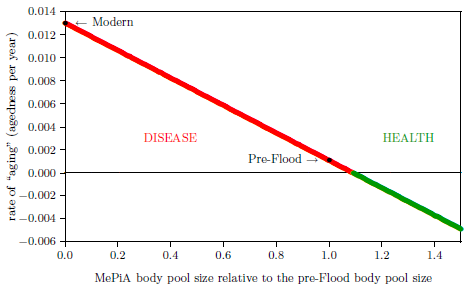 |
The duration of the Spike was not a free parameter of the mathematical optimization of the goodness-of-fit parameter, Χν2, because it is a discrete, integer variable. Nonetheless, it could be optimized by giving it different values in separate runs of the computer model.When this was done, 69 years was found to be the duration yielding the best goodness of fit, as mentioned above. This implies that the large spike of MeP which the Flood released from the sediments surrounding Antarctica took 69 years to clear from the atmosphere.
Normally, MeP, which is easily oxidized, has a lifetime of only a few days at best in the atmosphere. The implication seems to be that the atmospheric chemical reactions which normally keep the atmosphere free of unwanted gases were significantly overloaded during the Spike interval. This overloading would have been contributed to by other anaerobic sea floor gases, especially methane.[40] Thus, it appears that earth's atmosphere may have been significantly polluted globally during much of the Spike.
The amplitude of the oceanic reservoir free parameter is the contribution of the oceanic reservoir to SC (the specific concentration of MePiA in drinking water) in 3519 B.C., immediately following the year of the Flood. The model yields a value of 0.838±0.006 for this parameter. (All error estimates from the model are ±1σ.)
If there had been no Flood, then the value of this parameter would have been 1.0, as it had been throughout the pre-Flood interval. The fact that it is less than 1.0 implies that the oceanic flux of MeP to the atmosphere was reduced immediately following the Flood, compared to what it had been before the Flood.
This parameter specifies the rate at which MeP was lost from the oceanic reservoir, not into the atmosphere, but internally due to chemical and biological degradation. The model yields a value of (8.63±0.07)×10-4 per year for this parameter. This means that it would take these internal processes about 900 years to deplete the oceanic MeP reservoir by a factor of two. Biological processes may have been dominant as "C-P lyase genes are abundant in marine microbes."[41]
The model finds a reservoir size for MePiA of 1.18±0.02 times the pre-Flood body content of MePiA (Figure 11.3). This implies that the body reservoir for MePiA was ([1 - 1/1.18]×100=) 15% underfull for pre-Flood humans. To top up the body reservoir for MePiA requires the intake of MePiA to be 18% greater than the pre-Flood intake, according to the model. It appears that this condition would have been easily met throughout the Spike interval, as discussed above.
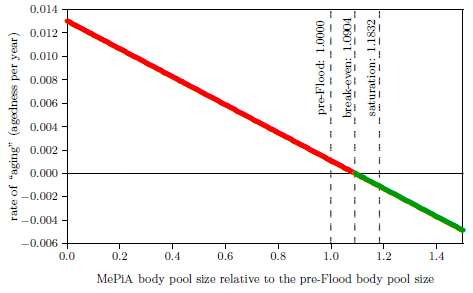 |
The model finds a lifetime for MePiA in the body of 194±4 years. The corresponding biological half-life is 135±3 years. This is a remarkably long biological half-life, as previously discussed, implying specialized mechanisms for conservation of MePiA.
It is clear, by visual inspection of Figure 11.4, that the fit of the model to the data is quite good. The model has succeeded in capturing the panoramic essence of the biblical life expectancy data and in quantifying its free parameters.
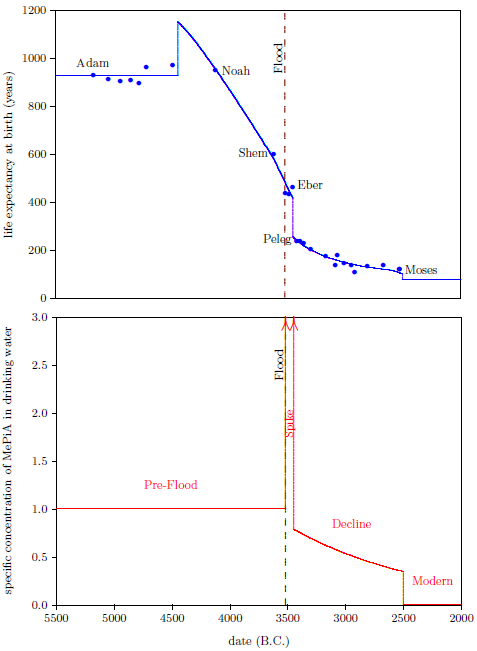 |
The model's greatest deficiency appears to be that it finds a life expectancy which seems certainly too low just prior to the Moses Drop.[42] The blue dot labeled "Moses" is actually two data points close together, one for Moses and the other for Aaron. While the actual life spans in these two instances were 120 and 123 years respectively, the model predicts a life expectancy of 107 and 108 years for these two individuals. This results, not from statistical scatter, but from over-simplification of this part of the model. Specifically, in real life there would have been an "echo" of the Spike during the lifetimes of Moses and Aaron which is not included in the model. This "echo" results from the fact that during the Spike newly formed ocean bottom water would be enriched in dissolved MeP precursor gas relative to prior and subsequent years. Recall that ocean bottom water is formed from dense (cold, saline) northern surface water sinking each winter. This water would be in contact with the atmosphere prior to sinking. Since the atmosphere contained an unusually high concentration of MeP during the Spike, bottom water originating during the Spike would also contain an unusually high concentration of MeP. This is not included in the model. The model treats the Decline phase as a simple exponential decay. To be more realistic, the Decline would need to be modeled as an exponential decay plus a "hill" (probably a Gaussian, resulting from diffusion of the elevated MeP during the roughly one thousand years needed for the Spike-formed bottom water to rise to the surface) at the end of the Decline. Such fine detail has been neglected in the current model to keep things as simple as possible at this stage.
The success of the model drives home the conclusion that the biblical life expectancy data cannot be mythological or otherwise fabricated. What did the ancient author of Genesis understand about methylphosphine (MeP), about its reactions with oxidants in the atmosphere yielding methylphosphinic acid (MePiA), about Antarctica, about upwelling phosphate and nitrate in the southern oceans, about vitamins and vitamin deficiency diseases, about anaerobic digestion within sea floor sediments, about the baring of southern ocean floors during the Flood? Yet, all of these things and more must be properly understood to explain the pattern of these biblical life expectancy data points. The only reasonable explanation of the amenability of these ancient biblical life expectancy data to quantitative, scientifically rigorous analysis is that they are accurately recorded, real-life observations.
Conversely, the goodness of the fit to these data validates the model and its theoretical underpinnings. The central hypothesis is strongly corroborated: human "aging" is a vitamin deficiency disease resulting from dietary insufficiency of a previously unknown vitamin—now understood to be MePiA—which was made globally deficient by the impact of Noah's Flood on earth's environment.
We call vitamin MePiA deficiency disease "Aging 1" for short.

Aging 1 is vitamin MePiA deficiency disease. To cure a vitamin deficiency disease requires only adequate intake of the deficient vitamin.
Normally, adequate intake of a vitamin is obtained by eating foods containing that vitamin in adequate amounts. This normal strategy will not work with vitamin MePiA because there is no natural food or drink which contains this vitamin today.
In the ancient past, this vitamin was naturally available in rainwater. As a result, it was present in the water ancient individuals drank from rainwater-fed sources such as ponds and rivers. But Noah's Flood broke the natural production of vitamin MePiA so that it is not available in rainwater-fed sources of drinking water any longer.
To obtain an adequate daily intake of MePiA today, it is necessary to supplement one's diet with this vitamin artificially. The chemical compound corresponding to vitamin MePiA is methylphosphinic acid. This is a known compound which can be chemically synthesized relatively easily. As a result, supplementation of our diets with vitamin MePiA may be easily accomplished today.
RDI (Recommended Daily Intake or Reference Daily Intake) is the normal metric used by nutritionists seeking to establish an appropriate consumption level of a given vitamin or other nutrient. In the case of MePiA, one may wonder whether daily intake is really necessary given the very long biological half-life of this vitamin. The short answer is that while daily intake seems unlikely to be necessary, it is nonetheless expedient. Since the biological half-life of MePiA appears to be over a century, it is theoretically possible that weekly, monthly, and even yearly supplementation with this vitamin, at increasingly large intakes as the interval is lengthened, might suffice to meet physiological needs. The main theoretical argument against such a practice is that the natural frequency of intake prior to the Flood was daily—indeed, every time one took a drink of water. The incredibly complex human body is likely to be best served by emulating this natural frequency. In addition, there is a practical argument, it being most convenient to take this vitamin daily, as will become clear in subsequent chapters.
Determination of the optimal daily intake of any vitamin is not a trivial exercise. For example, although the chemical compound corresponding to vitamin C, ascorbic acid, was discovered in the 1930's, the optimal daily intake is still an ongoing debate today.
The appropriate daily intake is still vigorously disputed by scientists, and recommended allowances not only vary from one country to another; they also change from time to time within the same country. Although everyone agrees that the minimal daily requirement for vitamin C is 10 mg or slightly less, there is little agreement regarding recommended intakes.[43]
Determination of the optimal intake of MePiA
seems likely to be an area of active research for years as initial, crude estimates are gradually refined.
The biblical life expectancy data are, once again, the best place to begin. But the modeling work discussed in earlier chapters of this book was necessarily carried out using "specific concentrations." These are concentrations which are relative to the (presently unknown) pre-Flood concentration of MePiA in drinking water.
What is now needed is some means of converting these relative concentrations to absolute concentrations. We would like to know how many micrograms of MePiA a person should take per day.
The most direct route to converting the model's relative concentrations to absolute concentrations appears to be via the measurement of MePiA concentration in pre-Flood glacier ice. This has complications, such as the susceptibility of MePiA to oxidation over the thousands of years which have transpired since its deposition in snow on the glacier. But natural oxidation of MePiA will yield MePA, which is stable, so that, even in the case of complete oxidation of MePiA in glacier ice, measurement of MePA can be used to obtain an upper limit for pre-Flood MePiA concentration. This is not the only potential complication. Once again, determination of the optimal intake of MePiA is not expected to be an easy task. It presently appears that multiple experimental routes will need to be explored, and the final determination seems likely to be a composite of multiple estimates from multiple routes. The measurement of MePiA and MePA in pre-Flood glacier ice seems attractive because it appears to hold potential to tell us most directly the intake rate prior to the Flood, which would allow replacement and saturation intakes to be calculated by the model.
A second route to absolute intake rates is via the measurement of MePiA in the urine and/or blood serum of treated human subjects.This method should allow the size of the body pool (or reservoir) of MePiA to be measured, from which the necessary daily replacement intake can be calculated using the biological half-life provided by the model.
A third route to estimate the daily needed intake of MePiA is to measure MePiA in human milk. Since MePiA is water soluble, it is likely present in the milk of a mother consuming MePiA. Milk is nature's perfect food, designed to meet the needs of the growing baby for most, if not all, nutrients.[44] Therefore, measurement of human milk MePiA concentrations would allow calculation of total MePiA intake for the baby. Based on all the other essential nutrients, this total MePiA intake can be assumed to be optimal for the baby. This estimate would provide a starting place from which to predict the MePiA requirements of humans at other ages and stages of life.
Work on these needed measurements has been given highest research priority at ARP for several years. Because of the very low concentrations of MePiA and MePA involved, plus the relatively high concentrations of potentially interfering substances in the samples being measured, the measurements are difficult to make. Nonetheless, we anticipate obtaining our first meaningful results probably within a year.
Fortunately, the model provides a method of estimating the maximum possible pre-Flood intake in absolute terms, and from this the maximum saturation intake can be calculated. This sets a useful upper limit for the recommended daily intake.
Recall that SC is the specific concentration of MePiA in drinking water. The model gave, as the amplitude of the oceanic reservoir free parameter contribution to SC, 0.838±0.006. This value for SC corresponds to the post-Flood oceans immediately following the Flood. These oceans had been homogenized by the Flood, as previously discussed. As a result, they could not have contained any more than a surface saturation concentration of MeP. Any amount of MeP above this would have bubbled out of solution during homogenization.
The behavior of a gas in a liquid is governed by Henry's Law. This scientific law says that the concentration of the gas in the liquid will be proportional to the applied pressure of that gas.The proportionality constant for MeP does not seem to be readily available. A value of 7.8×10-4 moles of MeP gas/(atm • cubic meter of water) was found when this constant was measured in our laboratory. This means that methylphosphine gas has a relatively low water solubility, similar to that of hydrogen gas.
Once the Henry's Law constant is known, the concentration of MeP in saturated surface water can be calculated. The result is 7.8×10-4 moles of MeP gas/cubic meter of water.
The average depth of the oceans is 3,800 meters. The turnover time of the oceans after the Flood was the time from the Flood to the Moses Drop. This is (3520 - 2504 =) 1,016 years. So the rate of turnover was 3.74 meters/year.
The surface area of the oceans is 3.61×1014 square meters. So MeP was being added to the atmosphere from the oceans at a rate of 1.0×1012 moles of MeP/year for SC = 0.838.
For the pre-Flood, SC = 1. So the maximum pre-Flood flux of MeP to the atmosphere was 1.3×1012 moles of MeP/year.
Now we need the atmospheric conversion efficiency for MeP to MePiA. This is difficult to measure. A 23% conversion efficiency for reaction with ozone was measured in our laboratory. Much smaller conversion efficiencies resulted for reaction with both hydroxyl and nitrate radicals. However, these measurements were for the gas phase only, while some of the chemistry which converts MeP into MePiA in the atmosphere probably takes place in aqueous phase on aerosols. This would likely boost the contribution of MePiA from both hydroxyl and nitrate radical reactions. Since we are presently trying to obtain a maximum possible pre-Flood intake, it seems best to adopt a value of 50% as the probable maximum conversion efficiency. This yields a maximum of 6.3×1011 moles of MePiA being produced in the atmosphere each year.
The total annual rainfall globally is 5.4×1017 liters/year. So the maximum pre-Flood MePiA concentration in rainwater was 1.2×10-6 mole MePiA/liter, which is 1.1×10-4 grams MePiA/liter. Assuming a consumption of 3 liters of water per person per day, and using the model's calculated relative saturation intake of 1.1832, yields a maximum saturation intake of 400 micrograms of MePiA per day.
The maximum saturation intake calculated above should be regarded as a true maximum, not as a representative actual intake. It could easily be a factor of ten or more too high. The actual concentration of MeP in the oceans immediately post-Flood is not presently known. It may have been significantly below the saturation level the calculation assumed. In addition, the conversion efficiency of MeP to MePiA in the atmosphere may be significantly below the 50% used in the calculation.
To help gain more of a feeling for what is likely to be a reasonable optimum daily intake of MePiA, two additional methods of estimation are presented below.
Measurements of methanesulfonic acid (MSA) in glacier ice may be used as a crude proxy for MePiA.
MSA is a product of atmospheric chemistry acting on dimethyl sulfide (DMS), as mentioned previously.
DMS is emitted biogenically from the surface waters of the oceans. In contrast, MeP is emitted anaerobically from sea floor sediments, as discussed previously. The source strengths of these two processes can obviously be very different.
In addition, the efficiency for conversion of DMS to MSA in the atmosphere can obviously be very different from the conversion efficiency of MeP to MePiA.
Nonetheless, the concentration of MSA in glacier ice provides a ballpark figure for what the biogenic ocean source plus atmospheric chemistry system is capable of.
Measured values from Antarctic cores show considerable variation geographically and temporally, but an average within a factor of two of 10 micrograms of MSA/liter seems representative.[45]
Assuming 3 liters of water consumed per person per day gives a ballpark (proxy) intake of 30 micrograms of MSA/day.
A second ballpark estimate may be obtained using the body pool size of the traditional water-soluble vitamins as an estimate of the body pool of MePiA.
Loss of MePiA from the body is assumed to be governed by the equation X = X0 e-λt, where t measures elapsed time from termination of dosing. Thus δ X = - λ X δt. This equation can be used to compute the daily replacement intake of MePiA by taking X to be the normal body pool of MePiA and δt to be 1 day.
The lifetime for MePiA in the body has been calculated by the model to be 194 years. This gives λ = 1/194 per year = 1.4×10-5 per day.
All that is needed to complete the calculation is the body pool size.
The largest body pool size we can find is for vitamin C at roughly 1,500 milligrams.[46] The smallest is for vitamin B12 at roughly 2.5 milligrams.[47]
These two extremes for X give ballpark intake extremes of 20 and 0.033 micrograms/day respectively. This is a range of roughly 600, showing that they are indeed just ballpark estimates.
A more representative body pool size, based on vitamins B1, B6, and folic acid is roughly 25 milligrams. This yields a replacement intake for MePiA of roughly 0.33 micrograms per day.
There seems to us to be less reason for concern about overdosing than there is for underdosing with MePiA. This compound displays low acute oral toxicity.[48] The results of a study with rats showed an LD50 (lethal dose, 50%) of 940 mg/kg body weight (BW) with a 95% confidence interval of 476 to 1,858 mg/kg BW. This places the acute oral toxicity of MePiA 1.9 times lower than the 501 mg/kg BW minimum required for a compound to be considered to be in the low acute oral toxicity category.[49] For general reference on the toxicity scale, comparing to other oral LD50 values in rats, the LD50 of MePiA is 3.2 times more toxic than table salt (NaCl)[50], and 4.9 times less toxic than caffeine[51] (Figure 12.1).
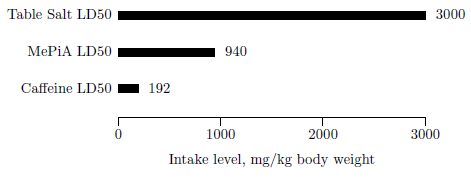 |
In addition to being of low acute oral toxicity, MePiA is not mutagenic.[52]
MePiA does not display chronic toxicity. MePiA will be highly dissociated at physiological pH based on its pKa value (pKa = 3.08), and will therefore not bioaccumulate over time. Experiments with mice at very high MePiA intake levels (designed to look for increased longevity rather than toxicity) have revealed only increased longevity and no evidence of chronic oral toxicity. For example, supplementation of the diets of 27 Hsd:ICR (CD-1®) female mice at 100 milligrams of MePiA per liter of drinking water beginning at 24.7 weeks of age and ending at 109.6 weeks of age served only to increase maximal life span by 20% relative to the control group not treated with MePiA.[53] This is equivalent to a human intake of roughly 250,000 micrograms of MePiA per day.
Water-soluble vitamins, especially those which are small acids, do not generally pose a risk of overdose, as any excess is readily removed from the body by the kidneys.
This property seems to be shared, as expected, by MePiA. Tolerance to overdosing with MePiA in humans seems to have been demonstrated by the full life spans enjoyed by both Noah and Shem who lived through the Spike, as well as by the first three post-Flood generations who were born during the Spike. All of these individuals appear to have been subject to very high MePiA intakes during the Spike, and these high intakes seem to have done them significant good and no harm, at least as far as longevity is concerned.
Meanwhile, underdosing is known to produce the ravages of "old age," dramatically reducing potential life expectancy.
We humans tend to fear the unknown. Compared to "aging," which has become comfortably familiar, vitamin MePiA is relatively unknown. But make no mistake, "aging" is the thing to be feared in this instance—it is the dread disease, and MePiA the welcome cure.
Taken together, the several estimates provided in this chapter suggest that the optimal intake of MePiA will likely be found to be within a factor of ten of a few micrograms per day—say 0.3 to 30 micrograms MePiA per day. With insufficient intake as the primary concern, prudence suggests that it would be better to err on the high side of this large range. We presently recommend 20 micrograms MePiA per day for adult males.[54] This recommendation, while functional at present, will, no doubt, be refined further in the future.
The model shows that, beyond a mere halting of Aging 1, rejuvenation takes place at high enough intakes of MePiA.
The life expectancy extension enjoyed by individuals living into the Spike was longer than the duration of the Spike itself (Figure 12.2).According to the model, the life expectancy extension for someone born in 4444 B.C. was 225 years. The duration of the Spike was just 69 years. These individuals were partially rejuvenated.
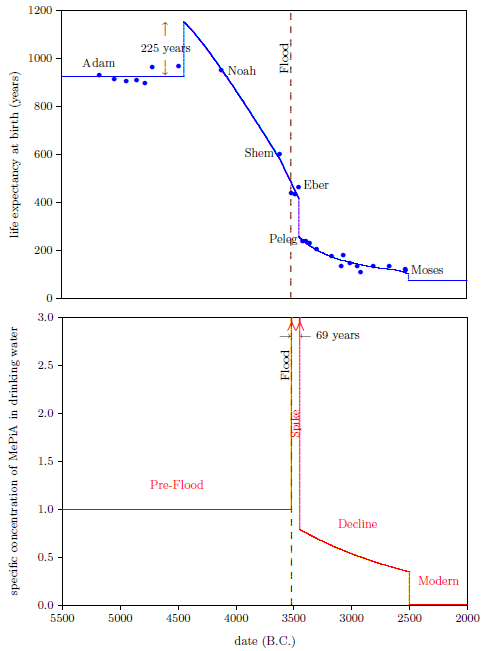 |
Thus, the model implies that the high intake rate of MePiA during the Spike did not merely halt further progression of Aging 1 during the Spike.Rather, the accumulated biological harm which had been caused by Aging 1 prior to the Spike was partially mended during the Spike—a net healing of free-radical damage took place during the Spike.
The cure for Aging 1 disease—vitamin MePiA—is in hand. To make it easily possible for individuals and families to obtain vitamin MePiA, we have made it commercially available in a dietary supplement called "Dr. Aardsma's Anti-Aging Vitamins."[55]

Humanity possesses reliable multicentenarian life span data in the Bible. These data show unequivocally that the human body is not designed to waste away and die by age 70 or 80 years.
The relatively brief current human life span may be explained as the inevitable outcome of a tragic, global, congenital vitamin deficiency disease of a previously unknown vitamin, dubbed "vitamin X." Thus, the ancient, biblically-recorded life span data instigate a quest for vitamin X. And these data, when coupled with modern scientific principles and methods, reward us with the discovery of vitamin X—methylphosphinic acid (MePiA).
MePiA is well suited to fill the role demanded of vitamin X. Significantly, it readily satisfies four criteria essential to the definition of a vitamin—not an easy thing to do. Traditionally, of the nearly infinite number of chemical compounds, only thirteen have been found able to satisfy these four criteria.
Vitamins are (1) organic compounds, (2) which must be obtained through the diet, (3) which are vital to the health of the organism, and (4) which are needed in tiny amounts only. That MePiA satisfies these four criteria is explicitly seen as follows:
It would be very nice if this were the end of the story. Aging would then be no more than a simple vitamin deficiency disease—methylphosphinic acid deficiency disease—and the cure for aging would then be no more difficult than to supplement one's diet with an adequate daily amount of this missing vitamin. The expectation would then be prevention of aging for the young, and of a more or less guaranteed, slow, steady progression back toward youthful health and vigor for the aged.
Unfortunately, real life is seldom simple, and the cause of aging is no exception. The biblical life span data have yet more to say about aging, and what these ancient data say is that aging is not only MePiA deficiency disease. They inform us that there is a second aging disease, Aging 2, to be reckoned with.
As we venture into discussion of this second aging disease, it will no longer be necessary to place the word, aging, in quotes. The quotes can be phased out because enough discussion has happened by this point to have corrected the meaning of the word. The popular misapprehension of aging as an abstruse natural process by which time converts youthful bodies into elderly bodies has now been displaced by the correct understanding of aging as universal congenital disease which converts healthy bodies into disease-ridden, disfigured bodies.

A second aging disease, Aging 2, is revealed by the shape of the survival curve for pre-Flood males.
Table 14.1 shows the seven data points used to construct the survival curve of interest, which includes only deaths due to aging. Enoch is excluded from this dataset because he did not die of aging.[57] Lamech is excluded because his low age at death (777 years) relative to the other pre-Flood males listed in Genesis 5 makes it seem unlikely that he died of aging.[58] In fact, it seems more probable that he perished in the Flood.[59] Methuselah, who may also have perished in the Flood, is nonetheless included in the list because his age at death, the largest in the list, implies that a misassignment error in his case would make little difference. Noah is excluded from the dataset because he lived both before and after the Flood.[60]
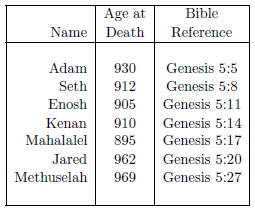 |
Figure 14.1 shows the resulting ancient survival curve dataset as orange dots. The vertical orange lines through the orange dots are error bars, indicating the (1σ) uncertainty in the percentage of survivors. For each data point, the life span of a single male individual is being used as an estimate of the average life span for the whole male population back at that time, and this gives rise to these error bars.
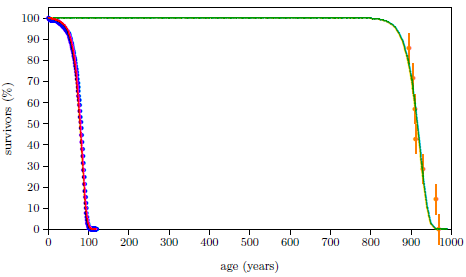 |
Also shown on the graph as blue dots is a modern survival curve for human males. The dataset for this curve is taken from the U.S. Social Security Administration's 2016 actuarial life table.[61]
A best-fit, smooth curve[62] is drawn through the data points in each case, providing further visualization of the shape of the survival curves in each instance.
The shape of the biblical dataset, made explicit by its fitted, green curve, is not what one would naturally expect. The pre-Flood deaths are all grouped together near 925 years. Since deaths due to aging today are spread out mainly between say 60 and 90 years, and since pre-Flood lifespans due to aging seem to be a little more than ten times modern lifespans due to aging, one naturally expects pre-Flood deaths due to aging to be spread out mainly between say 650 years and 950 years, not all gouped together near 925 years.
To see this most simply, imagine, for a moment, that Figure 14.1 represents two-cycle weed eater or trimmer machines rather than human males. Two-cycle engines require oil to be mixed with the fuel to lubricate the engines' moving parts during operation. If oil is not mixed with the fuel, or if too little oil is added, the rate of wear will increase, and the engines will "die" more quickly. Change the "age" axis scale in Figure 14.1 from years to something more suitable, like minutes of operation. Then the red curve represents weed eaters run with no oil in the fuel, and the green curve represents weed eaters run with some (but still not enough) oil in the fuel.
The green curve immediately seems to be wrong. One does expect weed eaters having some oil to live longer than weed eaters having no oil, but one does not expect weed eaters having some lubrication to die off in a tight time band similar to the tightly grouped die-off time band for unlubricated weed eaters. One naturally expects lubrication both to lengthen the average life span and to spread out the time between deaths. We expect this because lubrication slows wear, and, after the first machine has died due to wear, it is additional wear which causes the death of the second machine. Since wear has been slowed down, the spacing between deaths should spread out.
The magenta curve in Figure 14.2 shows what might naturally be expected for lubricated weed eaters. This is what one would naturally expect if wear had been reduced by a factor of 10. It is simply the red curve stetched along the time axis by a factor of 10.
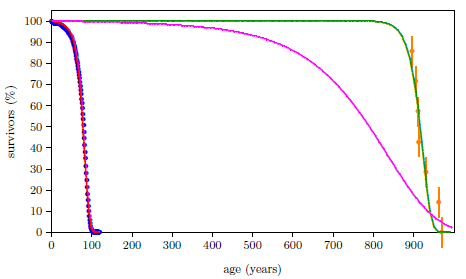 |
The ancient survival curve data are clearly not in harmony with this natural expectation. The green curve is significantly different from the magenta curve. The spacing between deaths for the red and the green curves in Figure 14.2 seems too similar for the large increase in average life span exhibited by the green curve.
These Genesis survival curve data are telling us something important about the nature of the aging process in humans. They are saying that aging prior to Noah's Flood was not simply modern aging stretched out over a longer time. Rather, pre-Flood aging seems to have been more like modern aging delayed by about 800 years. Evidently, pre-Flood individuals, after growing to adult maturity in the first few decades of life much as we do today, retained youthful adult bodies out to 800 years of age before they began to suffer (and ultimately to die of) aging.
Automobiles provide a useful mechanical example at this point, illustrating how survival curves shaped like the green curve may come about.
Imagine a cohort of 100 identical automobiles, fresh from the factory. As is normal for automobiles, they have an electrical system. Principal components of the electrical system are an alternator to generate electric current, a battery to store and supply electric current, a belt and pulley arrangement for turning the alternator armature, and wires delivering electric current to electrical devices.
Fresh from the factory, the automobiles are functioning normally, but the electrical systems are already aging. Of specific interest in the present context, the alternator belts suffer a tiny amount of wear with each revolution of their respective armatures. The automobiles are suffering alternator-belt-wear disease.
After many hours of constant operation, the alternator belts finally wear out and break, all pretty much at the same time. With the alternator armatures no longer turning, no electric currents are being generated, but the batteries keep supplying electric current, so the automobiles continue to run. Unfortunately, this results in a new pathology: drained-battery disease.
Drained-battery disease progresses at different rates for the individual automobiles, depending on the use of the various electrical devices such as radios, fans, interior lights, signal lights, headlights, etc. Symptoms of progression of the disease include slowly dimming headlights, slowing of turn signal blink rates, eventual stalling of the engines, and only clicking sounds when ignition keys are turned to restart the engines. Eventually, drained-battery disease runs its course, ending with the "deaths" of all of the automobiles.
Take-home points from this analogy are:
The biblical life span data reveal that human aging is a two-phase disease. It has an induction phase, (Phase I, the long plateau of the green curve in Figure 14.2), followed by an induced phase (Phase II, the short ski slope portion of the green curve in Figure 14.2). During Phase I, Aging 1 disease (i.e., MePiA deficiency disease) is active. The insidious presence of Aging 1 in Phase I results in the induction of another, separate aging disease, Aging 2, in Phase II.
Two-phase human aging can be explained fairly simply by the theory of vitamin X introduced previously. Recall that, in the theory of vitamin X, MePiA is the specific antioxidant needed to prevent free-radical damage to mitochondria (energy-producing organelles in cells). It is now clear that the simple idea of progressive damage to generic mitochondrial tissue must be abandoned. It cannot be responsible for two-phase aging all by itself because it is a single-phase phenomenon. Dysfunction of mitochondria in the case of accumulating damage to generic mitochondrial tissue would be expected to be simply proportional to the amount of accumulated generic damage. We now realize that we need a two-phase phenomenon. As with the automobile example, it is not general wear and tear on all parts which induces drained-battery disease. Rather, it is wear of one specific part: the alternator belt. Evidently, some specific mitochondrial tissue suffers free-radical damage over a long time before it finally breaks, triggering the onset of Aging 2. It is as if a switch has been flipped, starting up the Aging 2 disease process.
How does one suddenly switch on a biological disease? The simplest answer to this question is, "Genetically." Many biological diseases result from mutation of a single DNA base pair. Change a DNA base pair, and you may very well switch on a biological disease.
Now most DNA is in the nucleus of the cell. This is called "nDNA." But, as it turns out, mitochondria have their own relatively small amount of DNA. This is called "mtDNA."
Putting this all together leads to the postulate that the specific mitochondrial tissue being damaged by free radicals responsible for the onset of Aging 2 is mtDNA. Phase I then corresponds to free-radical damage of mtDNA, and Phase II corresponds to Aging 2, a disease of mtDNA, induced (switched on) by excessive accumulated damage of mtDNA by free radicals. Phase I is lengthened by addition of vitamin MePiA to the diet. This happens because of reduced mtDNA damage due to free-radical scavenging by MePiA in the mitochondria. Vitamin MePiA is acting as an exogenous nutrient antioxidant like vitamins C and E, but it plays a special mtDNA-protective role. Death of the organism results from progression of the induced mtDNA disease, Aging 2, not from free-radical damage per se.
This two-phase adaptation of the theory of vitamin X needs a new name, to differentiate it from previous theories of aging. Let us call it the "two-phase theory of human aging."
Two-Phase Theory of Human Aging: Human aging is a composite of the progression of two diseases:
- Aging 1: congenital vitamin MePiA deficiency disease, and
- Aging 2: an mtDNA disease induced by Aging 1.
Aging 1 alone is active in Phase I. Both Aging 1 and Aging 2 are active in Phase II.
Aging 1 may contribute in a minor way to aging deaths, but progression of Aging 2 is responsible for most aging deaths.
Vitamin MePiA is able to slow onset of Aging 2 or, if available in sufficient amount, MePiA is able entirely to prevent onset of Aging 2 by protecting mtDNA from free-radical damage.
Vitamin MePiA may exert its antioxidant activity throughout the body, but to prevent Aging 2, it is needed specifically in the mitochondria.

The two-phase theory of human aging developed in the previous chapter says that human aging involves two diseases:
We have seen that the cure for Aging 1 is in hand. The global eradication of this aging disease is now only a matter of time. Since Aging 1 induces Aging 2, the global eradication of Aging 1 will be accompanied by the global eradication of new cases of Aging 2. This is very good news. But, what about existing cases of Aging 2? Can the billions of individuals presently suffering from Aging 2 be cured, or are they yet doomed to die of "old age"?
To answer this urgent question, one must first understand the pathophysiology of Aging 2—the mechanism by which it originates in the body, and the mechanism by which it progresses in the body. The purpose of the present chapter is to elucidate this pathophysiology—to develop a theory of Aging 2.
According to the two-phase theory, aging progresses in humans as if it were controlled by mechanical outlet timers (commonly used by homeowners to turn lights on and off in their homes while they are away on vacation). Each member of the survival curve cohort has his own timer. In this analogy, the timers are all set to turn their outlets on at the same preset time and then leave them on. During Phase I, the mechanical timers run, ticking off the seconds, until the preset time is reached. At that point, they all flip their switches more or less at the same time, turning the power on to the Phase II process. The first task for the two-phase theory is to demonstrate that it can give rise to a plausible biological timed switch explaining Figure 15.1.
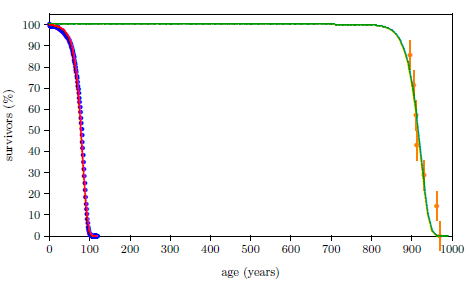 |
The simplest idea for a hypothetical switch design is probably that Aging 2 is switched on by a specific, free-radical-induced point mutation of mtDNA. Call this specific mutation mtDNAx. Free radical attack of mtDNA results in mtDNAx, and mtDNAx expresses itself clinically as aging of the organism. Without the mutation, there is no Aging 2 disease. With it, there is Aging 2 disease.
This simplest switch demonstrates that the two-phase theory provides a framework within which switches are possible. Beyond this, however, this simple switch is not very functional. It has the problem, for example, that there is no obvious timer associated with it. Free radicals are being generated all the time. Why, pre-Flood, did this switch turn on in all members of the cohort only at age 800 years?
A timer function demands a more complex type of switch. To obtain a functional timer, it seems necessary to postulate some sort of Poisson process. A Poisson process describes the occurrence of sequential unlikely events randomly in time. Here is an example of a Poisson process resulting from the bursting of water balloons.
Imagine using an air rifle to shoot pellets at water balloons. A single, water-filled balloon is suspended from an overhead, slanted rod by a string. If the balloon gets hit by a pellet, it bursts. At that point, another suspended water balloon slides down the rod and takes its place. There are 9 balloons total. The target balloon is hard to hit because it is pretty far away. In fact, a hit is scored only once per 100 shots on average. The shooter keeps firing until all balloons, one after the other, are burst. A list of the number of shots it took to burst each balloon is a Poisson sequence. The significance of this will be made clear in a moment. For now, notice merely that it takes some time for all the balloons to be hit.
Now imagine that there are lots of these water balloon setups, each with equally-skilled, independent shooters who start shooting at the same time and shoot at the same constant rate. We are trying to design timers which have the property that they will all flip their switches at nearly the same time. In the present example, we want all shooters to finish bursting all the balloons at nearly the same time.
Since the probability of scoring a hit is 1 in 100 (i.e., 0.01), on average it will take (9/0.01=) 900 shots for a given shooter to burst his 9 balloons. This is the average. In practice, some shooters will get lucky and do better than average, and some will get unlucky and do worse than average. Now here is the important thing, in the present context, about a Poisson process. Not only the average number of shots can be foretold but also the spread (technically, the standard deviation) in the actual number of shots around the average can be foretold. For a Poisson process, the spread is just the square root of the average. This allows us to know, for example, up front, before any shooting begins, that two-thirds of the shooters will have burst all 9 balloons within (√ 900 =) 30 shots of 900 total shots, and less than 1 shooter in 100 will take either fewer than 810 shots or more than 990 shots. Thus, this balloon-bursting timer has the property that we are looking for. All shooters will finish shooting at nearly the same time.
The timer can be improved by increasing the number of balloons. For 9 balloons the relative spread is (√ 900 /900*100=) 3.3%. For 900 balloons it is (√ 90,000 /90,000*100=) 0.33%. The relative spread can be made as small as we please by starting with a large enough number of balloons.
This example can be ported over into the two-phase theory of human aging as an analogy by identifying the individual shooters with free-radical-generating processes in individual mitochondria, the pellets with free radicals, and the water balloons with mtDNA within the cell.
The essential idea of the analogy then becomes irreparable damage of mtDNA due to free-radicals (this may correspond to DNA double-strand breaks, for example, and may take more than one "hit" to accomplish) necessitating replacement of the mtDNA copy. Irrepairable damage of the mtDNA copy, like the bursting of the balloon, is the Poisson event. Fundamental to the design of the switch is that there be a large but finite number of sequential replacement cycles possible before something happens, switching on Aging 2.
The idea of a finite number of replacement cycles of mtDNA suggests the possibility of a limit on the number of times mtDNA can be copied, presumably beginning back at the stem cell. This suggests that there may be some sort of mechanical problem with excessive copying of mtDNA, analogous to the telomere-shortening problem for nuclear DNA (nDNA).
Telomeres cap nDNA chromosomes. Replication of nDNA causes a mechanical shortening of the telomere cap. This shortening causes no loss of genetic information as the telomere carries no genetic information. But shortening of the telomere cap cannot go on indefinitely because the telomere has a finite length. Eventually, the telomere will be used up, and mechanical shortening will begin to eat into the genetic information section of the chromosome. Thus, somatic cells (i.e., body cells; not germ-line cells) cannot replicate nDNA indefinitely. (Germ cells have ability to add length back on to telomeres, so the germ cell line can go on replicating indefinitely, generation after generation.)
Human mtDNA does not have telomeres. Human mtDNA is a circular molecule having no ends needing to be capped. Clearly, something other than telomere shortening is going on with mtDNA. But the important concept to be grasped at this point is only that mtDNA may run into a replacement problem which is due to a mechanical limitation rather than being due to an mtDNA genetic mutation problem.
An important input to timed switch design is the length of the Poisson sequence that it must furnish. In the balloon analogy, this amounts to the question of how many balloons are needed to get the shooters to finish close enough to the same time. For human aging, the answer turns out to be a minimum of 2,200. That is, at least 2,200 sequential mtDNA replacement cycles must be possible. This number is calculated from the ancient and modern survival curves as follows.
Figure 15.2 shows death rates versus age resulting from the modern and ancient fit curves of Figure 15.1. For the modern dataset, the full width at half maximum of the frequency distribution of age at death is 28 years. For the ancient dataset, it is 49 years. Let X be the maximum allowable additional spread in full width at half maximum due to the switches not all switching at the same time. Then error propagation theory says that 492 = 282 + X2, which gives X = 40 years.
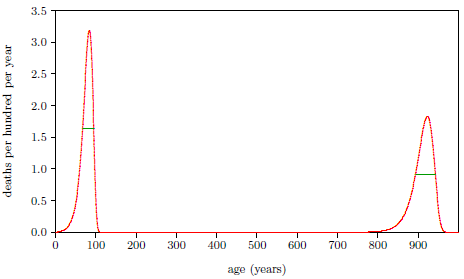 |
For the ancient dataset, the switch flips on after roughly 800 years. Let M represent the number of mtDNA replacements for a given mtDNA copy lineage beginning back at the stem cell during that 800-year counting interval. The standard deviation in this count, according to Poisson counting statistics, will be √ M . For large M, the Poisson distribution and the normal distribution become approximately the same. The full width at half maximum for the normal distribution is 2.35 times its standard deviation. This allows the standard deviation in the present case to be converted to full width at half maximum by multiplying by 2.35. So we wish to find M such that
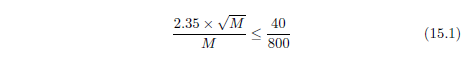
This yields M ≥ 2,200.
It is possible, at this point, to estimate another quantity of interest to the theory of Aging 2. This is the age of onset of Aging 2 at present. It is found to be 14 years, using the following method.
First, visually estimate the age of onset of Aging 2 for pre-Flood human males using the survival curve dataset shown in Figure 15.1. This is the point on the fitted green line when the plateau stops and the ski-slope begins. One finds roughly 800 years. (A more objective, mathematical analysis, finds 800 ± 1 years.[63])
The present interest is in the modern case. Before the Flood, MePiA was relatively abundant. Today, it is lacking. Given this pre-Flood estimate for the start of Aging 2, at what age might Aging 2 be expected to set in today?
Simple linear scaling can be used to transform this pre-Flood estimate into a modern estimate. First, use the Figure 15.1 graph again to estimate the age when there are 50% survivors. The objective mathematical analysis mentioned above gave 915.7 years of age for pre-Flood males. Thus, for the pre-Flood dataset, Aging 2 began (916 - 800 =) 116 years prior to the 50% survival age. The dispersion in the modern data is less than in the pre-Flood data, as shown in Figure 15.2. The full width at half maximum of the death rate distribution for the modern dataset is 28 years, while for the ancient dataset it is 49 years. Thus, Aging 2 may be estimated to begin (116 × 28 / 49 =) 66 years prior to the 50% survival age for modern males. Actuarial life table data for U.S. males in 2016 gives 50% survivors at 80.2 years.[64] Hence, this method of estimating the age for onset of Aging 2 for modern males yields (80 - 66 =) 14 years.
This is a first estimate only, mainly useful as a ballpark figure. The main thing it shows is that onset of Aging 2 today happens at a very young age.
Substantial progress has now been made toward development of a theory of Aging 2, but, at this point, a roadblock is encountered.
Prior to Noah's Flood, the age of onset of Aging 2 was 800 years. It is estimated to be 14 years today. Clearly, the onset of Aging 2 disease is regulated by a timer which is running very much faster today than it was pre-Flood. The ancient and modern life span data demand a biological timed switch, and this demands a Poisson process. We have tentatively identified the rate at which the timer ticks with the rate of free-radical damage of mtDNA, and the Poisson event with the replacement of mtDNA. Remaining to be identified is the biological switch responsible for the onset of Aging 2. It turns out to be surprisingly difficult to come up with a biological Poisson process giving rise to a suitable switch.
Not just any sequential process will be a Poisson process. A Poisson process involves sequential events whose exact timing from one event to the next is randomly distributed around some average time.
Imagine unlocking three equally spaced doors. Suppose the doors are side by side: Door 1 next to Door 2 next to Door 3. Then the time it takes to unlock the three doors will depend on the order in which the doors are unlocked. The order 1–2–3 will be faster than 1–3–2, for example, because of the extra time it takes to walk from Door 1 to Door 3 and then back to Door 2. The time it takes to unlock each of the three doors in this case is not randomly distributed in time—it depends on the order in which the doors are unlocked—so the unlocking of the side-by-side doors is not a Poisson process.
Now suppose that the three doors are located behind one another, blocking passage down a hallway, for example. It is then necessary to unlock Door 1 before one can get to Door 2 to unlock it, which then allows access to Door 3 to unlock it. In this case, the time it takes to unlock each door will be randomly distributed around some average door-unlocking time, and the unlocking of these sequential doors will be a Poisson process.
To find a biological Poisson process is, in itself, not a trivial exercise. To find a biological Poisson process followed by a suitable biological switch has proven to be extremely difficult.
Somewhat frustratingly, the telomere candidate mentioned previously works nicely, while no other candidate process seems to work at all.
As mentioned previously, telomeres are non-coding bits of nDNA which provide the ends of chromosomes with protective caps. Shortening of telomeres happens each time the cell divides and the chromosomes need to be replicated. Thus, shortening of a given telomere is a sequential process, and, like the sequentially unlocked doors or the sequentially burst balloons, the ordering of events is physically constrained. The second shortening can happen only after the first shortening has happened, etc. Taking cell replication for the adult body to be randomly distributed around some average replication time, a Poisson sequence results for a given chromosome from the times between the shortening of its telomeres.
A further boost for this candidate is that the fundamental, repeated, coded sequence for telomeres, TTAGGG, "is usually repeated about 3,000 times."[65] This implies a potential for about 3,000 balloons per telomere. That is, from a software engineering design perspective, one would expect the smallest removable piece to be TTAGGG, and there are 3000 of these pieces. This piece is expected to be a unit which is coding for something, otherwise it could just be TTTTTT. The TTAGGG sequence seems to be coding for a whitespace character—something like a blank (i.e., a space between words). To a computer, a blank is 00100000. The actual algorithm which executes each time a cell replicates may remove more than one blank at a time. But, from a software engineering design perspective, the potential is there to remove just one blank at a time, and this gives 3000 potential cycles, which is immediately in the same ballpark as the minimum 2,200 needed by the Aging 2 timed switch. A large number of balloons is evidently not a problem for telomeres.
Finally, excessive shortening of a telomere will not only remove it completely but will also result in loss of some of the coding region of the chromosome, inducing a guaranteed genetic mutation, which does act as a biological switch.
But there is a major problem with the telomere candidacy. As noted above, human mtDNA does not have telomeres. Human mtDNA is a circular molecule having no ends needing to be capped.
This yields a conundrum. The two-phase theory of human aging says that Aging 2 is an mtDNA disease. To begin to explain Aging 2 biologically seems to require that telomeres exist on human mtDNA, but human mtDNA is a circular molecule and thus has no ends and no end caps—no telomeres.
Much reflection on this conundrum suggests only one possible solution. The only way to solve this conundrum is via the bold postulate that human mtDNA must possess "in-line telomeres" (Figure 15.3).
 |
This postulate is "bold" because "in-line telomeres" seem to be previously unknown, making them to be purely hypothetical entities, newly invented to meet the present need.
The first and most obvious question which this bold postulate raises is, "What are 'in-line telomeres' for—what purpose would they serve?"
Actually, this question needs to be flipped on its head. A much bigger and more obvious question is, "How could human mtDNA manage to make do without any telomeres?" Allow us to explain the problem.
Maintenance of the blueprints is necessarily of highest priority for any organism.
A house is constructed by working from a set of blueprints. The blueprints code the idea of the building. Change the blueprints, and the idea changes. A different building gets constructed. The physical building is simply the tangible expression of the idea coded by the blueprints.
Living organisms are also constructed from blueprints. While house blueprints are made of paper and ink, biological organisms' blueprints are made of DNA.
The DNA codes the idea of the organism. The fish idea is quite different from the bird idea, for example, and the DNA code for the two is quite different.
In writing, we use words and sentences to code ideas. Here is an example of an idea coded in writing using a typewriter:
The box was red.We could use writing to code the idea for a house we wish to have built. We don't normally do so because the idea is much more easily and efficiently coded by means of a drawing—a blueprint.
Now here is the important point in the present context. Coded ideas are rather fragile things.
Watch what happens to the idea
The box was red.if a simple typing error is accidentally made while coding it:
The fox was red.Notice that the original idea has vanished and been replaced by a whole new idea.[66] A tiny coding error can be majorly destructive of a coded idea.
Most coding errors result in something unintelligible, like this:
The gox was red.In this case, the original idea has vanished and been replaced by gibberish.
For cells, coded gibberish represents erosion of the idea of the organism with concomitant loss of functionality. Cells are marvelously resourceful and able to withstand some loss of function, but the principal expectation of accumulating gibberish is eventual death of the cell.
For this reason, maintaining the fidelity of its DNA code is necessarily of highest priority for any organism.
Now, DNA risks loss of fidelity every time it is replicated. Replication is carried out by dedicated copy machinery within the cell. While this replication machinery is extraordinarily good—much better than any human typist—it is not perfect. For human mtDNA, the error rate is less than one mistake per million "letters" of the code copied.[67] This is a very low error rate, but when you consider that all humans start out as a single cell and wind up being comprised of about 37 trillion cells, typically containing thousands of mtDNA copies per cell, which cells then get replaced every 7 to 10 years, mtDNA copy errors are clearly a problem which must somehow be addressed if the organism is to survive and prosper.
How might a self-replicating biological machine be designed to minimize the impact of unavoidable mtDNA copy errors?
There seems to be only one possibility. It seems necessary, when making a new mtDNA copy, to use as a template the lowest generation-number mtDNA copy available.
Take the stem cell to be generation 0. The first mtDNA replication gives rise to a generation 1 copy. If the generation 1 copy is used as a template, the next new copy will be generation 2. If the generation 0 copy is used as a template again, one gets another generation 1 copy, distinct from the first generation 1 copy. Since each mtDNA nucleotide-copying event (analogous to copying the individual letters in a typed word) has a non-zero probability of producing a copy error, a generation 2 copy is likely to contain more copy errors than a generation 1 copy. A generation 2 copy will contain both the copy errors the generation 1 template had (which the copying machinery has copied to generation 2) plus any new copy errors made in the production of the generation 2 copy. Thus, the copy error density is proportional to the generation number.
To minimize copy error density, best design calls for the use of the lowest generation template available when making a new copy.
To implement this best design strategy, it appears necessary to tag each and every mtDNA copy with a generation number, so the lowest generation number copy can be selected from available copies for use as the template for making the next new copy.
Telomeres provide precisely this sort of tagging function. They not only cap and protect the ends of nDNA, they also keep track of how many times a cell has replicated.
Now you can see the problem. How does human mtDNA manage to get along, as the organism progresses from one initial cell to over 37 trillion cells, without this telomere function of keeping track of the generation number of each mtDNA copy so copy errors can be minimized?
The generation-number-tracking function of telomeres appears to be essential. This implies that it is essential for mtDNA to have "in-line telomeres."
This generation-counting function for "in-line telomeres" allows this new, hypothetical entity to be christened with a new name. A friend, Tom Godfrey (PhD linguistics), was asked to help with this. He suggested the Greek word "psephos." (The leading p is silent.)
Another English word, psephology, contains a good candidate. The initial Greek root in this word refers to a pebble. The Greek verb for the action of counting has the same root (https://en.wikipedia.org/wiki/Psephos), evidently because pebbles were once used for voting or counting. You may already know that calculus means small pebble in Latin, but I think you want to stick with Greek.[68]When asked about a "mere" ending, to place this entity in the same class as the telomere, Tom replied, "The Greek roots in telomere mean end and part, respectively, so psephomere works out to pebble/counter part."
"Psephomere" seems well suited to this mtDNA mechanical generation counter, based, as it is, on physical tokens (i.e., short bits of nucleotides). We will use it in place of "in-line telomere" from now on.
A second obvious question is, "Why is no psephomere evident in the published map of human mtDNA?"[69]
The answer to this question requires a second bold postulate. The second bold postulate is that the published map of human mtDNA is for mtDNA which has shortened its psephomere to zero.
This postulate is "bold" because it seems so unlikely at the outset. But, here again, this flips on its head. As soon as this postulate is advanced, a complete theory of Aging 2 suddenly emerges, and this theory immediately shows that mtDNA which has shortened its psephomere to zero is normal (which is not to imply healthy) today, so that a modern map of mtDNA is most likely to show the zeroed-psephomere molecule. Said simply, the zeroed-psephomere molecule is Aging 2—the shortening of the psephomere to zero is the event which flips the switch inaugurating Phase II—and because Aging 2 disease is presently normal to the global population for all individuals past roughly age 14 years, randomly chosen mtDNA is most likely to be of the zeroed-psephomere type.
The newly emerged theory of Aging 2 looks as follows.
Theory of Aging 2: Psephomeres keep count of the generation number of every mtDNA copy, enabling mtDNA copy errors to be minimized. The count is kept, as in the case of telomeres, by successively shortening the psephomere of each new mtDNA copy. Because generation 0 psephomeres are of finite length, excessive copying of mtDNA (due, for example, to a high incidence of free-radical damage necessitating frequent replacement of mtDNA copies) will result in eventual loss of psephomeres from the mtDNA population. These are Aging 2 diseased mtDNA copies. Loss of psephomeres breaks generation number counting, which breaks ability to minimize copy error density, which allows mtDNA copy errors (i.e., mtDNA genetic point mutations) to multiply rapidly, which results in exponentially progressing clinical Aging 2 disease over ensuing decades. That is, the expression of accumulated mtDNA copy errors results in increasing mitochondrial dysfunction, which results in increasing cellular dysfunction, which results in increasing whole organ and whole organism dysfunction, culminating ultimately in death of the individual.
In this theory, the shortening of the psephomere to zero is the long-sought mechanical biological timer, and the final decrement to zero of the psephomere is the associated switch which initiates Aging 2. The time required to shorten the psephomere to zero will depend on the rate of replacement of mtDNA, which will depend on the rate of damage of mtDNA due to free radicals, which will depend on cellular concentrations of vitamin MePiA. The depletion of the psephomere to zero breaks the generation counting function, which switches the mtDNA copy from healthy to Aging 2 diseased.

The most obvious test, to check the veracity of the theory of Aging 2, is to look in cells of young humans for healthy mtDNA. The younger the human, the longer the psephomeres are expected to be.
Because this test requires specialized equipment and techniques, a result from it seems likely to be many weeks or months away.
Another test, which may be carried out immediately, is to look for vestiges of psephomeres on the Aging 2 diseased-mtDNA map and its associated replication algorithm.
Imagine living on a remote island in the early 1920's. The island's population has heard about the wonders of the Model T Ford. Though never having seen one, they pool their resources and buy one. It arrives some months later after a long sea voyage. Having never seen a Model T, the islanders fail to realize that the exposure to salt spray on the long sea voyage rusted away the bolts holding the headlamps, causing the lamps to fall off and be lost at sea. The Model T is driven around the island for some weeks, but only during the day, since it is too dark to see where the car is going at night. Eventually, one islander gets the idea that headlights seem necessary for the car to be fully useful. He wonders why Henry Ford would fail to include them in the design of the car. He postulates that the car must originally have had headlights. How can he check his theory? He can look for headlamp vestiges on the car.
Going over the car carefully, he soon finds symmetrically placed sets of holes on the front of the car, one set on the left and one on the right. These holes seem to have no purpose. He surmises that they may have been put there to hold headlamps. Then he discovers broken-off wires dangling behind the holes. Tracing them, he finds that they run back to an on/off electrical switch on the dashboard. The islanders had wondered what the switch was for. Switching it on and off hadn't seemed to do anything. Now he has an explanation for the switch for the first time.
He takes the holes, the wires, and the switch—all essential components of an automotive headlamp system—to be vestiges of headlamps, and deems his theory, while not proven, to be confirmed beyond reasonable doubt.
Imagine a single mitochondrion containing multiple mtDNA copies. Imagine that one copy gets damaged and needs to be replaced.
The mitochondrion is a highly complex biological machine, much more complex than an automobile, for example. It cannot think, but it can sense its own state and choose between various options depending on what it has detected. This ability of a machine to choose options depending on conditions should not seem shocking. The thermostat on your home furnace does the same thing all the time, and it is a very simple machine. It senses the air temperature in your home. If the air is too cold, it flips a switch calling for more heat. When the air gets warm enough, it flips the switch back, shutting off the heat. Think of the mitochondrion as a very complex machine equipped with many sensors and many switches controlling many processes.
The mitochondrion senses the irreparably damaged mtDNA copy and signals for it to be replaced. How the damaged copy is scrapped is not of interest in the present context. What is of interest is the replacement process. Can vestiges of psephomeres be found in this replacement process?
To answer this question, it is necessary to review what is known about human mtDNA replication. Only those details deemed to be important in the present context have been selected in the following brief review. As we go through this review, bear in mind that what has been identified by us as Aging 2 diseased mtDNA is regarded as normal, healthy mtDNA within the published scientific literature on mtDNA at present.
Figure 16.1 shows a simplified schematic diagram of Aging 2 diseased (i.e., zeroed-psephomere) mtDNA in what we will call its resting state. Details of the various components of the diagram follow.
 |
Each mtDNA copy is made up of two, distinguishable strands called the L strand and the H strand. These two strands may be thought of as parallel rails of a railway track. The fact that, in real life, they spiral around each other as they go may be ignored for the present purpose, but the fact that they fit together must not be ignored. The two strands are complementary. They fit together the way two pieces of a jigsaw puzzle fit together, except that each strand is comprised not of just two puzzle pieces but of more than sixteen thousand puzzle pieces. These thousands of pieces interlock with the complementary strand all along the length of the track. Thus, the track may be thought of as held together like the two sides of a zipped up zipper.
The track goes in a circle. Think of the L strand as the inside rail and the H strand as the outside rail of this circular track.
An important thing to get right at the outset is that it is not the case that a template mtDNA is simply copied during replication to yield the original template as Copy 1 plus one entirely new mtDNA copy, Copy 2. Rather, the two strands of the template get split up. One of the template strands goes to Copy 1, and the other strand goes to Copy 2.
We will adopt the convention that the template L strand goes to Copy 1 and the template H strand goes to Copy 2. This is the order in which the two new copies are made—first Copy 1 and then Copy 2.
The mtDNA non-coding region (NCR) is of particular interest in the present context. The NCR takes up about 7% of the length of the track in the map of Aging 2 diseased mtDNA.
For purposes of copying mtDNA, mitochondria possess multiple copies of a special apparatus, called POLγ, which may be thought of as a monorail engine which rides one strand of a template mtDNA to make a new complementary strand, synthesizing one small segment of the complementary strand at a time. Synthesizing new strands of track is what POLγ is mostly, but not exclusively, about. It can also cut out segments of track, for example.
A side entrance to the NCR (called LSP, for light strand promoter) is situated toward one end of the NCR. It employs special apparatus to help POLγ get onto the track. This gets POLγ mounted on the L strand, facing down the track in the clockwise direction. POLγ is then set to begin scanning the L strand and synthesizing a new complementary H strand. When the new H strand is finished, these two strands, one old and one new, will then become Copy 1.
POLγ is observed to halt at TAS (termination-associated sequence) 95% of the time rather than going full circle to complete Copy 1. TAS appears to be a gate which is regulated by the mitochondrion, keeping POLγ parked or letting it continue down the track synthesizing the new H strand. The reason for this gated behavior is regarded as uncertain. It appears to us to be a vestige, as discussed below.
Parking POLγ creates a short three-strand section of track called the "D-loop." This is shown in Figure 16.1 as the short section of track at the top having three rails. The function or purpose of the D-loop is unknown. It appears to us to be another vestige.
When the TAS gate is opened, POLγ continues slowly around the track clockwise, scanning the template L strand to make the new complementary Copy 1 H strand as it goes (Figure 16.2). When it has completed about five eighths of the circular track, a mechanism automatically triggers for another POLγ to mount the template H strand out at that distance, in another, short non-coding region, to begin to copy the template H strand, producing its new Copy 2 L strand. The distance of five eighths—greater than one half of the circle—allows Copy 1 to be completed and the two template strands to be completely separated from each other so that POLγ 2 can complete its synthesis of Copy 2 without ever encountering—colliding with—the POLγ 1 synthesis.
 |
The synthesis of Copy 2 proceeds in a counterclockwise direction, back toward the NCR. Meanwhile, POLγ 1 completes its journey, encountering the starting end of the new complementary Copy 1 H strand it has been synthesizing. It is time to join the ends together. At this point, a curious behavior is observed with Aging 2 diseased mtDNA. POLγ is observed repeatedly to delete and remake a segment of strand at the joint. This behavior is currently called "idling." Idling appears to us to be yet another vestige.
From an engineering design perspective, idling makes no sense. It seems inefficient at best and pointless at worst. We suggest that idling may have been misnamed and misunderstood. We suggest that it may rather be "stuttering." It appears to us to be a vestige of the psephomere shortening step.
In a healthy mtDNA, once synthesis of the Copy 1 H strand has been completed, the new H-strand psephomere needs to be shortened to increment the generation counter. We suggest that on Aging 2 diseased mtDNA, POLγ 1 is unable to complete the psephomere-shortening step because the psephomere has already been shortened to zero. It tries—it deletes—but this triggers an error condition. So it rebuilds the deleted segment. Then it tries again, only to trigger the error condition again.
POLγ 1 sits there stuttering in this way because it is trying to deal with a worn out part. The psephomere has been used up. Eventually, it completes its preprogrammed number of delete cycles, which were intended to shorten the psephomere a fixed amount, and moves on.
This explanation of idling places the psephomere for healthy mtDNA at the very beginning of Copy 1, at the point where POLγ 1 first begins synthesizing the new H strand (Figure 16.3). This point is normally called OH, for the origin of H-strand replication. This location appears to us to be another vestige. It is in the non-coding region (the NCR), and like telomeres, psephomeres are non-coding.
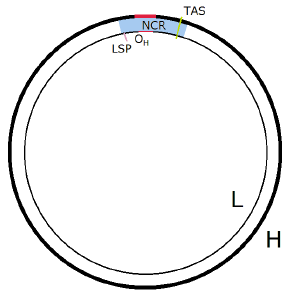 |
The existence of a loop in this region then also appears as a vestige. The shortening of the Copy 1 psephomere H strand will cause it to be shorter than the Copy 1 psephomere template L strand. To match the complementary H and L strands properly on either side of the shortened region—to be able to zip them together properly—requires that a loop be left to accommodate the extra L strand length.
The fact that Copy 2 does not begin copying at OH in the NCR, but rather five eighths of the circle away in another, short non-coding region, suggests the possibility that Copy 2 may not shorten either of its psephomere strands. Stuttering is seen only at the end of the synthesis of a strand, when a strand's two ends need to be joined together, and, for Copy 2, the psephomere is in the middle of the synthesis, not at the end (Figure 16.4), so there appears to be no psephomere shortening action for Copy 2.
 |
If this is correct, then the template L strand emerges as the "queen bee" strand. In sequential mtDNA replication events, this L strand produces "brood" H strands, all of the same generation, one generation higher than the queen.
To see this, focus on Copy 1 (e.g., the pair of H and L strands closest to center in Figure 16.4). On completion, Copy 1 contains a new H strand, but the queen (the template L strand) has survived and is now resident in the new Copy 1. This passage forward of the queen in the new Copy 1 may repeat itself over and over, for however many replications may be needed. This means that the template L strand is the template, which gets used over and over. It makes a Copy 1 H strand, which is always just one generation higher than itself each replication, but then this newly synthesized H strand gets cast off as Copy 2 in the next replication. Thus, the Copy 1 L strand is the queen, which is capable of casting innumerable Copy 2 brood, all of the same generation number, one generation higher than the queen.
Because Copy 2 replicates its template H strand without shortening either strand of its psephomere, the new Copy 2 L strand psephomere winds up recording that it is one generation higher than the queen. One may think of Copy 2 L strands as princesses—daughters of their queen mother.
Unfortunately, queens are not immortal. The template L strand is certainly as much subject to fatal free-radical damage as any other L strand, and it may be even more susceptible to fatal free-radical damage during copying than non-queen L strands. When the queen is killed by free-radical damage, she must be replaced. A new queen must be chosen from her mitochondrion hive. The logic of generation counting to this point implies that the mitochondrial hive will be inhabited only by sisters to the queen or by princesses (Figure 16.5). If a sister exists, then she needs to be chosen as the next queen since she will be one generation closer to the original blueprint than a princess. The eventual loss of all sisters will cause a princess to become queen. This increments the generation number of the queen and corresponds to the bursting of one balloon.
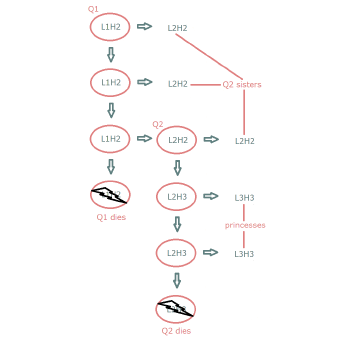 |
Now notice that it is because all sisters must be lost before the generation number can be incremented, in the case of functioning psephomeres, that multiplication of mtDNA copy errors is slow for functioning psephomeres and rapid for zeroed psephomeres. This explains why mitochondria have multiple copies of mtDNA rather than just a single copy, making this also to be a telltale psephomere vestige. The mechanics of choosing a new queen appears to us to expose yet another vestige. To choose the next queen from the mitochondrial hive of available copies, it is necessary to read and compare the psephomere L strands of all existing mtDNA copies in the mitochondrion. To read the psephomere L strand, POLγ must scan it. Like the on/off electrical switch and dangling wires behind the holes in the hood of the Model T, this suddenly gives a reason for the existence of the D-loop. To read the psephomere length, POLγ must scan the NCR L strand. In doing so, it simultaneously synthesizes the complementary H strand, up to TAS. This creates a short, three-stranded region continuous with the initial loop—creating the D-loop.
And now another vestige appears. Once the psephomere L strands have been read, all POLγs of all mtDNA copies must wait upon a decision of who is to be queen. This explains why TAS exists. TAS opens only to the new queen.
The replication algorithm detailed above is incomplete. For example, it does not specify how queenship is bestowed, nor how queenship opens just the queen's TAS gate while all others remain closed. In addition, it seems unlikely that every detail of this replication algorithm is correct. For example, it seems at least possible that queen sisters may cast brood simultaneously rather than sequentially. But the point seems adequately made nonetheless. The map of Aging 2 diseased mtDNA and its accompanying algorithm for replication do indeed appear to exhibit psephomere vestiges. The curious idling delete cycles just when and where psephomere shortening might be anticipated, the unavoidable need of a loop to take up the slack once the Copy 1 psephomere H strand has been shortened, the apparent lack of psephomere shortening for Copy 2 implying a queen bee mitochondrial hive necessitating multiple mtDNA copies, the need to scan the psephomere to read the generation number thus giving rise to a three-stranded D-loop region, and the existence and placement of TAS all appear to be vestiges which, while likely not an exhaustive list, are nonetheless sufficient to credibly support the theory of Aging 2.

The theory of Aging 2 refines the two-phase theory of human aging, making more specific its previously generic "Aging 2: an mtDNA disease…" as follows:
Two-Phase Theory of Human Aging: Human aging is a composite of the progression of two diseases:
- Aging 1: congenital vitamin MePiA deficiency disease, and
- Aging 2: zeroed-psephomere disease, induced by Aging 1.
The two-phase theory is explicit about the role played by vitamin MePiA deficiency disease in inducing Aging 2. The fact that a dietary deficiency of vitamin MePiA induces Aging 2 immediately implies a preventive role for MePiA with respect to Aging 2—one might say that vitamin MePiA is nature's prophylactic for Aging 2 disease. While this has been clear since discovery of Aging 2, the urgent question has been whether MePiA might also play a curative role with Aging 2.[70]
This question is urgent because Aging 2 disease is by far the leading cause of death today. The advent of the theory of Aging 2 allows this urgent question finally to be answered. Happily, the answer is found to be affirmative. According to the theory of Aging 2, vitamin MePiA can both prevent and cure Aging 2 disease.
The theory of Aging 2 says that loss of mitochondrial DNA (mtDNA) psephomeres is the fundamental cause of Aging 2. Psephomeres, somewhat analogous to telomeres, keep track of the generation number of each mtDNA copy as part of a mitochondrial copy error suppression mechanism. Loss of psephomeres breaks this mechanism, which allows copy-error mutations to accumulate rapidly within the mtDNA pool. To cure Aging 2, restoration of mtDNA psephomeres is required.
The idea of reaching into the mitochondria of all 37 trillion cells of the human body to restore psephomeres to thousands of copies of mtDNA per cell does not appear to be technically possible at present, nor would one wish to employ this approach even were it technically feasible. Loss of psephomeres results in accumulation of mtDNA genetic mutations. Restoring psephomeres to mutated mtDNA restores ability to suppress further mtDNA copy-error mutations, but it does not correct already existing copy-error mutations. Thus, Aging 2 would be cured, but existing mitochondrial genetic diseases induced by Aging 2 would not be cured. To do the job right, not only the psephomere but also the rest of the mtDNA molecule needs to be restored to its original condition.
Fortunately, according to the theory of Aging 2, the body's own healing mechanisms, when armed with an adequate provision of vitamin MePiA, do the job right. This is revealed by the temporary reversal of aging which Noah experienced during the Spike.
The modeling study of pre-Flood life spans made explicit the fact that all pre-Flood individuals were afflicted with congenital Aging 1 disease. They were all somewhat deficient in MePiA, though not nearly so deficient in this vitamin as are modern humans.
The modeling study also revealed a brief period of time (69 years), called the Spike, immediately following the Flood, when natural environmental levels of MePiA were temporarily very high.
Noah had Aging 1 disease when he entered the Spike. Though he was 600 years old when he entered the Spike, he did not have Aging 2 disease. Aging 2 was induced in individuals living before the Flood only at age 800 years, as discussed previously. The only aging disease which Noah had when he entered the Spike was Aging 1 disease, with which he had been afflicted since birth.
Because of his congenital Aging 1 disease, Noah was three-quarters of the way (i.e., 600 years of 800 years total) to onset of Aging 2 disease when he entered the Spike.
Upon entering the Spike, Noah's chronic dietary deficiency of MePiA was quickly replaced by dietary sufficiency of MePiA because of the large environmental abundance of MePiA during the Spike. Noah's Aging 1 disease was healed at that point. The free-radical flames which had been burning deep inside Noah's cells were quenched by MePiA. But 600 years of accumulated cellular damage remained. Happily, the biblical life span model found that Noah's body began to reverse this damage during the Spike. Noah experienced a partial rejuvenation because of the Spike. By the end of the Spike, the extent of the damage had, in fact, reverted back to what it had been 70 years prior to the Spike.
Noah's partial rejuvenation during the Spike is an extremely important result. How robust is this result?
To answer this question as objectively as possible, the model of the biblical life expectancy data was run again, this time excluding potential for rejuvenation. The model includes data from 19 ancient males, but only two of these, Noah and Shem, were born before the Flood and lived through the Spike. Since, in real life, rejuvenation was only operable during the Spike, and since neither Noah nor Shem were seriously deficient in vitamin MePiA prior to the Spike, the sensitivity of the model to this test was not expected to be large. Nonetheless, the no-rejuvenation test case resulted (1) in a worsened goodness-of-fit parameter,[71] implying that rejuvenation was 55% more likely to be the truth than no rejuvenation, and (2) in the actual life spans of both Noah and Shem being longer than the no-rejuvenation case predicted, Noah by 10 years and Shem by 32 years, further implying that, in real life, some rejuvenation had likely occurred.
This objective mathematical result is reinforced by common experience with the traditional vitamin deficiency diseases, all of which show healing—dramatically for some of the vitamins—when the deficient vitamin is restored to the diet in adequate amounts.
Finally, healing of the damage caused by Aging 1 may be expected based on the basic biology of normal cell replacement. Reversal during the Spike of the damage caused by Noah's prior Aging 1 disease would mean that his biological timer, regulating the length of Phase I, not only stopped advancing but began to run backwards during the Spike. Shortening of psephomeres is the mechanism by which the biological mechanical timer controlling onset of Aging 2 decrements time remaining on the timer. For the timer to run backwards, incrementing its time remaining, would mean that Noah's psephomeres, which had been declining in average length during the first 600 years of his life, increased in average length during the Spike. Basic biology shows that this may be expected due to normal replacement of cells once adequate cellular levels of MePiA have been achieved halting excessive free-radical damage of mtDNA.
Biological tissues typically need to maintain a fixed number of cells. For many tissues, this is accomplished by cellular replacement. While the details of cellular replacement vary enormously by tissue and cell type, cells that are damaged or dead are generally able to be replaced, either on a fixed schedule or as needed by events such as tissue injury.[72]
In a simplistic version of cell replacement, when a cell in a tissue dies, adult stem cells in the same tissue provide a new replacement for the dead cell. This could, in principle, be a direct replacement of one stem cell dividing asymmetrically to maintain the stem cell while producing a daughter cell that terminally differentiates (matures) and replaces the dead cell. Or it could involve one stem cell dividing asymmetrically to maintain the stem cell while producing a daughter cell which then goes through a series of divisions in a process called transient amplification, producing many precursor cells. One of these precursor cells of an appropriate generation (correct number of divisions) will terminally differentiate into the replacement for the dead cell. This terminally differentiated cell is what actually performs the work of a particular cell type (e.g., transporting oxygen if it is a red blood cell). The precursor cells are there only as part of the process of making replacement differentiated cells.
Cellular replacement is typically an infrequent process. The average age of cells in the human body today is 7 to 10 years. This is an average. Some body tissues replace themselves much more rapidly while others replace themselves much more slowly. But the important point is that stem cells have potential naturally to replace differentiated cells.
It is well known that cell replication leads to shortening of telomeres. Telomeres keep track of how many generations a given cell is removed from the stem cell. When a stem cell divides to produce a daughter cell—the daughter dividing to ultimately produce many terminally differentiated cells—the daughter will start out with long telomeres. Thus, it seems reasonable to expect that, when stem cells produce daughter cells, the daughter cells will also have long psephomeres.
Because psephomeres are parts of mitochondria, and mitochondria are parts of cells, normal cell replacement is thus expected to result in slow restoration of long psephomeres, once adequate cellular levels of MePiA have halted the accelerated shortening of psephomeres caused by excessive free-radical damage.
Putting this all together, the results of the model, the example of the traditional vitamins, and the expectation from the basic biology of cell replacement all appear to support the case for rejuvenation during the Spike reasonably strongly.
Noah's rejuvenation during the Spike implies that not all cells of the body advance in psephomere shortening at the same rate. This means that stem cell psephomeres shorten more slowly (if at all) than differentiated cell psephomeres. This is likely because adult stem cells of many tissues are largely in a dormant state, only "waking up" when they are needed to produce replacement cells for the tissue. This, coupled with ability of a single stem daughter cell to divide into multiple precursor cells, limits the number of divisions (and thus generations) stem cells experience. In addition, many stem cells have telomerases, enzymes designed to keep the telomeres from shortening. Thus, it seems likely that stem cells are either not subject to Aging 2 at all, or that the induction of Aging 2 in stem cells is significantly delayed relative to differentiated cells. And this means that as long as at least some of the stem cells are free of Aging 2 in a given tissue, once the body's internal free-radical inferno has been quenched by adequate cellular levels of vitamin MePiA, Aging 2 diseased cells in that tissue, rendered dysfunctional to varying degrees by their disease, will slowly be replaced by healthy cells furnished by the healthy stem cells, curing Aging 2.
To check our understanding of the principles and processes underlying the progression and healing of Aging 2 discussed to this point, a quantitative computer model was developed.
The model tracks progression and healing of Aging 2 in a multi-cellular biological "organism." The organism is extremely rudimentary. It may be thought of as a spherical ball of cells, though the algorithm, in fact, specifies no geometrical shape. The organism matures instantly and lives in a steady state maintenance condition. That is, dysfunctional cells are constantly being replaced via stem cells. One may picture, for example, a single, immortal, generation-zero stem cell in the center of the spherical body. This generation-zero stem cell, through a process of transient amplification of "stemmy" cells, provides, after 12 generations, 4,096 terminally differentiated working cells for the organism, all of the same basic tissue type. This is, clearly, an extremely rudimentary organism. The human body has 37,000,000,000,000 cells of many different tissue types. Nonetheless, this simple model is able to provide the check of basic principles and processes needed at this point.
Stemmy cells are assumed to be relatively few in number. The algorithm does not need to keep track of them, as they are assumed not to be subject to Aging 2. The idea here is that the differentiated cells are responsible for doing the wear-and-tear, environment-interacting business of the organism, while the stemmy cells are largely responsible for maintaining the fidelity of the organism to its design idea, coded in its DNA blueprint. As a result, stemmy cells spend much of their time in a low-work "sleep" mode, while differentiated cells are constantly at work.
To use this digital model organism to study Aging 2, it is necessary to keep track of what is happening with the psephomeres within each cell. To keep things simple, the model allocates 100 copies of mitochondrial DNA to each cell in an intracellular mitochondrial network. ("Mitochondria are dynamic organelles with the ability to fuse and divide (fission), forming constantly changing tubular networks in most eukaryotic cells,"[73] and "Fusion of mitochondria serves to mix and unify mitochondrial compartments."[74]) This means that the computer algorithm needs to keep track of the lengths of 100 psephomeres per cell of the organism. This comes to 409,600 psephomeres total.
The algorithm loops through time cycles, replacing terminally differentiated cells each time cycle. Each cell is reviewed for replacement every cycle. During the review, a random number is generated and compared to the preset probability of replacement of the cell. If the random number is greater than or equal to the preset probability, the cell is replaced.
The probability of cell replacement is made proportional to the average generation number of the mtDNA copies within the cell. This codifies two ideas: (1) that cells are replaced only because they have become dysfunctional, and (2) that mtDNA copy errors are taken to be the sole cause of dysfunction, the average generation number of mtDNA copies within the cell being proportional to the number of copy errors (i.e., mutations) made to the mtDNA. This is, once again, an obvious oversimplification compared to the physiology of the human body, but it is nevertheless functional for the present purpose of checking our understanding of Aging 2.
The generation-zero stem cell ultimately yields generation-twelve terminally differentiated cells having psephomeres of length (100 - 12=) 88, the psephomeres starting out with length 100 and having been shortened once for each mtDNA replication event resulting from the twelve cell divisions leading to the terminally differentiated cell. Thus, when a cell is randomly chosen for replacement, it is replaced by a cell having all psephomeres of length 88. This means that a generation-twelve cell's psephomeres can each be shortened 88 times before reaching the Aging 2 disease condition of zero psephomere length remaining.
Replacement of randomly chosen mtDNA copies also happens each time cycle, with consequent shortening of psephomeres. The probability of mtDNA replacement is a constant, reflecting the uniform random risk of non-repairable mtDNA damage from free radicals. However, the rate of advancement of mtDNA generation number is not constant. A relatively low rate of advancement of mtDNA generation number pertains up to the advent of Aging 2, due to the use of the "queen bee" replication mechanism, which always uses the lowest generation number copy as a template for making the replacement copy. Once the advent of Aging 2 breaks this mechanism, replacement mtDNA copies are made from any other copy in the mitochondrion, chosen at random. This results in a much increased rate of advancement of the mtDNA generation number, which yields a much increased rate of production of mtDNA copy errors. Since the probability of cell replacement in the model is based on mtDNA generation number, this increased rate of advancement of mtDNA generation number results in an increased probability of cells being replaced, which results in an increased rate of cell replacement.
The algorithm keeps track of the average psephomere length of the organism with time. This measures progression of the organism toward induced Aging 2 disease. The algorithm also keeps track of the percentage of psephomeres which have been shortened to zero with time. This measures progression of Aging 2 in the organism. Note, however, that progression of Aging 2 in the organism is not equivalent to progression of Aging 2 deaths in a population of organisms. The spread of zeroed psephomeres to more and more cells of the organism does not immediately translate into increasing sickness and probability of death of the organism. Zeroed psephomeres are, in and of themselves, harmless. Zeroed psephomeres are bad only in that they allow copy errors to accumulate rapidly. It is these copy errors, resulting in genetic mutations, which, over time, are ultimately responsible for the death of the organism.
As a first check, the model was used to see whether onset of Aging 2 near 800 years could be achieved. Figure 17.1 shows that it could. The blue line shows the average length of the organism's psephomeres. The red line shows the percentage of the organism's psephomeres which have been shortened to zero. The incidence of the first zeroed psephomeres represents onset of Aging 2 disease in the organism.
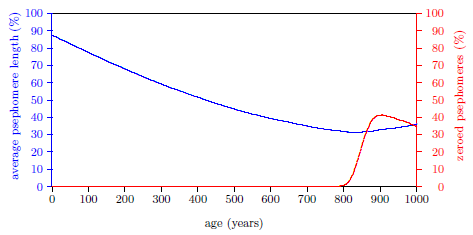 |
For this solution, the half-life for mtDNA replacement was set to 1.1 years.
The second check asked whether, leaving the half-life for mtDNA replacement at 1.1 years, the model could yield regression away from induction of Aging 2, similar to that which Noah experienced during the Spike, (i.e., regression of average psephomere length by the end of the Spike back to what it had been when Noah was 530 years of age). To test this case, the probability of psephomere shortening was set to zero for 69 years beginning at 600 years, simulating Noah's age and his experience with very high intake levels of MePiA during the Spike yielding cellular saturation of MePiA. Figure 17.2 shows that this was also possible. By the end of the Spike (brown vertical line), average psephomere length had returned to what it had been at 530 years (green vertical line).
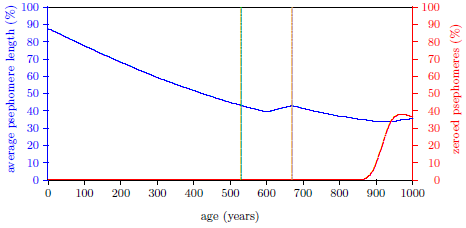 |
Notice that this regression of average psephomere length caused Aging 2 induction to be significantly delayed beyond 800 years. This emulates the lengthening of Noah's life span due to the Spike, as found by the biblical life span data model.
For this test run, the probability of cell replacement in a given year was set to 1.61×10-5 times the average mtDNA generation number per cell.
The third check asked whether the model could yield onset of Aging 2 near 14 years if the half-life for mtDNA replacement was decreased (to emulate the high rate of free-radical damage in modern times) while all else was left the same. Figure 17.3 shows that this was also possible.
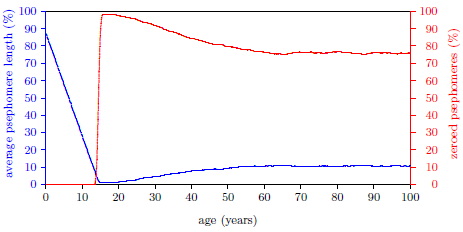 |
The red curve peaks initially, before declining to a steady state. This same behavior is hinted at in the previous two graphs, but it is made conspicuous here. This results from the mtDNA generation number continuing to advance beyond the zeroing of the psephomere. (The mtDNA copy is no longer able to keep track of this number once the psephomere has been zeroed, but the program is able to keep track of it.) As the generation number continues to advance, the probability of cell replacement continues to increase, so steady state in the rate of cell replacement, and hence in the number of zeroed psephomeres, is achieved only subsequent to the onset of Aging 2. The half-life for mtDNA replacement for this solution was 0.019 years (i.e., about 1 week). Thus, the model finds that the mtDNA replacement rate is 58 times faster for the modern emulation than it is for the pre-Flood emulation.
The foregoing, first-approximation digital organism model neglects a lot of biology. For example, it entirely ignores mitophagy, by which individual, dysfunctional mitochondria within a cell may be selectively replaced. Nonetheless, this simplistic model is able to reproduce key features of the progression and healing of Aging 2 disease. This success validates our understanding of the principles and processes underlying the progression and healing of Aging 2. It also recommends use of the model for learning more about Aging 2.
The first helpful use of the model is in providing a second, independent estimate of the modern age of onset of Aging 2. This is accomplished by making use of the observed 7 to 10 years average age of modern human cells. Figure 17.4 shows the average cell age (green line) calculated by the model for the foregoing case of onset of Aging 2 at 14 years.
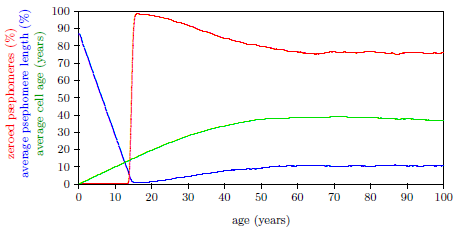 |
The average cell age is too large for much of the time range of interest, approaching 40 years by age 50 when it should stay below 10 years the whole time.
To decrease the average age of the digital organism's cells, it is necessary to increase the rate at which cells are replaced. The computer program constant which directly adjusts the rate at which cells are replaced has been "calibrated" using Noah's rate of rejuvenation during the Spike (Figure 17.2). It is no longer free to be adjusted. To increase the rate at which cells are replaced, an indirect adjustment is necessary. This can be accomplished by increasing the rate at which mtDNA is replaced. This will cause cells to become dysfunctional at a faster rate, which will cause them to be replaced at a faster rate. Increasing the rate at which mtDNA is replaced implies increasing the rate of free-radical damage of mtDNA. Increasing the rate of free-radical damage of mtDNA will obviously bring on the advent of zeroed psephomeres more quickly, shifting the age of onset of Aging 2 to younger ages.
Figure 17.5 shows a solution for which the average cell age lies between 7 and 10 years from early childhood onward.
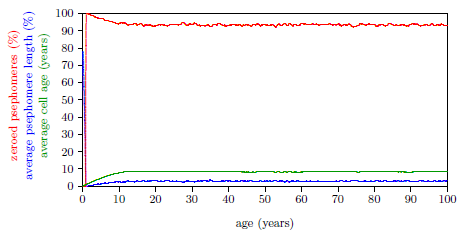 |
The age of onset of Aging 2 for this solution is roughly 0.9 years. The half-life for mtDNA replacement for this solution was 0.00117 years (i.e., about 10 hours). This yields a replacement rate for mtDNA 939 times larger than for the pre-Flood emulation.
This new estimate of the modern age of onset of Aging 2 is significantly different from the first estimate of 14 years. This is not too surprising. The first estimate relies on a large extrapolation of pre-Flood data into modern times, and this second estimate relies on a seriously oversimplified model.
It is difficult, at this stage, to know which of these two estimates is likely to be more accurate. The average of the two is 7.5 ± 9.3 years. Notice that this opens the possibility that the onset of Aging 2 may occur in the prenatal period. Prevention of Aging 2 in such a case would require that the mother be supplementing her diet with MePiA during pregnancy. But the main take-away from this average is that the age of onset of Aging 2 for modern individuals not supplementing their diets with the anti-aging vitamins is not well known at this stage, other than that it is probably somewhere in the first two decades of life.
The digital organism model, most importantly and helpfully, provides a means of estimating the healing rate of Aging 2. It allows the recession of zeroed-psephomere cells (once MePiA supplementation has begun, halting further free-radical damage of mtDNA) to be calculated. The consequent decline in the percentage of zeroed-psephomere cells is shown in Figure 17.6 for a number of representative initial supplementation ages: 5, 35, 65, and 95 years.
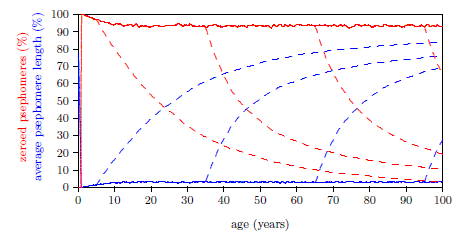 |
We have used the previous solution (Figure 17.5) as a basis for this calculation deliberately to cover the worst case scenario of needing to heal Aging 2 from very early childhood.
These curves illustrate mainly that the healing of Aging 2 is a slow process, lasting decades. For most starting ages, it takes 11 years for half of the zeroed-psephomere cells initially present to be replaced. For the 5-year-old case, which begins before steady state has been achieved, this takes 17.5 years, and there remains a few percent of zeroed-psephomere cells even at 100 years of age.
It seems likely that the human body, which is much more sophisticated than this simple model when it comes to dealing with dysfunctional cells and organelles, will do better at clearing out Aging 2 diseased cells than the model calculates. Nonetheless, it seems clear that the healing rate of Aging 2 will be measured in decades, not days, weeks, months, or even a few years.
The digital organism model implies that modern humans adequately supplementing their diets with MePiA can expect their bodies slowly to heal of Aging 2 disease. Notice, however, that the impact of this healing of Aging 2 on health and longevity is complex. While Aging 2 is simply zeroed-psephomere disease, zeroed-psephomere disease induces mtDNA-copy-error disease. Copy errors yield mtDNA genetic mutations. Thus, Aging 2 may induce a host of potential mtDNA genetic mutation diseases, some of which may have ultimately catastrophic consequences.
Thankfully, Aging 2 induced mtDNA mutations are not congenital genetic mutation diseases. They have not propagated from the zygote (the organism's very first, starting cell). While congenital, hereditary mtDNA genetic mutations might be expected to be present in every cell of the body containing mitochondria, Aging 2 induced mtDNA genetic mutations will initially be local to specific (unlucky) mtDNA copies in specific (unlucky) mitochondria in specific (unlucky) cells. One may think of progression of the clinical expression of Aging 2 disease as being due to new mtDNA genetic diseases of varied sorts sprouting up in local tissues all over the body. These diseases then get propagated by replication in several steps:
Meanwhile, a certain "inertia" seems inevitable with the progression of this mess—progression of this mess will not be brought to a halt overnight. Aging 2 seeds cells with mtDNA mutations. This starts happening early in life. These seeds take decades to come to their potentially dreadful harvests. Any given mutation itself takes years to spread in the body as the diseased cell gets replicated, making more cells having the same mutation, and the biochemical mess-ups a mutation causes take decades to cascade to their final harmful potential. Vitamin MePiA does not have the power immediately to stop any of this already existing mtDNA mutational mess. MePiA immediately stops the further compounding of trouble which would have resulted from further free-radical damage and further copying of mtDNA seeding yet more mtDNA mutations in the absence of MePiA. Additionally, MePiA immediately slows down the spread of existing mutations—probably pretty dramatically, but not to zero. Consequently, initial, slow progression of Aging 2 induced mutation diseases seems likely.
Whether this mess can be cleaned up in its entirety by adequate cellular levels of vitamin MePiA is, so far, unknown and seems likely to vary from individual to individual. Nevertheless, two important things are known and should not be lost sight of:

The ancient, biblical life span data reveal that human aging is not due solely to progression of vitamin MePiA deficiency disease. These data reveal that human aging is a two-phase disease. Congenital Aging 1 disease induces a second aging disease, Aging 2.
Aging 1 disease is insufficient cellular levels of MePiA, with excessive free-radical damage of mtDNA the result. Aging 2 disease is zeroed psephomeres, with excessive mtDNA copy errors the result.
The physiological outworking of Aging 2 is the major cause of aging deaths. Morbidity and mortality due to Aging 2 disease may be attributed to multitudinous mtDNA mutations, due to excessive mtDNA copy errors, due to loss of mtDNA psephomeres, due to an excessive need for replacement of mtDNA, due to excessive free-radical damage of mtDNA, due to cellular insufficiency of MePiA.
Adequate dietary supplementation with MePiA appears not only to prevent onset of Aging 2 but also slowly to cure this disease.

Aging 1 and Aging 2 are sufficient to explain the ancient biblical life span data, but they are not sufficient, by themselves, to explain modern life span data. Human aging as it is experienced at present involves yet a third aging disease. This third disease is MePA (methylphosphonic acid) deficiency disease.
At this point in this development of the causes and the cures of the human aging diseases, one might expect this third aging disease to be called "Aging 3." In fact, because it was discovered prior to the discovery of Aging 1, we call it "Aging 0." "Aging 1" and "Aging 2" were chosen as names to match with Phase I and Phase II when the fact that human aging is a two-phase disease was finally discovered. This made "Aging 0" the logical choice for the previously discovered MePA deficiency disease.
Aging 0 (MePA deficiency disease) is not expected to have been a part of human aging prior to the Flood because MePA is expected to build up and persist in rivers, lakes and other surface drinking-water sources.
Recall that MePiA and MePA are both water-soluble products of atmospheric chemistry operating on the trace gas methylphosphine (MeP). This atmospheric chemistry was operating before the Flood, and it is still operating today, but MeP was an abundant trace gas before the Flood relative to today. As a result, MePiA and MePA were relatively abundant natural products of atmospheric chemistry prior to the Flood, while they are negligible today.
Once formed, both MePiA and MePA will wash out of the atmosphere in raindrops, and both will wind up in natural sources of drinking water. Rainwater runoff collecting in a pond, for example, will add both MePiA and MePA in tiny amounts to the pond. Because MePiA is easily oxidized, it will naturally, slowly convert from MePiA to MePA in the pond. As a result, the concentration of MePiA in the pond is likely to be less than the concentration of MePiA in rainwater. Also as a result, the concentration of MePA in the pond is likely to be greater than the concentration of MePA in rainwater. In fact, because MePA is a stable molecule, it will persist for a long time in the pond, and evaporation of water from the pond could raise the MePA concentration in the pond considerably higher than its concentration in the original rainwater.
The tendency of MePA to build up in freshwater reservoirs has been emphasized in a curious way in modern times. While there are no known natural dietary sources of either MePiA or MePA today, there is a man-made environmental source of MePA today. Recall that MePA is a phosphonate—it is, in fact, the simplest phosphonate—and recall that the phosphonates, as a class, tend to be potent biological compounds. Not surprisingly, humans have found numerous novel uses for synthetic phosphonate compounds. One of the novel uses they have found for some synthetic phosphonate compounds is as warfare nerve gases. Also not surprisingly, when these synthetic phosphonate compounds get into the environment, they tend to break down into smaller, more stable molecular fragments. Finally, not surprisingly, environmental breakdown of complex synthetic warfare phosphonates tends to yield the simplest phosphonate, MePA, as a breakdown product. This has made MePA to be one of several intensely studied products of the natural, environmental breakdown of synthetic warfare agents.[75] The research question of interest, in this context, was whether these breakdown products might have harmful effects when they get loose in the environment. Are they toxic, for example? Do they cause cancer? Do they cause birth defects? MePA was found, in fact, to be in the same general safety category as table salt. But the important point in the present context is that all this weapons-initiated research into MePA has heightened awareness that (1) MePA will persist in the environment, and (2) it will tend to wind up in freshwater sources which may be used for drinking water.
Because MePA tends to concentrate in natural, freshwater sources of drinking water, and because the natural dietary intake of MePiA was found by the biblical life span data model to be nearly sufficient pre-Flood, it seems unlikely that MePA deficiency disease was part of the human aging experience prior to the Flood.
In contrast, once MeP has been lost from the atmosphere, synthesis of both MePiA and MePA in the atmosphere ceases, and both MePiA and MePA deficiency diseases become inevitable. This was not the case pre-Flood, but it is the case at present, making it necessary to differentiate between ancient human aging and modern human aging. Modern human aging appears to be ancient two-phase human aging with Aging 0 added in.
The foregoing conclusion needs to be modified by a caveat to avoid confusion in the future.
It is not likely that the Flood obliterated every last bit of natural source of atmospheric MeP. For example, Figures 9.1 and 9.2 showed that the Bering Sea enjoys high surface concentrations of nitrate and phosphate similar to ocean water surrounding Antarctica. The Flood is not expected to have significantly disturbed sea floor sediments in the northern hemisphere. Thus, it is possible that sea floor sediments in the Bering Sea are a minor source of atmospheric MeP today.
The Flood dramatically reduced the source of atmospheric trace gas MeP, perhaps by a factor of a thousand, or a factor of ten thousand, but it likely did not reduce the source of atmospheric MeP completely to zero. MeP will be present as a naturally-sourced trace gas at some tiny level, however small, in the modern atmosphere.
This implies that MePiA and MePA are yet being produced at some tiny level, however small, in the atmosphere today, and this implies that naturally-produced MePA especially, because it tends to build up in natural water bodies, may be measurable at tiny concentrations in such water bodies. Indeed, detection near 0.5 microgram MePA per liter of river water has been reported.[76] Because MePA is a natural breakdown product of phosphonates, as previously mentioned, and because phosphonates are used widely today in industry, agriculture, and warfare, it is not clear whether this MePA is natural or anthropogenic. Nonetheless, the idea that there is no (i.e., zero) naturally produced MePA in the environment today should obviously be treated as a convenient first approximation only.
One final point needs to be made before concluding this chapter.
The idea that the natural dietary source of MePA was sufficient prior to the Flood while natural dietary MePiA was not quite sufficient implies that MePA is not able to be converted to MePiA by body cells in a general way. If it were, then dietary sufficiency of MePA would have resulted in complete cellular sufficiency of MePiA and no need of dietary MePiA.
It seems likely to be biologically necessary that cells not be able to convert MePA to MePiA in a general way. MePA is itself a vitamin necessary for normal metabolism, as the next chapter shows. If cells converted all MePA to MePiA, a cellular MePA deficiency would result.
Meanwhile, we have previously seen that the very long half-life of MePiA in the body implies that MePA resulting from free-radical oxidation of MePiA in the mitochondria is likely recycled to MePiA.
These considerations imply (1) that conversion of MePA to MePiA happens only in the mitochondria, not throughout the cell in general, and (2) that MePA must not be able to penetrate from the cytoplasm into the mitochondria.
To be as clear as possible going forward, it is necessary to distinguish between the general two-phase theory of human aging and the more specific two-phase theory of modern human aging.
Two-Phase Theory of Human Aging: In general, human aging is a composite of the progression of two diseases:
- Aging 1: congenital vitamin MePiA deficiency disease, and
- Aging 2: zeroed-psephomere disease, induced by Aging 1.
Two-Phase Theory of Modern Human Aging: Modern human aging is a syndrome of three aging diseases:
- Aging 0: congenital vitamin MePA deficiency disease,
- Aging 1: congenital vitamin MePiA deficiency disease, and
- Aging 2: zeroed-psephomere disease, induced by Aging 1.
At present, for individuals not supplementing their diets with the anti-aging vitamins MePiA and MePA, Aging 0 disease, like Aging 1 disease, is active and progressing throughout life, taking its own deadly toll, as subsequent pages will show.

Aging 0 is the nutritional deficiency disease one gets as a result of insufficient dietary intake of vitamin MePA. Because this condition pertains congenitally and universally to modern humans, it is counted among the aging diseases for modern humans, in accord with the general theory of aging for biological organisms.[77]
Recognition of MePA as a vitamin results from decades of research into the cause of human aging. MePA and MePiA have only in recent years emerged from this research as a vitamin duo, the lack of which is responsible for modern human aging.
By definition, a molecule is a "vitamin" depending on what it does, not on whether it is listed as one of the thirteen traditional vitamins. This is necessary because the thirteen traditional vitamins were discovered over a protracted period of time, and there has never been any scientific reason to suppose that the discovery process has ended or that the traditional thirteen vitamins exhaust the vitamin category.
Casimir Funk, whom many regard as the "father of vitamin therapy,"[78] observed that:
The study of degenerative pathological changes of old age… may well belong to a future chapter of vitamin research.[79]Given that Funk would have been intimately familiar with the displayed phenotypes and physiological symptoms of human vitamin deficiency cases as well as animals experimentally deficient in specific vitamins (Figure 20.1), this statement by Funk is not surprising. MePiA and MePA appear to be properly classified as a duo of anti-aging vitamins in fulfillment of Funk's conjecture.
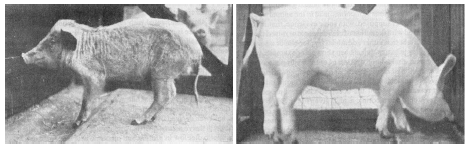 |
MePiA appears to be an exogenous antioxidant vitamin (like vitamin C or vitamin E). MePiA is easily oxidized in vitro to MePA, implying that it will be oxidized by free radicals in vivo to MePA.
Dietary supplementation with microgram per day amounts of MePA has been found to relieve symptoms of many age-related ailments, implying that MePA operates in its own biochemical pathways within the body in a typical vitamin capacity.
MePA, a water-soluble weak acid, behaves in a way which is characteristic of the water-soluble vitamins in general. Compare it to dietary supplementation with niacin (or nicotinic acid), another water-soluble weak acid, for example.
Clinically advanced pellagra is rare in the U.S. today, but before the discovery of nicotinic acid in the latter half of the 1930's, pellagra was common in the southern states where corn was a major staple in human diets. Following is a description from back at that time[81] of the effects on pellagra patients of treatment with nicotinic acid.
A comprehensive report has been made by Spies, Bean, and Ashe, based on observations at the Cincinnati General Hospital, and the Hillman Hospital, Birmingham, Alabama, on the nicotinic acid treatment of hundreds of cases of classic pellagra. It is stated that:"The administration of adequate amounts of nicotinic acid or one of its compounds is followed by the disappearance of many symptoms of the disease. Within 24 to 72 hours [1 to 3 days], the fiery redness and swelling of the tongue, gums, mouth, throat, and vagina subside, and the associated Vincent's infection disappears. Within 24 to 72 hours, nausea and vomiting cease, the increased salivation decreases, and bowel movements become normal. Abdominal distention, pain and discomfort disappear and, in most cases, the desire for food returns. The acute, fiery red erythematous [reddening of the skin, usually in patches] dermal lesions, in which the epithelium [thin tissue forming the outer layer of a body's surface] is intact, blanch within 48 hours after the administration of nicotinic acid, but where the continuity of the skin is broken and the lesions are moist, ulcerated, dry or pigmented, there seems to be no specific benefit. Perhaps the most dramatic response of the pellagrin to nicotinic acid therapy is the disappearance of the acute mental symptoms. These symptoms, varying from slight confusion to delirium and mania, disappear rapidly, often over night. The maniacal patients become calm and the confused patients, mentally clear. After therapy they become readjusted, and often have excellent insight and memory of their actions, ideas and surroundings during the psychotic period. Apathy and lassitude give way to interest."
Relief of diverse symptoms with no hint of any negative side effects due to treatment is characteristic of the cure of a deficiency disease by a water-soluble vitamin. This same behavior is seen with MePA. Following is a summary of experience gained to date with volunteers taking MePA in microgram amounts per day, deliberately imitating the form of the report on the nicotinic acid treatment of pellagra cases quoted above, to show the similarity of the two cases.
A compilation of testimonials has been made by Aardsma at https://agingcauseandcure.com/summary-reports/ on the methylphosphonic acid treatment of dozens of cases of modern human aging. It has been found that:Experience gained to date with MePA demonstrates relief of diverse symptoms with no hint of any negative side effects due to treatment. This is characteristic of the cure of a water-soluble vitamin deficiency disease.The administration of adequate amounts of methylphosphonic acid (MePA) is followed by the disappearance of many symptoms of the disease. Within a few weeks to a few months, the sleep disorders characteristic of aging subside: there is less trouble getting to sleep (i.e., reduced insomnia), sleep is deeper and more refreshing, and less sleep is needed. Associated fatigue is reduced or disappears. The rate of wound healing is remarkably increased, and accompanying inflammation and pain are decreased. The incidence of headaches and migraines is reduced. Within a few weeks to a few months, diseases which have taken hold because of agedness, such as heart failure, cancers, and autoimmune disease, may begin to be slowed, reversed, or cured. Numerous skin disorders disappear: skin becomes more moist and supple; chronic skin infections begin to clear up within a month after the administration of methylphosphonic acid. Perhaps the most dramatic response of the elderly to methylphosphonic acid therapy is the disappearance of chronic mental symptoms. These symptoms, varying from "brain fog" to depression and anxiety, disappear rapidly, sometimes within the first week. The depressed become more happy, the anxious, more calm, and the "brain fogged," mentally clear. Apathy and lassitude give way to interest and creativity.
Additional evidence indicating that MePA is a vitamin is provided by the observation that the physiologically active intake range is very low, and, simultaneously, its toxicity is also very low. This is a distinctive characteristic of many of the vitamins, with 6 out of the 13 traditional vitamins having microgram per day recommended intakes but limited toxicity.[82]
Vitamin B12 is a particularly striking example. Vitamin B12 has the lowest recommended daily intake of any of the vitamins (2.4 micrograms per day for an adult male) but has no apparent oral toxicity in humans.[83] In mice, its acute oral toxicity exceeds 5 g/kg body weight (BW).[84] To put this in perspective, assuming the same toxicity level, the safety margin is 152 million for a person weighing 73 kg and taking 2.4 micrograms of vitamin B12 per day.
The physiologically responsive range for MePA is on par with that of vitamin B12, with intakes as low as 1 microgram per day eliciting marked physiological response. When an LD50 (lethal dose, 50%) of 1888 mg/kg BW[85] is used in conjunction with a recommended daily intake of 20 micrograms per day, the safety margin for intake of MePA is 6.9 million.
These very low recommended intake levels coupled with very large safety margins for vitamin B12 and MePA are not characteristic of most drugs. Most drugs require intakes in the mg per day range in order to elicit the desired physiological response. There are, however, a limited number of drugs that are capable of a physiological response at very low doses. For example, the drug carfentanil can elicit a response in humans starting around 1 microgram.[86] However, a general characteristic of these extremely potent drugs is that the range of safe intake levels around the physiologically responsive range is very small. For example, while human toxicity data is limited, carfentanil is potentially lethal at a dose of 20 micrograms.[87] Thus, the safety margin for intake of carfentanil is only about 20 relative to the minimum amount needed to see a physiological response.
While MePA shares its microgram-range-physiologically-responsive-intake property with both vitamin B12 and the drug carfentanil, it does not have the small-safety-margin property of carfentanil, but rather has the large-safety-margin property of vitamin B12. In this respect, MePA is similar to the vitamin, B12, not the drug, carfentanil, thus providing further evidence that MePA is a vitamin.
We previously listed, in relation to MePiA, four criteria which serve to define a vitamin. Vitamins are (1) organic compounds, (2) which must be obtained through the diet, (3) which are vital to the health of the organism, and (4) which are needed in tiny amounts only. These four criteria, when taken together, are highly restrictive. Of the myriad of molecules known to chemistry, to the present time all have been eliminated from the vitamin category by these four criteria save just over a dozen diverse chemical compounds. That MePA easily fulfills these restrictive criteria is seen as follows:
A literary tablet from Abu Salabikh dated 2600–2500 B.C., contains a list of proverbs including this seemingly enigmatic prohibition:[89]
Do not make a well in a field, the water will do damage to you (?)
This seems to say that drinking well water, not from some specific well, but rather from wells in general, harmed one's health. This implies that it was observed back at that time that people whose supply of water was from wells suffered negative health consequences which were not experienced by people whose supply of water was from other sources such as rivers or cisterns.
This is now explicable. As mentioned previously, well water would be expected to be depleted of the anti-aging vitamins due to sorption of these small organic acids by soils or their utilization by microbes as rainwater passed through the ground into the well. Reliance on well water, depleted of the anti-aging vitamins, would put one on a path equivalent to modern aging, not only accelerating ancient aging, but also invoking the unique symptoms and heightened mortality consequent to Aging 0 disease.
Further support for this understanding of the Abu Salabikh proverb comes from the Proverbs of Solomon found in the Bible. We learn that Solomon, who lived in the first millennium B.C., long after composition of the Abu Salabikh tablet, "pondered, searched out and arranged many proverbs."[90] Not surprisingly then, the Proverbs of Solomon contain some striking similarities to the Abu Salabikh proverbs list. Both contain prohibitions against sex with a prostitute and against guaranteeing another person's loan, for example. But the prohibition against well water is missing from Solomon's proverbs. In fact, one finds the opposite:[91]
Drink water from your own cistern,While Solomon is clearly speaking metaphorically in this proverb, the parallelism used in the metaphor shows that Solomon held to no general prohibition against drinking well water. Why should there be this stark difference between the Abu Salabikh proverbs and Solomon's proverbs?
And fresh water from your own well.
When the Abu Salabikh proverb was written, the observation that well water produced negative health symptoms not experienced by people drinking river or cistern water would have been both generations old and current. Environmental MePiA and MePA fell essentially to zero only at the Moses Drop, which happened roughly 2504 B.C., at the end of the 2600–2500 B.C. date range given for the Abu Salabikh tablet. In contrast, when Solomon's proverb was written, early in the first millennium B.C., the anti-aging vitamins would have been missing from the environment for more than one and a half thousand years. Thus, by Solomon's time, as it is today, there was no difference between well water and cistern water—both were equally deficient in the anti-aging vitamins—and hence there would have been no reason for Solomon to warn against drinking well water. The prohibition against drinking well water should be found only in the more ancient Abu Salabikh proverbs list—which, indeed, is the case.
The cause of Aging 0 is dietary insufficiency of MePA (methylphosphonic acid), a previously unknown vitamin. Today, dietary insufficiency of MePA is a congenital condition of the global human population, making Aging 0 a universal disease which actively progresses from birth on. In the ancient past, when MePA was still naturally present in surface water, dietary insufficiency of MePA with consequent symptoms of Aging 0 seems possibly to have been the plight only of individuals dependent on wells to meet their daily need of drinking water.

Because Aging 0 is vitamin MePA deficiency disease, supplementation of the diet with an adequate daily intake of MePA constitutes the cure.
People supplementing their diets with MePA report many and varied health improvements.[92] For this reason, vitamin MePA, while certainly not a panacea, may be regarded as the "general health" anti-aging vitamin. Looked at properly, this says merely that vitamin MePA is involved in multiple biochemical processes in the body—as is true of most of the vitamins—so that, in its absence, multiple things go wrong in the body, and multiple ailments result. The resulting ailments of MePA deficiency disease are not trivial. They contribute their own load of morbidity and mortality to "old age" in modern humans, as the next chapter will show.
Experience with this disease to date indicates that, in contrast to Aging 2, healing of Aging 0 is relatively prompt—days, weeks, or months for most symptoms.
The best way to determine how much of an anti-aging vitamin is needed by the human body is probably by titration experiments with humans to determine how much the body retains. Unfortunately, determination of the optimal daily intake for vitamins in general is no trivial task. Scientists today are still working to fine tune the daily recommended intake of vitamins discovered several generations ago. The empirical determination of the optimal daily intake for the anti-aging vitamins is also not a trivial undertaking. It remains a key priority for Aardsma Research & Publishing (ARP).
General health is something that individuals can sense about themselves. When there is improvement in physiological function in areas such as sleep quality, we are often able to perceive the change. Anecdotal data over the last several years from individuals supplementing with vitamin MePA at various levels have indicated that an adequate daily intake is likely between 1 and 10 micrograms per day for an adult male. Rapid and dramatic health improvements reported at 1 microgram per day[93] make it clear that 1 microgram per day is within the beneficial range, so it is expected that the adequate daily intake is not far away. Also informative has been the September 16, 2018, testimonial from one user who experimented with reducing his daily vitamin MePA intake from 6 to 4 micrograms per day.[94] The results clearly indicated that, for this mature male, 6 micrograms of vitamin MePA per day was better than 4. While other individuals have supplemented with vitamin MePA at levels above 6 micrograms per day, there have been no additional reports of benefits at higher intakes. Therefore, it appears unlikely that the daily need for vitamin MePA is above 10 micrograms per day. This places the adequate daily intake of vitamin MePA between 1 and 10 micrograms per day for an adult male. This is still a large range and needs further refinement.
The environment probably provided more MePA than needed by the human body pre-Flood in some instances. As previously mentioned, this is because (1) rainfall would have supplied water sources with both MePiA and MePA, (2) from experiments conducted in the ARP lab (unpublished data) we know that MePiA in water slowly converts to MePA, and (3) because MePA is environmentally persistent, continual evaporation of water from drinking water sources would leave behind increasingly concentrated MePA. This suggests that at least some pre-Flood humans were routinely consuming more MePA than they needed, with the excess being eliminated from the body, presumably in urine. This leads to the conclusion that the current recommended daily intake of MePA should tend towards the upper range of the estimated physiological need (10 micrograms per day) rather than the lower end (1 microgram per day).
Most vitamins have a large safety range of intakes around the physiological requirement. This is likely why vitamin supplements are readily available without a prescription or extensive regulation, unlike drugs, which typically have a much smaller safety range around the recommended intake. This means that, for vitamins, the risk of inadequate supplementation is typically greater than the risk of over-supplementation. As all the available evidence indicates that this is also true for vitamin MePA, the rational approach is to supplement at the higher end of the estimated intake range.
Since the concentration of MePA was probably equal to or greater than the concentration of MePiA in pre-Flood drinking water, a reasonable approach to mimic what was happening pre-Flood is to supplement with at least as much MePA as the amount of MePiA needed by the body, as long as the best estimate for the adequate intake of MePiA intake exceeds 10 micrograms per day. Taking this approach reduces the risk of inadequate intake of MePA, and any excess MePA will be eliminated in the urine, as it was pre-Flood.
We presently recommend 20 micrograms MePA per day for adult males, equal to our present recomendation for MePiA.[95] Dr. Aardsma's Anti-Aging Vitamins dietary supplement[96] presently provides MePA and MePiA in equal amounts, making it easy to meet these recommended daily intakes. Refinement of these recommendations seems likely in the future. Bottles of Dr. Aardsma's Anti-Aging Vitamins include the most current intake recommendation on the label.
As with Aging 1 and Aging 2, the cure for Aging 0 disease is in hand and is commercially available.

While human aging prior to the Flood appears to have involved only Aging 1 and Aging 2, modern human aging involves a third aging disease, Aging 0. This was the first aging disease to be discovered. It is a nutritional deficiency disease resulting from cellular insufficiency of the vitamin methylphosphonic acid (MePA), and it may be cured by dietary supplementation with vitamin MePA.
Because (1) oxidation of MePiA produces MePA, (2) MePA is likely recycled back to MePiA within the mitochondria, (3) both MePiA and MePA are products of atmospheric chemistry operating on the single trace gas methylphosphine, and (4) both MePiA and MePA, being small water-soluble organic acids, tend to travel together in the environment and end up together in drinking water sources such as ponds, lakes, and rivers, it is natural to regard MePiA and MePA as a vitamin duo.
The addition of Aging 0 to Aging 1 and Aging 2 completes the set of diseases which are responsible for the clinical expression of the syndrome we moderns call "aging."

Human aging may be likened to a three-headed dragon.
The first head is named "Aging 0," the second head "Aging 1," and the third "Aging 2." These three heads are not triplets. They are each unique.
When we were born, our personal aging dragon, residing deep within our physical body, was embryonic and undeveloped. It would have remained indefinitely embryonic and unable to do us any harm if the two closely related, recently discovered vitamins, methylphosphonic acid (MePA) and methylphosphinic acid (MePiA), had been present in adequate amounts in our diets. But because these two vitamins are no longer naturally available in any food or drink, our personal aging dragon began to develop. Early on, it sprouted two heads: Aging 0 and Aging 1. Aging 1 fed insidiously on our living flesh until, after some years, the dragon had grown sufficiently to sprout its third head, Aging 2. Aging 2 soon outstripped the other two heads, greedily gobbling away at us. If the dragon continues to have its way, once it has grown large enough, it will devour us completely, killing us in the process.
The existence of these dragons has been known from very ancient times by the havoc they wreak on our bodies. How to vanquish them has been the object of dreams and schemes, also from very ancient times. A proper scientific understanding of ancient life spans recorded in the Bible now enables us to be the first to know enough about these dragons to wield effective weapons against them. That they may be kept dormant by vitamins MePA and MePiA has only recently been discovered. The discovery that they are three-headed dragons is even more recent.[97]
The discovery that these dragons are kept perpetually dormant by adequate daily intake of vitamins MePA and MePiA constitutes the fundamental cure for human aging. This discovery means that the human aging syndrome may be eliminated from Earth in a single generation by the simple expedient of universal lifelong dietary supplementation of vitamins MePA and MePiA in adequate daily amounts.
This expedient should, of course, be seized upon at the highest levels of government, and it should, of course, be promptly implemented. Aging is, after all, humanity's number one pandemic, flourishing virtually unimpeded from very ancient times. Compared to it, COVID-19 was a Sunday school picnic. American deaths involving COVID-19 from early March 2020 through early March 2021 averaged less than 1,500 per day. Meanwhile, aging has been claiming well over 6,000 American lives per day for a very long time, year after year.
But do not hold your breath waiting on appropriate government action. Because of its unique utilization of ancient biblical life span data, this book is decades ahead of mainstream science in its understanding of aging. Anti-Bible prejudice coupled with normal bureaucratic inertia poses a high hurdle to appropriate government action in this particular instance.
At present, we must look after ourselves in regard to our personal aging dragon, and then we must do what we can to help others around us look after themselves in regard to their dragons. To make self-help possible, Aardsma Research & Publishing (ARP) has made a dietary supplement, Dr. Aardsma's Anti-Aging Vitamins, commercially available.[98] We are doing what we can do now, from our end, to begin to fight these dragons, while we wait on government and the rest of the world to come up to speed.
Meanwhile, research into human aging, informed by both ancient biblical and modern U.S. life span data, carries on apace at ARP. The present chapter communicates a number of important, recent discoveries resulting from a modeling study utilizing 2016 actuarial life table data for the U.S. population. Most importantly and urgently, it exposes a current national health emergency. It reveals that some 74,000 U.S. males and 212,000 U.S. females are currently dying needlessly each year of Aging 0 alone. It finds that these deaths could easily, immediately be prevented by a nationwide program of universal lifelong supplementation with Dr. Aardsma's Anti-Aging Vitamins.
According to the two-phase theory of modern human aging, what is currently called "aging" is a syndrome of three individual aging diseases:
According to this theory, human aging unfolds in two phases: first a pure vitamin deficiency disease phase (Aging 0 and Aging 1) and then a second phase during which all three aging diseases are operating simultaneously.
Development of this theory has relied heavily on ancient human life span data. The task for this chapter is to determine whether the two-phase theory of modern human aging is able to explain modern human life span data. The question is not whether the two-phase theory can explain the dramatic shortness of modern human life spans relative to ancient biblical life spans. It does that at the outset. Remove a vitamin, such as vitamin C, from a person's diet, and you can be sure that person will die young. The more vitamins you remove, the younger that person will die. We modern humans are dying tragically young relative to our ancestors, who enjoyed youthful life spans of nearly 1,000 years, because vitamins MePA and MePiA are missing from modern diets. The present question is whether the two-phase theory of modern human aging can account for the detailed pattern of modern shortened life spans. The dramatic shortness of modern human life spans is already amply explained.
Modern human life span data reveal a distinctive pattern.
For each calendar year, or cohort, death rates are relatively high in the first year after birth, decline very rapidly to a low point around age 10, and thereafter rise, in a roughly exponential fashion, before decelerating (or slowing their rate of increase) at the end of the life span.[99]
Is the two-phase theory of modern human aging able to explain this pattern, and not just in a qualitative, hand-waving, approximate sort of way, but in detail and in a precise quantitative fashion? If it cannot do so, then something is yet wrong with the theory, and it needs further development. If, on the other hand, it can do so, then development of the theory may reasonably be regarded as complete, and the theory itself may reasonably be regarded as functional.
The next two sections are necessarily heavy with math and technical details. Readers interested only in the results may wish to skip ahead to Section 23.4.
The data of interest are shown graphically in Figure 23.1. This dataset is for the year 2016 from the United States Social Security Administration's actuarial life tables.[100] It is an extraordinary dataset, incorporating death data from 2,744,248 individuals.[101] This large number of individuals results in very small measurement uncertainties in most of the individual data points, allowing small effects to be clearly resolved.
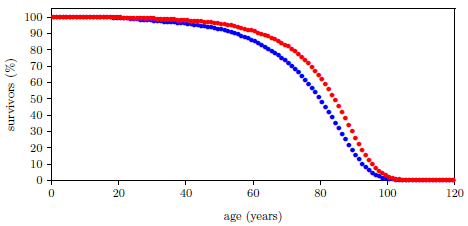 |
The Aardsma model for generic aging disease provides the needed foundation for explaining this dataset. This model is given by the equation:[102]
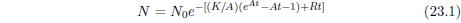
In the present context, this equation includes two contributions to death of individuals within a total population of N0 individuals all of the same age. The first contribution is due to an aging disease, controlled by K and A, and the second contribution is due to a constant background rate of random extraneous (i.e., not due to aging) deaths controlled by R.
Figure 23.2 shows that the Aardsma model is able to explain the gross features of the pattern of the dataset reasonably well. That is, the fitted curves follow the data points reasonably well. This means that the modern human survival curve is dominated, in panoramic view, by an incidence of death due to aging which increases exponentially with age.
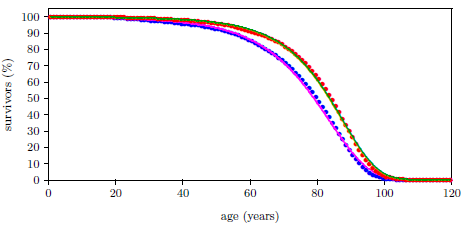 |
While the coarse pattern of the dataset is explained by the Aardsma model, the fine details of the dataset are not explained at all. This is most forcefully revealed by the goodness-of-fit parameter, Χν2, for the two curves of Figure 23.2. For a good fit, this parameter is expected to be less than 1.5. For the fit to the male data it is 17,462, and for the female data it is 8,494.
The fits are poor at the fine-details level because actuarial life table data include deaths from many other causes than just aging. The Aardsma model includes an age-independent, random deaths term, but it neglects all age-dependent extraneous causes of death. This is suitable for much laboratory animal data, but it is not suitable for real-life human life span data. While laboratory animals do not kill one another in wars, for example, real-life humans do.
To explain these modern life span data quantitatively, using the Aardsma model, it is necessary either (1) to upgrade the Aardsma model by adding more terms needed to describe additional causes of death or (2) to eliminate these extraneous deaths from the dataset in some way.
In the following analysis, the second method will be used. Because the analysis is no longer seeking to explain just the broad outline of the dataset—because it is now digging into the details—this is an unavoidably complex undertaking. To keep things as simple as possible, we will proceed in small steps, chipping away at the analysis a piece at a time, to arrive at a final solution through a series of successive approximations.
The Aardsma model, as it is presented in Equation 2, describes aging in a generic way. The two-phase theory of modern human aging is not generic. It describes three specific aging diseases—Aging 0, Aging 1, and Aging 2—each of which contributes to aging in its own specific way. All three of these diseases need ultimately to be represented in the model, each by its own individual term patterned on the Aardsma model. As a first step in this direction, include in the model at this stage only the dominant disease of these three.
The pre-Flood biblical dataset has revealed that Aging 2, induced by Aging 1, was by far the primary cause of aging deaths pre-Flood.[103] However, as previously discussed, Aging 0 (MePA deficiency disease) would not have been a part of human aging back at that time the way it is today, making it hypothetically possible for Aging 0 to be the dominant cause of aging deaths today. Life-span data from laboratory mice argue against this possibility. As shown earlier in this book, in an experiment designed to look for a large increase in life span, mice treated with MePiA (i.e., cured of Aging 1 and thus protected from Aging 2) did show a clear increase in longevity, while mice treated with MePA (i.e., cured of Aging 0) did not. Thus, Aging 2 appears to be the primary cause of aging deaths at present, just as it was in the past. As a first step in modeling the modern actuarial life table data, generic aging should be replaced by Aging 2 disease.
The generic aging model of Equation 2 is not far from what is needed to describe Aging 2 deaths. Deaths due to Aging 2 are expected to increase exponentially with age, as Equation 2 describes. The only real difference is that Aging 2 is expected to begin subsequent to birth (i.e., not at t = 0). Aging 2 is induced by Aging 1. Aging 1 must operate for some length of time before Aging 2 is induced. Our best current estimate of when this happens is 7.5 ± 9.3, as previously discussed. It is still correct to have random deaths beginning at birth, as they do in Equation 2, but the onset of Aging 2 deaths needs to be able to be delayed for some length of time. To make this happen, Equation 2 needs to be modified (generalized) by introduction of a fourth parameter, t2, specifying the time at which Aging 2 begins. This necessary modification yields the first-approximation model shown in Equation 3.
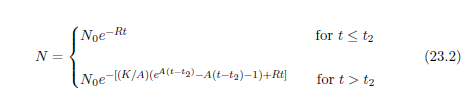
The next objective is to fit this first-approximation model to the 2016 actuarial dataset to see how well it does at this level of approximation. To keep things as simple as possible, focus on just the male dataset for now.
It is clear that this simple model will not do well if fit to the entire male dataset because of the presence of extraneous (i.e., not due to aging) deaths, as mentioned previously. To make progress toward the present objective, a method of eliminating extraneous deaths from the dataset must be devised.
The Aardsma model already accounts for age-independent extraneous deaths. This is what the R is all about in Equations 2 and 3. These are deaths that can happen to anybody at any age with equal probability. Being struck by a meteorite is a good, if rare, example of an age-independent extraneous death.
Missing from the Aardsma model are age-dependent extraneous deaths. Neonatal deaths are a good and, sadly, not so rare example in this category. The 2016 U.S. actuarial life table for males reveals, for a starting population of one million males, 6,364 deaths in the first year of life and only 429 deaths in the second year of life. Clearly, the first year contains a very large, age-dependent, neonatal contribution which is not due to Aging 2.
The most obvious way of eliminating neonatal deaths from the dataset is by the simple expedient of excluding the data point for the first year of life from the least squares fit of the first-approximation model to the dataset.
Further pondering of the problem of age-dependent extraneous deaths soon clarifies that this method of removing neonatal deaths can be extended to other age-dependent extraneous deaths. Data points which are clearly too "contaminated" by age-dependent extraneous deaths can simply be excluded from the fit. This will allow aging deaths to have primary influence over the fit. Of course, this approximation will fail if too many data points are so contaminated that they have to be excluded, but this outcome seems unlikely. The dataset has over 110 data points. A model including all three aging diseases would be expected to have fewer than a dozen free parameters. Well over half of the data points could be excluded, and still there would be a large number of degrees of freedom remaining for the least squares fit.
Up to the present time, the Aardsma model has been used to model survival curves constructed from total-survivors-versus-time data. It is much easier and more intuitive to use this data-exclusion method with probability-of-death-per-unit-time data points than it is with total-survivors-versus-time data points, so the next task is to transform the problem into its probability form.
The actuarial life table data are conveniently given in both forms. Figure 23.3 shows the 2016 actuarial life table dataset for U.S. males in probability-of-death-per-year form.
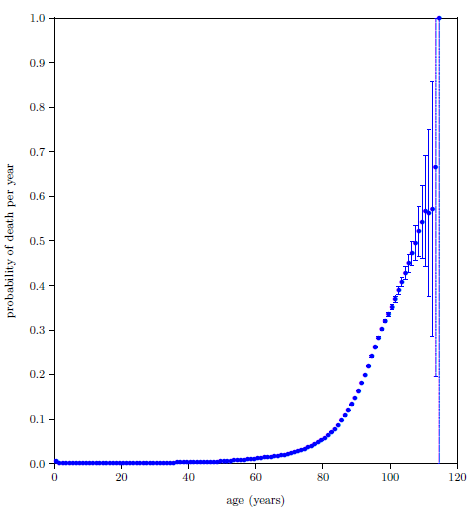 |
The probability of death per year, P, is seen to be quite small for the first four or five decades of life, but by 80 years the probability of death per year is well above zero and bending sharply upward.
The error bars are calculated as the square root of the actual number of deaths per year divided by the number of survivors at the beginning of the year (measured from the annual birthday of the individual), starting from the actual total number of U.S. males who died in 2016. The published actuarial table specifies deaths per year from a starting population of 100,000 individuals, not the actual number of U.S. males who died in 2016. The actual number of deaths in the United States for 2016 reported by the CDC was 2,744,248.[104] Taking half of these to be male yields an actual starting population of 1,372,124. Thus, to get the error bars right, the actual number of deaths per year was calculated from the published table using this starting population size rather than 100,000.
The calculated error bars are too small to be seen in Figure 23.3 for most of the data points, but they become quite large as age increases beyond 100 years. These large error bars result from the relatively small numbers of survivors beyond 100 years today.
Now the Aardsma model needs to be transformed from its survival form to its probability form.
The defining differential equation for the Aardsma model has been previously specified as:[105]
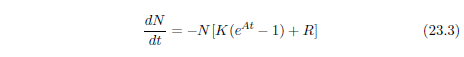
This can be transformed to probability of death per unit time by dividing both sides of the equation by -N0.
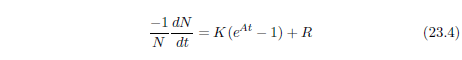
The left side of the equation is now the probability of death per unit time. Call this simply P from now on. In practice, P is just the number of deaths in a given time interval divided by the number of survivors at the beginning of the time interval and divided by the length of the time interval. For the 2016 actuarial life table for males, P for the first year of life is just (6364 / 1,000,000 / 1 =) 0.006364, and for the second year of life it is (429 / (1,000,000 - 6364) / 1 =) 0.000432.
It is helpful at this stage, for clarity and mnemonic value, to make a slight change of notation. The present context is concerned with the application of this model specifically to Aging 2. This has already instigated generalization of this equation by the introduction of t2 as a fourth parameter. Following the lead of this subscripted notation, replace A in Equation 5 with E2 and replace K with A2. The result of these changes is:
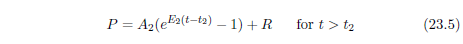
Just as R controls random extraneous deaths, so A2 controls Aging 2 deaths. Meanwhile, E2 is an exponential growth constant, controlling how rapidly the probability of death due to Aging 2 increases with age.
The transformed generalized Aardsma model may now be specified from birth onward as:
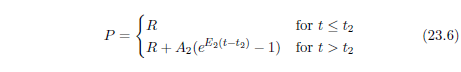
The next task is to select the data points to be included in the least squares fit. The goal is to minimize the impact of age-dependent extraneous deaths on the fit.
Each data point gives a probability of death for a given age. The probability of death for a given age is the sum of the probabilities of death for numerous causes of death at that age. Potential causes of death include both Aging 2 and extraneous deaths such as murder and automobile accident. All of the data points are contaminated with extraneous deaths to some extent, but some points are much more contaminated than others. The contribution of extraneous deaths to the probability of death for a given age becomes less and less noticeable as age increases. In the first four or five decades of life, Aging 2 is not very developed, so most deaths are due to extraneous causes. But this reverses in the latter decades of life. Development of Aging 2 is then advanced, causing most deaths to be due to Aging 2 disease, and causing the contribution of extraneous deaths to the probability of death to become relatively minor.
Figure 23.4 allows the typical magnitude of extraneous deaths to be estimated. It shows the 2016 actuarial life table dataset for U.S. males with the probability axis expanded by a factor of one hundred. The probability of death has gone off scale by age 60. Aging 2 is significantly developed by age 60, so data points up near age 60 aren't very helpful for the present purpose because they mix together both significant Aging 2 deaths and significant extraneous deaths. But below about age 40, Aging 2 is not very developed, so these ages provide a good estimate of the typical size of the probability of death due to extraneous causes. This suggests that extraneous deaths can be expected to account for a probability of death per year of say less than about 0.0025.
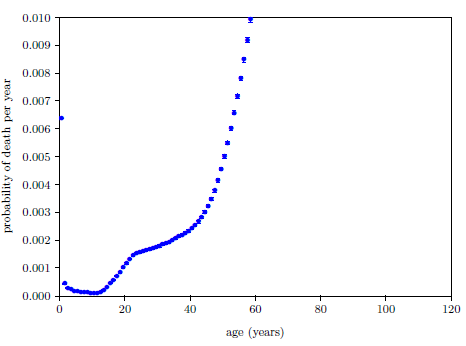 |
This means that data points having a total probability of death per year greater than 0.025 can be expected to be contaminated at less than a (0.0025/0.025×100 =) 10% level, data points having a probability of death per year greater than 0.05 can be expected to be contaminated at less than a 5% level, and data points having a probability of death per year greater than 0.25 can be expected to be contaminated at less than a 1% level.
For the 2016 dataset for males, the 10% (0.025) level corresponds to age 71, the 5% (0.05) level corresponds to age 79, and the 1% (0.25) level corresponds to age 95. Adopt the 5% level as the cutoff at this stage. That is, keep all data points from age 79 on.
Next, the fit needs some data points in the low age range. Figure 23.5 provides visual assistance with this.
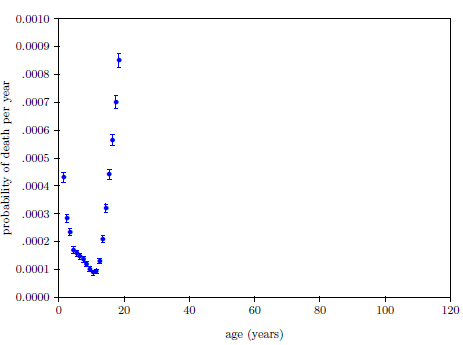 |
It shows the 2016 actuarial life table dataset for U.S. males with the probability axis expanded by a factor of one thousand. Extraneous deaths fall off fairly rapidly from both sides to a minimum between 10 and 11 years of age. This minimum—below one death per 10,000 individuals per year—seems likely to be mainly due to random extraneous deaths, which are included in the model by the R parameter. So points near this minimum should not introduce any large error into the fit.
Keep the three points closest to this minimum. That is, keep the data points for ages 9, 10, and 11.
These choices retain 39 data points at this stage: the 36 data points from age 79.5 (i.e., for the time interval from 79 to 80 years of age) to the end of the dataset, plus the three data points displaying the lowest P values of the entire dataset, from ages 9.5, 10.5, and 11.5 inclusive.
If the Equation 7 model is fit to the selected data points at this stage, the least squares fitting program finds for the age at which Aging 2 begins, t2 = 9.5 ± 64.7 years. The very large relative error found for this parameter results from the fact that few data points have been able to be retained in the fit down at the young ages where the true value of t2 is expected to be found. The least squares fit mathematics shows that there aren't enough input data points where they are needed to be able to nail this parameter down. This seems likely to add a lot of variability to subsequent fits as the model is developed below, obscuring the effects of further improvements, so it seems best to deal with this at this stage.
The problem can be worked around by estimating t2 independently and then using the estimate as a fixed parameter rather than as a free parameter of the model. Previously, by scaling the pre-Flood value of t2 (800 years) to modern times, the modern value of t2 was estimated to be around 14 years. Subsequently, the digital organism model found a value of roughly 0.9 years. These two estimates were averaged to obtain 7.5 ± 9.3 years as the current best estimate. Use this estimate as a fixed parameter for the present actuarial modeling study. That is, set t2 = 7.5 years in the model from now on.
Figure 23.6 shows the fit of Equation 7, with t2 = 7.5 years, to the selected data points.
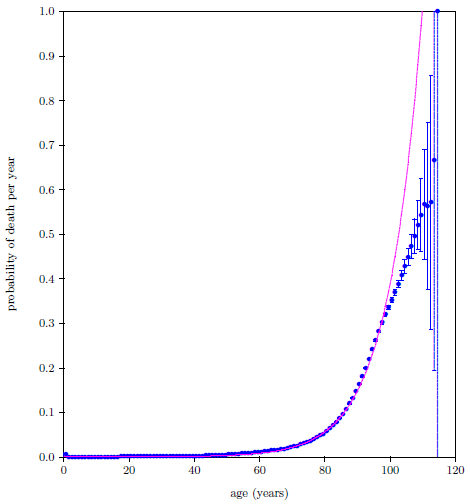 |
This gave Χν2 = 54.8 with 36 degrees of freedom—still a long way from 1.5, signaling a surprisingly poor fit.
The reason for the poor fit is visually apparent from the graph. The data deviate from the expected exponential growth, shown by the magenta line, beginning somewhere after age 90.
The most obvious question to ask in such a case is whether the data can be trusted. This unexpected slowing of the rate of increase of P happens in the nineties, and not a lot of people make it to their nineties. Is it possible that the slowing is just a statistical anomaly with this particular dataset?
No, it is not possible. We are assured by the quotation from the Social Secuity Administration given previously that, for the general case, the "death rates… rise, in a roughly exponential fashion, before decelerating (or slowing their rate of increase) at the end of the life span."[106] This plainly means that the slowing down is always seen in all datasets. The problem is clearly with the model, not with the data.
These data are insisting that the death rate due to Aging 2 does not continue to progress exponentially indefinitely, as the Aardsma model assumes. Even though Aging 2 still continues to progress past age 90, its rate of progression slows down.
This is a significant discovery. It means that the development of Aging 2 disease is subject to one or more limiting factors—that progression of Aging 2 disease is a saturating exponential process rather than a simple exponential process. (A saturating exponential process first grows exponentially, but then growth slows down and, if given enough time, ceases entirely). We must now pause development of the present actuarial table model to deal with this unexpected discovery.
Why should the progression of Aging 2 deaths be a saturating exponential process rather than a simple exponential process? We went back to the digital organism model to try to find an answer to this question.
The digital organism model, as originally conceived, had no way of calculating the death rate due to Aging 2. It merely tracked the length of psephomeres versus the age of the organism, and psephomere length is not the same thing as death rate. To study why the Aging 2 death rate should be a saturating exponential, it was necessary to add to the digital organism model a means of converting from psephomere length to Aging 2 death rate. This was done as follows.
First, by way of a general improvement of the model, the production rate of free radicals was changed from constant to variable. The idea here is simple. Free radicals are produced in the mitochondria as a result of the mitochondria doing their job of producing energy for the cell. As the free radicals damage the mitochondrial DNA, they hinder the ability of the mitochondria to do their job. This is expected to result in (1) loss of energy to the cell and (2) concomitant reduction in free-radical production in the mitochondria. To implement this idea, the free-radical production rate was made inversely proportional to the average mtDNA generation number for the cell. The higher the generation number of the mtDNA, the more copy-error mutations it is likely to contain and, hence, the more dysfunctional the mitochondrion is likely to be, resulting in less energy production and lower free-radical production.
Second, explicit calculation of the probability of Aging 2 deaths per year was added to the model. The idea here seems to be most easily illustrated by considering cancer deaths resulting from Aging 2. Imagine that an mtDNA copy error produces an mtDNA mutation which produces cancer. The copy error may happen fairly early on in life, but the cancer will take many years to develop and become fatal. Thus, each copy error contributes to the probability of aging deaths per year. The individual contributions were modeled as tiny, bell-shaped (Gaussian) probability distributions. A tiny Gaussian originates each time a new mtDNA copy is made. The shape of all these Gaussians is the same (i.e., σ is fixed), and their centers (i.e., μ ) are all shifted a fixed number of years into the future from the age at which the mtDNA mutation happens. The amplitudes, however, are variable, made proportional to the generation number of each new mtDNA copy. This codifies the idea that the further removed from the original an mtDNA copy is, the more copy errors it is likely to have, and hence the more likely it is to do harm to the organism.
This allows a probability of death per year of the organism at a particular age to be calculated as the sum of the individual probabilities at that age from all of the tiny Gaussians which have accumulated up to that age. The calculated probability is not properly normalized, but this is of no concern in the present context. Only the shape of the final probability of death per year is currently of interest. Is it initially exponential, and does it saturate or not? Figure 23.7 shows that, yes it is initially exponential, and yes it saturates.
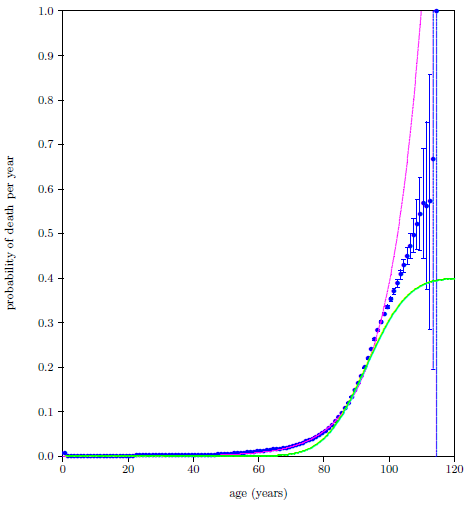 |
Remarkably, once again, the highly simplified digital organism model has successfully emulated an observed, real-life effect. It seems that the principles underlying the model cannot be too far off the mark.
The digital organism model implies that Aging 2 saturation happens because replacement of dysfunctional cells brings the organism eventually to a steady-state total probability of death per year. Healthy replacement cells continue to infuse new life, diluting the dominantly morbid effect of the much larger number of diseased cells.
Dissecting what is going on in the digital organism model a little more deeply yields the following more detailed explanation. When the organism is young, the average generation number of its mtDNA copies is relatively low. Summing the corresponding low-amplitude tiny Gaussians results in a low probability of death per year. (Recall that the amplitude of these Gaussians is proportional to the generation number of the mtDNA copy.) As the organism advances in age, the average generation number of its mtDNA copies initially increases, causing the corresponding total probability of death per year to begin to increase. However, average mtDNA generation number does not increase indefinitely, because cells having mtDNA of high generation number have a high probability of being replaced, and they get replaced by cells with mtDNA having a low generation number. Eventually, with increasing age, a steady state is achieved in which the number of high-generation-number mtDNA cells being replaced per year is constant. This causes the average amplitude of the tiny Gaussians to become constant. Once this happens, each subsequent year then produces the same average number of mutations having the same average probability of harm down the road. Summing these similar tiny Gaussians year by year then results in the same total probability of death year by year—which is just another way of saying that the probability of death per year then saturates.
Unfortunately, in real life, the steady state probability of death at which Aging 2 saturates is so high that nearly everybody dies before getting anywhere near it. The onset of saturation begins to be seen only subsequent to 90 years of age. The probability of living into the fully saturated regime is vanishingly small, so the practical benefit of Aging 2 saturation on longevity turns out to be minimal.
The actuarial table model is now in need of an alteration. The description of death due to Aging 2 needs to be improved to allow for saturation.
Briefly, let ts be the time at which exponential progression of Aging 2 stops and saturation begins. Use the functional form 1-e-x to describe saturation. Too little is known about Aging 2 to be able to derive the functional form which should be used here at present, so this is merely a convenient first approximation. This choice preserves the exponential growth part of the description of Aging 2 which the model already has and merely adds saturation to it. Demand that the resulting curve and the slope of the curve be continuous at ts. This yields:
The model is now:
The fit of the Equation 9 model to the selected data points is shown in Figure 23.8. This is now a five-parameter fit (R, A2, E2, Es, and ts) with t2 fixed at 7.5 years. The fit is remarkably improved. The value of the goodness-of-fit parameter, Χν2, reflects this. It has now dropped below 1.5 for the first time, to just 0.46 with 34 degrees of freedom.
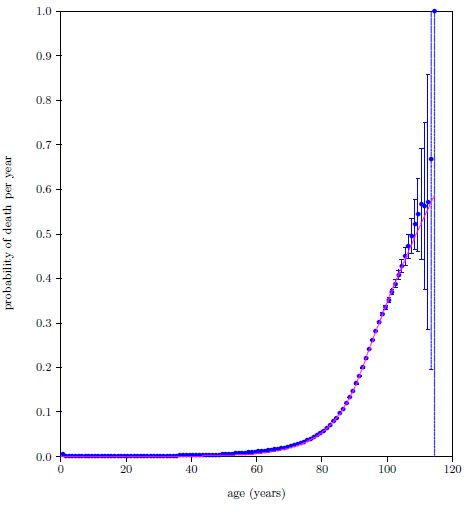 |
But, even though the fit is now statistically good for the first time, the data still indicate that something is not quite right with the model. The final ten data points are all visibly above the magenta line. The female dataset (not shown) behaves in a similar way at this stage. This suggests that there is another cause of death for which the model is, so far, not accounting. This cause must be an aging disease, because the extraneous death contribution to these high age data points has dropped well below the 1% level. Recall that the 1% cutoff is back at age 95.
It is good news for the two-phase theory that the data imply this. It means that the data independently say the same thing the two-phase theory says, that aging, as it is experienced today, is made up of more than just a single aging disease. This, of course, is independent corroboration of the theory of two-phase human aging.
The two-phase theory has two more aging diseases ready to go: Aging 0 and Aging 1. Which of these two is the next most dominant disease?
Real-life observation argues in favor of Aging 0. For example, I (GEA) had the fortunate misfortune, several years ago, of having vitamin MePA be the first anti-aging vitamin discovery and mistakenly thinking it was the only anti-aging vitamin. This resulted in a significant number of individuals, myself included, taking vitamin MePA by itself for some months or years. The results of this unintended, single-vitamin experiment was that many health improvements resulted for many individuals, revealing the importance of this vitamin to health.[107] In my own case, the results were somewhat dramatic, as recounted in the epilogue of this book. I have since, on several occasions, commented in private conversation that I felt that I would likely be dead were it not for vitamin MePA. My health before vitamin MePA was trending steadily downward. I was having to exercise more and more personal discipline with food and work habits to stay healthy enough to be productive, yet I felt that I was fighting a slowly losing battle. This all reversed with the advent of MePA.
Vitamin MePA is the cure for Aging 0. Said simply, experience with MePA supplementation implies that Aging 0 can ultimately be debilitating to the point of death. And this means that Aging 0 should be a real—not just a hypothetical—part of modern actuarial life table data. It appears that, while Aging 2 is the most dominant cause of death, Aging 0 should be next in line and not too far behind.
The Aging 0 term is a relatively easy adaptation of the Aardsma model. It is simply an exponential increase in the probability of death per year, with the probability of death initially (at birth) being zero. Using subscript 0 to designate Aging 0 parameters, the needed term looks like this:
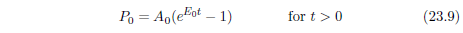
Figure 23.9 shows the fit of the upgraded model:
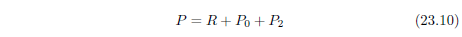
This is now a seven-parameter fit (R, A0, E0, A2, E2, Es, and ts) with t2 still fixed at 7.5 years. Though the visual fit is significantly improved in the final decade, Χν2 fails to show it, remaining the same (0.46, now with 32 degrees of freedom) because of the large error bars diminishing the weights of the data points in this region.
It might seem that development of the model could stop at this point. The data's demand for a second disease has been satisfied, with a reasonably good fit both statistically and visually being the result. But there is yet a problem. The model's placement of the second disease, Aging 0, does not agree with real-life, personal experience of this disease.
Figure 23.10 shows the problem graphically. It shows, as a black line, the contribution to the total probability of death the model has assigned to Aging 0. That is, the magenta line shows the total probability of death, made up of random extraneous deaths plus Aging 0 deaths plus Aging 2 deaths, while the black line shows just the putative Aging 0 contribution to the total probability of death.
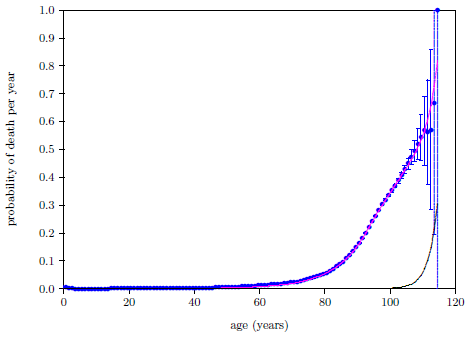 |
The black line implies that Aging 0 makes negligible contribution to deaths until very late in life, out near 100 years of age. My (GEA) personal encounter with Aging 0 started when I turned 50, with the onset of chronic inflammatory demyelinating polyneuropathy (CIDP), and intensified throughout the succeeding decade until it seemed unlikely that I would last yet another decade. This same conflict with reality exists for nearly all of the individuals who supplemented with vitamin MePA alone and experienced resulting health benefits, nearly all of whom were decades younger than 100 years.
It appears that, while a two-disease model is able to fit the dataset mathematically, the resulting solution does not correspond to the real world.
The two-phase theory specifies three aging diseases, not two. Is there possibly a three-disease model which both fits the data mathematically and more accurately reflects real-life experience?
Aging 1 is MePiA deficiency disease. MePiA is an antioxidant. The working hypothesis for vitamin MePiA at present is that MePiA fulfills a unique antioxidant function within the mitochondria, protecting mitochondrial DNA (mtDNA) from excessive free-radical damage. Loss of this vitamin from the diet results in loss of protection of mtDNA, with consequent, eventual induction of Aging 2, which is fundamentally a mitochondrial disease.
It seems unlikely that the role of Aging 1 in human aging stops with just induction of Aging 2. Free radical production in living tissue never ceases and, in the absence of MePiA, free-radical damage to mtDNA carries on with or without induction of Aging 2. This damage seems certain to have deleterious effects beyond just induction of Aging 2. Clearly, Aging 1 should be included in the model.
Aging 1 is modeled similar to Aging 0, using subscript 1 to identify Aging 1 parameters:
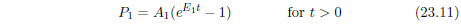
Inclusion of this third aging disease upgrades the model to:
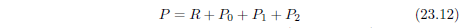
Note that the mathematical forms of the Aging 0 and Aging 1 terms, P0 and P1, are identical. This poses potential trouble for the least squares fit. It is possible for the least squares search algorithm to find two-disease solutions by setting either A0 or A1 effectively to zero, or by setting A0 = A1 and E0 = E1. Fortunately, these two-disease solutions, which are not of interest in the present context, can be avoided by choosing starting values for the free parameters judiciously.
Figure 23.11 shows the final weighted least squares fit of the two-phase, three-disease model to the 2016 actuarial life table datasets for U.S. males, and Figure 23.12 shows the same thing for U.S. females.
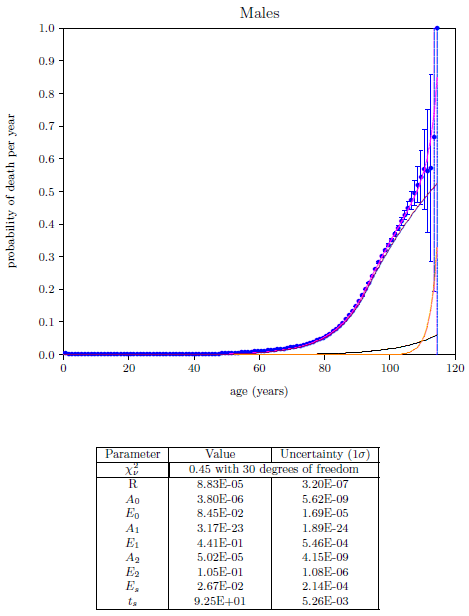 |
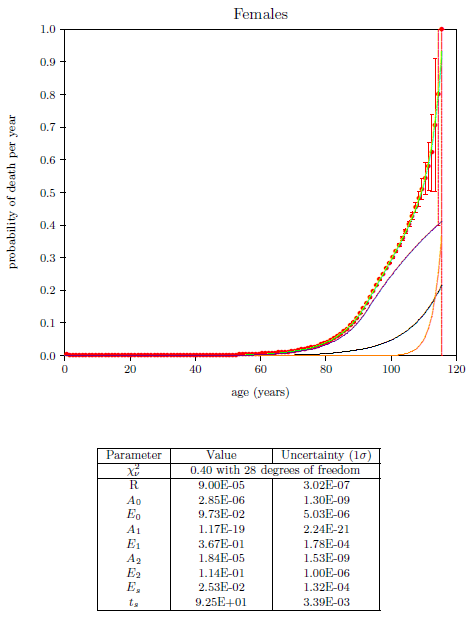 |
The male dataset was used to investigate the effect on the final fit of choosing different fixed values of t2. Values of 0.9, 7.5, and 14 years were investigated. The visual effect on the final fit was negligible in all three cases, but t2 = 14 years gave the best goodness-of-fit parameter value—0.45 compared to 0.50 for the other two choices—so it was used for these two figures. This result strengthens the possibility that onset of Aging 2 for modern humans may be near 14 years, and we will tentatively side with this choice going forward. Note, however, that this result of the modern actuarial table model cannot be regarded as definitive because of the lack of usable data points in the early decades, as previously discussed.
These two figures make it immediately clear that all three aging diseases are indeed present in the actuarial life table datasets and that the previous, two-disease model had mistakenly found Aging 1 as the second-found aging disease rather than Aging 0.
Because Aging 0 and Aging 1 share the same mathematical form, it is in principle possible for a two-disease model to find either of these two as the second-found aging disease. We inadvertently found Aging 1 when we had set out to find Aging 0. This, however, spurred us on to look for a three-disease solution, which worked out well. But, what would have happened had we found Aging 0 instead? Looking at the final graphs, it is clear that Aging 0, without Aging 1, is unable to fit the centenarian data properly. Thus, the visual fit of the centenarian data would not have been much improved, once again suggesting that a third aging disease was yet missing, spurring us on to look for a three-disease solution. So, either way, the actuarial life table data appear to solicit a three-disease model.
The fit of the proper, three-disease model to the male and female datasets immediately informs us that Aging 1 is indeed the least dominant of the three aging diseases—it shows up last and plays little direct role in aging deaths prior to 100 years of age. It, of course, plays a severe, indirect role in aging deaths by its induction of Aging 2 disease in childhood or early adolescence. Meanwhile, in stark contrast to its earlier quiescence, it quickly becomes a significant player in centenarian mortality for both males and females. The implication of this discovery is obvious. Aging 1 is MePiA deficiency disease. MePiA deficiency disease can be quickly cured by supplementing one's diet with an adequate daily amount of vitamin MePiA. The three-disease model shows that centenarians are especially sensitive to this deficiency disease. Centenarians, therefore, need to be especially careful to supplement their diets with the recommended daily intake of vitamin MePiA.[108] According to the model, centenarians following this advice significantly improve their chances of breaking current longevity records.
The proper, three-disease model now brings the fitted Aging 0 curve into harmony with real-life experience. The Aging 0 curve now rises above the level of random extraneous deaths at about age 40 (visible when the scale is expanded; not shown) and continues to increase exponentially from there. It accounts for about 11% of aging deaths in males and 25% in females at age 50, for example.
While mortality due to MePA deficiency disease (Aging 0) is very low prior to age 40, this does not mean that vitamin MePA is not needed prior to age 40. The actuarial life table data show only mortality, not morbidity. Optimal health at all ages is dependent on a proper daily intake of all of the vitamins. When a vitamin is deficient in the body, biochemical pathways become inaccessible, and cells are unable to manufacture biomolecules or perform key functions needed for growth and maintenance. This weakens every cell in the body. Early in life, most members of a same-age population will be healthy enough to survive despite the presence of vitamin MePA deficiency disease, but a few, suffering from other serious ailments, will not. In addition, ability to survive life-threatening events such as fires or automobile accidents will be diminished by the whole-body weakening resulting from the presence of vitamin MePA deficiency disease. In consequence, the probability of death due to Aging 0, while tiny prior to age 40, is still real and measurable even in the early childhood years. Thus lifetime, daily supplementation with the anti-aging vitamins, MePiA and MePA, is essential for optimal health and longevity at all ages.
Figures 23.11 and 23.12 show a clear disparity between males and females in relative sensitivity to Aging 0 (black lines). The model finds that women are more sensitive to vitamin MePA deficiency disease than are men. This provides further evidence of the validity of the two-phase theory of modern human aging. Aging 0 is vitamin MePA deficiency disease, and vitamin MePA deficiency disease seems to engender autoimmune diseases.[109] Meanwhile, women are far more susceptible to autoimmune diseases than are men.[110]
Autoimmune diseases are the third most common category of disease in the United States after cancer and cardiovascular disease, affecting ∼5 to 8% of the population or 14.7 to 23.5 million people. Conservative estimates indicate that ∼78% of the people affected with autoimmune diseases are women.The model's finding, that women are more sensitive to Aging 0 than are men, is clearly supported.
On the flip side, the model finds (Figures 23.11 and 23.12) that men are more sensitive to Aging 2 (violet lines) than are women. This further corroborates the two-phase theory of modern human aging. Specifically, the two leading causes of death in the U.S. are heart disease and cancer. The model makes it clear that Aging 2 is, by far, the leading cause of all deaths. This immediately implies that heart disease and cancer are symptoms of the more fundamental disease, Aging 2. (Note, in passing, that this, in turn, implicates mitochondrial dysfunction as the root cause of both heart disease and cancer.) Since, according to the model, men are more sensitive to Aging 2 than are women, an immediate prediction of the model is that the age-specific incidence of both heart attack and cancer will be higher in men than in women. This is found to be true in both cases.[111]
Perhaps the most conspicuous observation from Figures 23.11 and 23.12 is that Aging 2 deaths dominate human aging at present, far outstripping all other causes of death combined. Table 23.1 quantifies this.
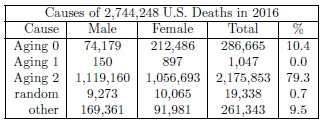 |
Roughly 4 out of 5 deaths are due to Aging 2. Yet again the implication is clear. To prevent onset of Aging 2 and to reverse the course of established Aging 2 disease, dietary supplementation with MePiA is essential.
The results of the two-phase, three-disease model (Figures 23.11 and 23.12 together with Table 23.1) make it clear that Aging 0 is responsible for many, many deaths at present. Table 23.1 ascribes to Aging 0 a total of 286,665 U.S. deaths in 2016. This means that more than one in ten U.S. deaths in 2016 was due to Aging 0. Of course, there is nothing particularly special about 2016. This same loss of life happens each year. The tragedy is that it continues to happen even though it is now totally unnecessary.
Once again, the implication is clear. Aging 0 is vitamin MePA deficiency disease. Just as scurvy can be cured in short order by adequate daily intake of vitamin C, so Aging 0 can be cured in short order by adequate daily intake of vitamin MePA. Dr. Aardsma's Anti-Aging Vitamins dietary supplement is able to eradicate Aging 0, just as vitamin C has eradicated scurvy, and just as vitamin B3 (niacin) has eradicated pellagra, and just as vitamin B1 (thiamin) has eradicated beriberi, etc., and the model finds that when this happens, over a quarter million U.S. lives, of all ages and both sexes, will be saved per year.
The two-phase theory of modern human aging has been shown to provide a quantitative explanation of the pattern of modern human actuarial life table data. This appears to be the first time the detailed pattern of human actuarial life table data has been scientifically and mathematically explained by any theory of aging.
A good fit of the dataset could not be obtained with a single exponential (i.e., a single-aging-disease) model. To get a statistically and visually good fit, two exponentials were required and the dominant exponential had to be capable of saturation. While this arrangement was able to yield a good fit statistically and visually, the resulting solution did not accord with reality. To obtain a true-to-life solution while retaining a good fit statistically, a third exponential had to be added. Thus it was found that the dataset demands more than a single-aging-disease model and solicits a three-aging-disease model. In this way, the modern dataset corroborates the two-phase theory of modern human aging, which theory predicts the simultaneous presence of three distinct aging diseases in modern actuarial data.
The process of fitting the two-phase theory of modern human aging to the 2016 actuarial life table datasets for males and females has generated a wealth of new insights into modern human aging. Most importantly, it has uncovered the possibility of immediately saving over a quarter million U.S. lives per year, currently perishing unwittingly due to now easily treatable vitamin MePA deficiency disease. This unnecessary loss of life is a national health emergency. To protect the population from this disease and, ultimately, to eradicate aging, a policy of universal, lifelong, daily supplementation of diets with vitamins MePA and MePiA should be implemented immediately. The alternative yields only massive, ongoing, unnecessary human suffering and death.
The 926-year life expectancy at birth enjoyed by pre-Flood individuals is an enormous improvement on our current 77-year life expectancy at birth, but the model for the ancient biblical life span data says that it is possible to do even better. This first comes into view with the sudden jump in life expectancy from 926 up to 1,151 years in 4444 B.C. (Figure 24.1).
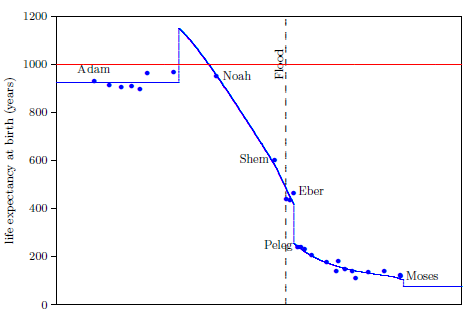 |
This jump is real, as mentioned previously. It results from the fact that individuals born close enough to the Spike lived into the Spike, assuming they did not get killed by the Flood, where they were subject to a very high MePiA intake. This high intake topped up their body reservoir of MePiA and thus increased their life spans.
The model says that the 926-year average life span of pre-Flood humans was not a limiting life span for humans any more than our current 77-year average life span is a limiting life span for humans. There appears, in fact, to be no limiting life span for humans.
This is not to say that MePiA grants immortality, for it most certainly does no such thing. MePiA does not stop speeding bullets, for example. Adequate daily intake of MePiA merely removes Aging 1 and Aging 2 as particular causes of death.
When the aging diseases, including Aging 0, have been removed, humans, in fact, have potential to live healthy, youthful lives lasting thousands of years. Aging is not a natural process for the human species. It is a disease process.
At present, we live in a world full of people who are at various stages of sickness due to the three aging diseases. Disease progression is nearly zero in infants and nearly terminal in centenarians.
The aging diseases are just now beginning to be driven back. Eventually, these diseases will be eradicated from our planet. In the meantime, what extension of life span might aging-diseased individuals anticipate who have begun to take Dr. Aardsma's Anti-Aging Vitamins? The following section undertakes to answer this question. The analysis is necessarily technical and mathematical once again. For readers not interested in the technicalities, a presentation and discussian of the results begins in Section 24.2.
There are no experimental data on this question at present, nor are there likely to be any experimental data on it any time soon. Experimental evidence for life lengthening due to the anti-aging vitamins has only recently been obtained from a pilot study with mice, discussed in an earlier chapter. The pilot study took nearly three years to complete. Because humans live about 30 times longer than mice at present, experimental evidence for life lengthening in humans seems likely to be decades away. At present, to predict the probable effect of the anti-aging vitamins on human life spans, it is necessary to rely on theory.
Fortunately, the scientific theories which must be relied on in this case—the two-phase theory of human aging, and the two-phase theory of modern human aging—appear to be sound. The two-phase theory of human aging has successfully explained and mathematically modeled the ancient biblical data recording multicentenarian life spans. It has given rise to substantial anecdotal testimonial evidence of health benefits realized by users of one or both of the anti-aging vitamins.[112] And it has received its first direct experimental corroboration in the form of life lengthening in animal models via the pilot study with mice mentioned previously. Meanwhile, its extension into modern times, the two-phase theory of modern human aging, has successfully modeled modern male and female actuarial data, apparently for the first time ever. While theory is all we have to go on at present, there are good reasons to trust the requisite theory.
No one can say with certainty how long anybody will live, of course, but on average at present, in our aging-diseased population, we expect infants to enjoy a life span of something like 70 or 80 years, and we expect 40-year-olds to live something like another 30 or 40 years. Because discrete, individual life spans cannot be predicted, it is necessary, for scientific purposes, to talk about the average life span of a group of individuals. The average life span remaining for a group of same-age individuals is called their "life expectancy." The life expectancy today for infants will be something like 70 or 80 years, and the life expectancy for 40-year-olds will be something like 30 or 40 years.
Questions about life expectancy are routinely answered today using actuarial life tables. An actuarial life table shows the probability of dying within the year from one birthday to the next. The present analysis uses the same (2016) Social Security Administration actuarial life table which was used in the previous chapter's modeling study.[113] Both the number of survivors and the life expectancy versus age are included in the 2016 SSA table. In the present chapter, the 2016 table is treated as representative of what is normal in the case of modern, well-cared-for humans whose diets completely lack the anti-aging vitamins from birth on.
Figure 24.2 shows the table's life expectancies versus age graphically.
 |
Life expectancies for males and females are graphed separately, reflecting their separation in the actuarial table. This graph shows that newborn male babies have a life expectancy of 76 years, and newborn female babies have a life expectancy of 81 years; 40-year-old males have a life expectancy of 38.6 years, and females 42.5 years; and 80-year-old males have a life expectancy of 8.3 years, and females 9.7 years.
What happens to these life expectancies when an individual converts from a diet devoid of the anti-aging vitamins to a diet containing adequate anti-aging vitamins?
To answer this question quantitatively, it was necessary to use numerical methods via a computer program once again. The program calculated the new life expectancies for males and females first beginning to take the anti-aging vitamins at some age between birth and 113 years. To understand how the computer algorithm works, it is first necessary to understand the idea of physiological agedness.
Calendar age is a count of the number of calendar years which have passed since a person was born. Physiological age is a measure of the physiological progression from birth to "old age" of a person's body. We are accustomed to equating these two at present because the two start out together at zero. Physical growth toward maturity then progresses at a roughly equal rate in all individuals, making it possible to guess a young person's calendar age fairly accurately based upon his degree of physical maturity. Meanwhile, at the same time as growth toward maturity has been happening, the aging diseases have been progressing, making it possible to guess an adult's calendar age fairly accurately based upon the extent to which his aging diseases have progressed.
While it is now necessary to break the careless habit of equating physiological age with calendar age, this habit provides a simple and convenient scale for discussing how youthful or otherwise a person's body appears to be. It is immediately clear what is meant if one says at present that a person looks to be one year old, for example. It is clear that one means that physical development toward maturity has just gotten started. It is also immediately clear what is meant if one says at present that a person looks to be 100 years old. It is clear that one means that physiological agedness, due to the aging diseases, has come very nearly to a terminal condition. It would also be clear what was meant should one say at present that a pre-Flood individual having a calendar age of 700 years had a physiological agedness of 30 years. It would be clear that this meant that a 700-year-old person back then had the physical body of a 30-year-old today.
The physiological agedness scale is defined to be a measure of the physiological progression of the aging-diseased human body from cradle to grave, conveniently calibrated by our everyday experience at present.
Related to the concept of physiological agedness is the idea of terminal maturity, when physical development toward adulthood ceases. What calendar age should be assigned to this point?
In the physiological agedness scale, growth toward maturity mixes together with decline due to aging, blurring this point. In the ancient past, when people lived much longer than aging-diseased modern humans do, this was not the case. Modern humans are born with severe, congenital Aging 0 and Aging 1 diseases, and Aging 2 disease starts up probably within a few years of 14 years of age, so that clinical symptoms of aging have already set in before maturation has come to completion. Before the Flood, humans were born with Aging 1 disease, as we are today, but their Aging 1 disease was slight compared to ours. They matured from infancy to adulthood in the first few decades of life, much as we do today.[114] Because there is almost no mortality due to Aging 1 until Aging 2 has become far advanced, pre-Flood individuals enjoyed a long interval of time between terminal maturation and the onset of clinical symptoms of Aging 2.
The computer algorithm adopts 34 years—two decades following modern onset of Aging 2—as the calendar age of terminal maturity for modern humans. The possible range for terminal maturity today seems to extend from the early twenties to the early forties, making 34 years seem an appropriate estimate.
Consider the example of a modern, 40-year-old male beginning to take the vitamins. His calendar age and his physiological age start out both together at 40 years. After he has taken the vitamins for one year, his calendar age has increased 1 year making him 41 years old. Meanwhile, because of the vitamins, his body has begun to heal of his aging diseases and has become a little more youthful. As a result, he has less chance of dying during his next year of life. His life expectancy has increased.
How much has his life expectancy increased? The computer program calculates this based on what is known about the rate of healing of the three aging diseases at present. Healing of both Aging 1 (vitamin MePiA deficiency disease) and Aging 0 (vitamin MePA deficiency disease) appears to be rapid, as previously discussed. To keep things simple, the program treats the healing of both of these aging diseases as instantaneous as soon as supplementation with the vitamins begins. The healing of Aging 2, which is a mitochondrial genetic disease rather than a deficiency disease, cannot be approximated so simply. It heals very slowly.
The zeroed-psephomere condition, which is Aging 2, heals, once supplementation begins, by the slow process of cell replacement, as previously discussed. Dysfunctional cells, including Aging 2 diseased cells, slowly get replaced by cells having long psephomeres produced by the stem cells.
How long does this replacement process take to eliminate Aging 2 diseased cells from the body? Figure 24.3 displays
 |
the calculation of this which was produced by the digital organism model in a previous chapter. This figure shows that, for starting ages other than the 5-year-old case, it takes 11 years for half of the zeroed-psephomere cells initially present within the digital organism to be replaced. This suggests the simple procedure of approximating the regression of Aging 2, once a person begins to supplement with the anti-aging vitamins, as a decaying exponential having a half-life of 11 years.
For the present purpose of estimating life expectancies, this decaying exponential is used both to estimate the change in a person's physiological age year by year and to calculate the person's probability of death per year due to Aging 2. The probability of death per year due to Aging 2 is calculated as:
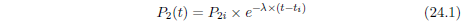
where t is the person's calendar age, λ is the decay constant ( = ln(2) / t1/2 = 0.693 / 11 = 0.063 per year), ti is the person's age when supplementation began, and P2i is the person's probability of death per year due to Aging 2 at ti. The decrease in the person's physiological age, for a person over the age of 34 years, is calculated each time step, δt, as δt × ( 1 - e-0.063 × (t - ti) ), where δt is in calendar years.
Summarizing to this point, after one year of supplementation, the 40-year-old man has become 41 years old in calendar years and (40 - 1 × ( 1 - e-0.063 × (41 - 40) ) =) 39.939 years old physiologically. He has lived another year and become slightly more youthful in the process.
This neglects the possibility that he may not have lived to his 41st birthday, an oversight which will be corrected shortly. Assuming he has survived to age 41, it is easy to calculate what happens in his 42nd year. His calendar age advances to 42, and his physiological age reduces further, to 39.878 years.
The program carries on in this way (albeit at much higher computational precision and with much smaller time steps) year by year, advancing the man's calendar age and reducing his physiological age. Now consider the possibility that the 40-year-old man died prior to his 41st birthday. This possibility is related to the probability of death per year for 40-year-old-men. This probability is made up of the probability of dying due to the aging diseases between age 40 and 41, plus the probability of dying due to other causes such as car accidents. For individuals not supplementing with the anti-aging vitamins, the SSA actuarial life table informs us that this probability is 0.00242. This means that out of 100,000 anti-aging-vitamins-deficient men turning 40 years old, 242 die in their 41st year for one reason or another: heart attack, brain aneurysm rupture, car accident, violent crime, etc. But the 40-year-old man in our example is not anti-aging-vitamins deficient. He has been taking the vitamins since he turned 40, curing his Aging 0 and Aging 1 diseases, and diminishing his probability of death due to Aging 2. The program sorts this all out by first subtracting from the actuarial table probability of death the calculated probability of death due to all three aging diseases. These probabilities are calculated using the fitted curves for Aging 0, Aging 1, and Aging 2 from the previous chapter. This leaves just the probability due to extraneous deaths. The program then adds to this the appropriate amount of Aging 2 probability of death per year, calculated from Equation 14, giving the appropriate probability of death for the anti-aging-vitamins-supplementing 40-year-old man. The program uses this probability as part of its calculation of the new life expectancy. It starts with 100,000 men of age 40 and keeps a total of how many survive year by year of the calculation. When none remain, it stops the calculation and calculates the new life expectancy value for age 40 males.
That explains most of what the computer program does. Only two further details need to be mentioned. First, notice that the probability of death for our example 40-year-old male in his 42nd year is not the value found in the SSA actuarial table for a 41-year-old male. Rather, it is the value one gets from the table for a 39.939-year-old male. (It is necessary to interpolate the table to get this value.) Because his body has become more youthful, his probability of death does not increase to the value shown in the table for a 41-year-old male but rather reduces to the value appropriate to a 39.939-year-old male. It is the physiological age which must be used in the 2016 actuarial life table for the calculation, not the calendar age. Said simply, the program is using the SSA actuarial table to learn the probability of death due to extraneous causes, and this probability is related to physiological age, not calendar age.
Second, notice that there are three distinct cases to deal with:
The new life expectancies, calculated by the computer program, are shown by the heavy dotted lines in Figure 24.4. The lighter lines show the life expectancies from the 2016 actuarial table, which are normal to aging-diseased individuals today.
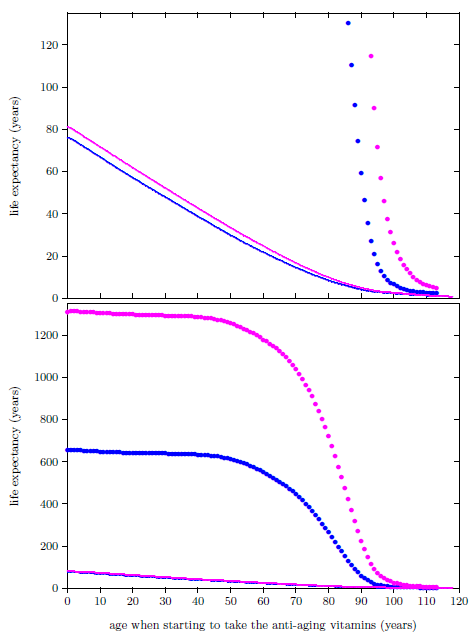 |
Returning to the teaching example of the male beginning to supplement with the anti-aging vitamins at 40 years of age, the graph shows that his life expectancy at age 40 jumps from the SSA actuarial table value of 39 years (giving a total life span expectancy of 79 years) to 633 years (giving a total life span expectancy of 673 years). For a female beginning to supplement with the anti-aging vitamins at 40 years of age, the graph shows that her life expectancy at age 40 jumps from the SSA actuarial table value of 43 years (giving a total life span expectancy of 83 years) to 1286 years (giving a total life span expectancy of 1326 years).
As a further teaching example, consider an elderly male beginning to supplement with the anti-aging vitamins at 90 years of age. The top graph shows that his life expectancy at age 90 jumps from the SSA actuarial table value of 4 years (giving a total life span expectancy of 94 years) to 59 years (giving a total life span expectancy of 149 years).
The new life expectancies for females come out roughly double the new life expectancies for males. This happens because of the large difference in the probabilities of extraneous deaths—the only remaining causes of death once aging disease deaths have been removed—for young adult males and females. Notice, again, that, as the computer algorithm is designed, once a person begins supplementation with the anti-aging vitamins, his physiological age converges on 34 years. This means that he will spend most of his long life at or near physiological age 34 and be subject to the extraneous causes of death normal to a 34-year-old. At age 34 years, the extraneous death probability for females is 0.00077 per year while for males it is double at 0.00156 per year. The three leading causes of death in this age group today are unintentional injury, suicide, and homicide.[115] The leading causes of unintentional injury death are unintentional poisoning (0.00019), dominated by drug abuse overdose, followed by motor vehicle traffic deaths (0.00012), no doubt dominated by alcohol abuse, and unintentional fall deaths (0.00011).[116] The fact that young males tend to take more unnecessary risks than young females is clearly reflected in these probability-of-death numbers.
This emphasizes that one's life expectancy, once aging deaths have been removed, is strongly influenced by one's lifestyle choices. This is further illustrated by the fact that pre-Flood males had life expectancies at birth of nearly 1000 years, significantly greater than the roughly 650 years calculated by the computer program for modern males. This results from lifestyle differences. Pre-Flood males did not do drugs and did not drive automobiles, for example. The computer program makes no allowance for how one chooses to live—its analysis is based entirely upon national averages for the year 2016—so Figure 24.4 should be viewed with this in mind. One can do better on average than the calculated life expectancies if one chooses to live in less risky ways.
Another thing to bear in mind is that the nature of life expectancy itself has changed. Its definition has not changed, of course. It is still the average life span for a same-age group of individuals. But the shape of the survival curve for such a group changes dramatically once the aging diseases have been removed. When aging deaths are dominant, as they are today, life expectancy means the age at which just about everybody dies. When aging deaths have been removed, the meaning changes, as Figure 24.5 illustrates.
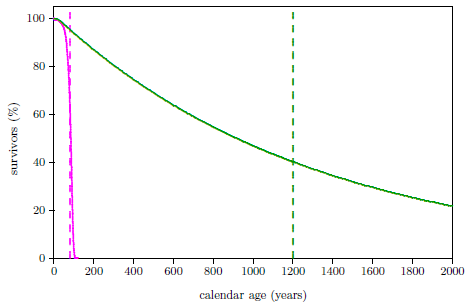 |
The dashed vertical lines show the respective life expectancies at birth. The dashed magenta line does, indeed, correspond to when just about everybody dies, but the dashed green line most certainly does not. Nearly one fifth of females supplementing with the anti-aging vitamins, shown by the solid green line, are still living 800 years after their life expectancy at birth, shown by the dashed green line. The reason nearly everybody dies at the same time when aging is present is that aging disease kills everybody off while they are all still tragically young.
Figure 24.5 also begins to illustrate the very long life spans which are possible once aging deaths have been removed. It shows more than one fifth of women living to 2,000 years of age. If the green curve were extended, it would show nearly one percent of women with life spans in excess of 6,000 years.
Such great ages may seem impossible or unbelievable, but logic and science insist on them. Because the aging diseases have been taking their toll on humans for thousands of years, we have grown up assuming that a 70- or 80-year life span is normal. But we are entirely mistaken in this assumption. We have grown up in abnormal times with abnormally shortened human life spans relative to the totality of human history. Earth's oldest written history, found in the biblical book of Genesis, records human life spans of nearly 1,000 years prior to Noah's Flood. These ancient observational data of long human life spans are every bit as meaningful as our modern observational data of short human life spans, and they immediately teach us that it is wrong to assume that our modern times are normal.
To be perfectly clear, these results, with individuals living in excess of 6,000 years, though calculated from theory, are meaningful in the real world today, and they apply to ordinary people like us.
What do 6,000-year-old people look like? Do they look very, very old? No. The "old" look is purely a consequence of the aging diseases. They have no aging diseases. They look like mature, youthful adults.
How do 6,000-year-old people die? They die from all the sorts of things not related to aging disease which kill normal mature youthful adults today: car accidents, homicides, suicides, lightning strikes, drownings, infections, natural catastrophes, wars, snake bites, etc.
But note that even a life span of 6,000 years does not mean that those so blessed run out of time and perish around 6,000 years of age. Running out of time happens only today. It happens because aging diseases progress exponentially. If you do not die due to some extraneous cause, then you can absolutely count on the aging diseases killing you, in the absence of the anti-aging vitamins, before you have lived even 125 years. And because aging diseases progress exponentially, you can absolutely count on most people dying today in their 70's or 80's. But once aging is removed, one does not run out of time. Rather, one eventually runs out of luck. For females with a 1,311 year life expectancy at birth, somewhat more than half will run out of luck sometime prior to 1,311 years, and somewhat less than half will run out of luck sometime after 1,311 years. When exactly they run out of luck will be randomly distributed, not all bunched close to 1,311 years.
The foregoing numerical results of the computer calculation should not be treated as highly precise certainties. They are not that. They are approximations showing likelihood, not knowns showing certainties. They entail several assumptions and approximations which cannot be guaranteed. For example (1) that Aging 2 sets in at 14 years of age, (2) that Aging 2 heals as a decaying exponential with a half-life of 11 years, and (3) that the probability of extraneous deaths for two individuals, both 34 physiological years old, but one 34 calendar years old and the other 340 calendar years old, will be the same.
Nonetheless, what the computer calculation communicates loudly and clearly is that the advent of the discovery of the anti-aging vitamins has opened a whole new era for the human species. It makes unquestioningly clear the fact that the seemingly refractory "threescore years and ten; and if by reason of strength… fourscore years"[117] time limit on life, which has dominated all of humanity for some four and a half thousand years, has been lifted at last.
Figure 24.4, shown previously, displays, in graphical form, a world in transition. The transition is away from the dominance of the severely life-shortening aging diseases to a world free of them. Ultimately, life spans measured in multiple centuries will be the normal experience of humanity. The ancient prophet, with poetic aptness, put it this way:[118]
For the youth will die at the age of one hundred
And the one who does not reach the age of one hundred
Shall be thought accursed.

Figure 25.1 is possibly the most succinct way of showing the benefit to longevity of beginning lifelong supplementation with the anti-aging vitamins at any age.
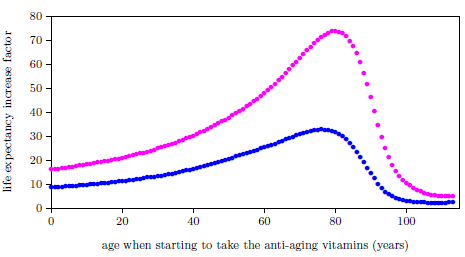 |
Because longevity is so intimately dependent on health, this graph may be interpreted as indicating benefit to health as well. It shows that a male who starts lifelong supplementation at age 40 will increase his life expectancy by a factor of 16, and a female who starts lifelong supplementation at age 40 will increase her life expectancy by a factor of 30. While many would give almost anything if somehow another 10 years could be added to their life, here is opportunity for more than another 10 lifetimes to be added to one's life.
Notice that there are no losers with this graph. Both males and females, regardless of their age when they begin supplementation with the anti-aging vitamins, increase their life expectancies. The smallest increase, for males aged 107–110 years, is a factor of 2.1.
Figure 25.2, copied here from the previous chapter, shows that, for individuals supplementing with the anti-aging vitamins, life expectancy declines with increasing initial supplementation age. The decline is shallow at first, but becomes more and more rapid at higher ages.
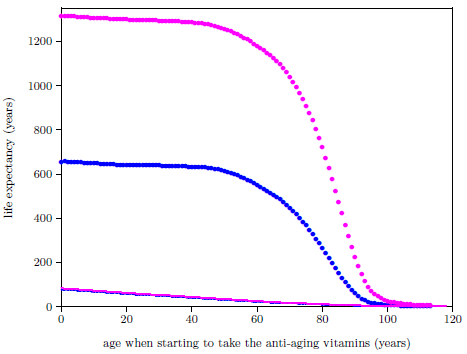 |
This observation leads immediately to a most important point of strategy for personal use of the vitamins. We will call this point of strategy Rule 1.
Rule 1: Whatever your age, begin taking the anti-aging vitamins without delay.The only exception to this rule would be nursing infants, who are expected to be furnished naturally with the vitamins through their mother's breast milk. It is the mother, not the nursing child, who should apply Rule 1. If you are concerned about possible interactions of the anti-aging vitamins with medications you are presently taking (none currently known), get your health professional involved to help you be able to follow Rule 1 safely and without anxiety.
The health and longevity cost of breaking this rule can be very large. Consider, for example, a female who is first introduced to the anti-aging vitamins at age 50. Unfortunately, life has made her a bit cynical. She thinks "snake-oil salesman" to herself, and turns a deaf ear. When she reaches age 60, she changes her mind. Her hair has gone white and her arthritis takes the joy out of life much of the time. Her friend, who started on the anti-aging vitamins a long time ago, has no arthritis and is simply more healthy and vibrant. She finally decides to start taking the anti-aging vitamins.
Had she started taking the anti-aging vitamins at age 50, her life expectancy would have been 1,258 years. At age 60, it has declined to 1,204 years. Of the 54 year difference, she has lived 10 years and lost 44 years. She has managed to squander 44 years of life expectancy—over half a lifetime, in present terms—in just 10 years.
It might be supposed that, nonetheless, this is not too bad—she still has 1,204 years of life expectancy remaining. But this is wrong-headed entirely. She now has a 60-year-old body to contend with. If she had begun taking the vitamins 10 years earlier, her aging diseases would have regressed rather than progressing another 10 years. It will likely take her several decades to get back to where she had been physiologically 10 years previously.
But, of course, she may never make it another several decades. The incidence of cancer is strongly correlated with physiological age. Imagine that her body had developed a cancerous cell in her 59th year which, had she begun taking the anti-aging vitamins at age 50, would not have arisen. Imagine that the cancerous cell has a doubling time of one year. Twenty years later, in her 80th year, this single cell has multiplied and become a pea-sized tumor. Her body is now growing slowly younger year by year, but she is still in her very late fifties physiologically, and her body has no natural defense against this particular cancer. Her only hope is removal of the tumor. This, of course, requires that the tumor be somehow detected. The longer it goes undetected, the greater the probability that it will metastasize, at which point removal of the tumor will do little good. Meanwhile, all of this might have been so easily avoided.
The aging diseases are not to be toyed with at any age. Wisdom calls for deliberate action against them at one's earliest opportunity.
There is only one other major point of strategy. We will call it Rule 2.
Rule 2: Take the anti-aging vitamins without fail every day.This rule does not need much elaboration. It is pretty obvious. Would you knowingly eliminate any of the other vitamins from your diet?
The only reason Rule 2 needs to be explicitly stated and emphasized is that it is easy to get off track with the anti-aging vitamins. Healing of Aging 2 is slow. One is not likely to notice much happening, especially after initial gains in health due to the healing of Aging 0 have become the new norm. It is all too easy to fall prey to the feeling that the anti-aging vitamins aren't doing anything for you anymore and to stop taking them. One must not give way to such impulses. Seriously now, would you intentionally eliminate any other vitamin from your diet?
People who unwittingly eliminated vitamin C from their diets (mostly long-ago sailors on long voyages failing to get fresh fruits and vegetables) soon became diseased with scurvy, suffering horribly from bone and muscle pain, easy bruising and bleeding, gum disease, depression, and anemia for weeks or months before succumbing to this ancient dread disease. Nobody ever intentionally eliminated vitamin C from his diet. Nobody wants scurvy.
People who unwittingly eliminated vitamin B3 (niacin) from their diets (thousands of Southerners in the 1920's, eating inexpensive milled Midwestern corn so they could convert more of their land to cotton)[119] soon became diseased with what is called pellagra, suffering horribly from dementia, diarrhea, and dermatitis for weeks or months before succumbing to this relatively modern dread disease. Nobody ever intentionally eliminated vitamin B3 from his diet. Nobody wants pellagra.
People who unwittingly eliminated vitamin B1 (thiamine) from their diets (by eating mostly polished, white rice) soon became diseased with what is called beriberi, suffering horribly from loss of appetite, irritability, mental confusion, peripheral neuropathy, swollen hands and feet, and chest pains from a malfunctioning heart for weeks or months before succumbing to this old-time dread disease. Nobody ever intentionally eliminated vitamin B1 from his diet. Nobody wants beriberi.
Put your intellect, not your feelings, in charge. The aging diseases are insidious and deadly. You must never let down your guard against them. Treat them as deadly enemies, for they are deadly enemies.
Many foods and drinks are fortified with the traditional vitamins today, making it difficult not to get an adequate daily intake of these vitamins. It is to be hoped that this will eventually be the case with the anti-aging vitamins as well—that governments will come up to speed with the anti-aging vitamins and put measures in place to make it difficult for citizens not to get an adequate daily intake of them. But history shows that governments generally have a pretty poor track record with this sort of thing. In the meantime, you must look after yourself. This is not hard to do. Simply follow Rule 1 and Rule 2:
Rule 1: Whatever your age, begin taking the anti-aging vitamins without delay.
Rule 2: Take the anti-aging vitamins without fail every day.

Once Aging 2 saturation is allowed for, the two-phase theory of modern human aging is able to explain the detailed pattern of modern actuarial life table data. This appears to be the first time a mathematically rigorous, scientific explanation of these data has been achieved.
When applied to modern actuarial life table data, the two-phase theory shows that approximately ten percent of the population is dying at present from Aging 0. These deaths are now unnecessary and easily, rapidly remedied via dietary supplementation with MePA.
The three aging diseases together account for ninety percent of deaths at present. Dietary supplementation with both MePiA and MePA is able ultimately to eliminate all of these aging deaths. This realization transforms the current concept of aging from an unfortunate, unavoidable, natural phenomenon to an intolerable, easily remedied, global pandemic.
Healing of the aging diseases begins as soon as daily dietary supplementation with MePiA and MePA at appropriate levels is initiated. Symptoms associated with Aging 0 and/or Aging 1 may be expected to resolve generally in under a year. In contrast, symptoms associated with Aging 2 may take decades to resolve.
The discovery of the causes and the cures of the aging diseases ushers in a new era for humanity. Eradication of these diseases will cause life expectancies to skyrocket.

In closing, we, the authors of this volume, want to share with you what we and our wives have personally experienced with the anti-aging vitamins. Of the three aging diseases, only Aging 0 (MePA deficiency disease) is expected to display rapid resolution of clinical symptoms upon initiation of daily supplementation with the anti-aging vitamins. Aging 1 (MePiA deficiency disease) is expected to resolve rapidly as well, but because we are unable to sense free-radical damage to our mtDNA in any direct way, there are expected to be no rapidly-resolving pains or other symptoms associated with this disease. Rapid resolution of Aging 1 allows Aging 2 to begin to heal, but healing of Aging 2 is a slow process, as previously discussed. So personal testimonials, at this still early stage of discovery of the anti-aging vitamins, will necessarily be dominated by MePA. Come back in thirty years, and we will tell you about what MePiA has done for us.
As briefly mentioned previously, MePA was discovered several years before the discovery of MePiA. When it was discovered, vitamin MePA was mistakenly thought to be vitamin X and to be the sole vitamin responsible for aging disease. As a result, it was tested with early volunteers by itself, without accompanying MePiA.[120] In consequence, the effects ascribed to MePA in the present chapter are, in fact, known not to be due to the second-discovered anti-aging vitamin, MePiA.
I (MPA) can remember Dad talking about "vitamin X" when I was a small child. Little did I dream then that someday I would be part of the Aardsma Research & Publishing research team working with the anti-aging vitamins or have the opportunity to contribute to the second edition of Aging: Cause & Cure. Given that this second edition, as did the first, relies heavily on the brilliant work of my father over the last several decades, both in Bible chronology and in human aging, it is indeed an honor to be a coauthor of this edition.
I am a healthy 31-year-old who makes an effort to live a healthy lifestyle, but I am not fanatical about exercise or health food. I started daily supplementation of my diet with MePA a little over three years ago, in late 2017. I began to supplement my diet daily with both MePA and MePiA in mid 2019, about 1.5 years ago, and continue to do so. I have been a Rule 2 follower (see Chapter 25) since I started MePA in 2017.
The anti-aging vitamins obviously play a role in health. What has my overall experience been with health while taking the anti-aging vitamins? Exactly what Chapter 24 predicts for a healthy individual beginning supplementation with the anti-aging vitamins in his late 20's: very little noticeable change. I continue to function as a healthy young adult. In fact, according to the model, this uneventful state of young adulthood, punctuated by life's normal bumps and scrapes, may be all I have to report for a very, very, very long time. The biggest impact of taking MePA which I have experienced is a reduced susceptibility to common illnesses such as colds and the flu.[121] While uneventful healthiness might seem a bit boring, a (now) rare bout of the common cold is all that is required to remind me that "uneventful healthiness" is a wonderful thing.
I will never know what diseases and ailments I would have had through life had I not started the anti-aging vitamins as a young person. Despite being a scientist, that is one experiment I am glad I am not performing. If I stop supplementation with the anti-aging vitamins, both Aging 0 disease (cured for me in late 2017) and Aging 1 disease (cured in 2019) will be back all too soon, and the slowly regained Aging 2 healing will be rapidly lost. It is a great blessing to have been able to start taking the anti-aging vitamins when I was relatively young and healthy.
This section is largely written for younger people like myself who are not yet feeling the "bite" of the aging diseases all that strongly.
I started out feeling that aging was all about old people. To align with reality, my thinking about aging had to undergo a paradigm shift. I, as a young person, had to realize, not that I would eventually grow old and have an aging problem, but that I already had an aging problem that I needed to do something about right then.
Once I was thinking about aging correctly and recognized my problem, I then had to change my behavior to address my aging problem. I had to modify my habits and begin to take the anti-aging vitamins every day. In order to continue to make effective progress against my aging problem, my behavior has had to stay changed long term. I have to take the anti-aging vitamins today and every day for the rest of my life. In short, I had to get motivated and stay motivated to take my unfelt-but-very-real aging problem seriously every day.
I had a professor in college who said that he always showed the mathematical equations in class because a small number of students "heard angels sing" when they saw the equations and immediately everything was clear to them. I am still waiting for those angels to start singing. Unlike Dad, I am not mathematically oriented, and I think mostly in pictures. We would be light-years back in our understanding of aging without Dad's brilliance for math and the intensive mathematical modeling in this book that underlies our current understanding of modern human aging. After spending a lot of time going through the technical details of this book and listening hard for the angels, allow me to share a non-technical mental motion picture that keeps me motivated to take my aging problem seriously.
First, a recap of my mental paradigm shift and an introduction to my mental motion picture.
It is a fairly natural thing to feel that aging is all about old people. As a young person, it is somewhat natural to wait to get serious about these anti-aging vitamins until the negative effects begin to be felt. However, as Dad and I have done our best to make clear in this book, this "natural" way of thinking is wrong. The most deadly of the aging diseases at present is Aging 2 disease. I think about Aging 2 as a tall waterfall with deadly rocks at the bottom. Without MePiA, our physiological boat goes over the falls around age 10–14 years. Once we are over the top of the falls and are headed towards the rocks at the bottom, it is no joke to attempt to stop the boat's deadly fall.
Unfortunately for me, even though I am young, my boat had already gone over the falls long before MePiA was discovered. Despite the appearance that all is well with my body, I know that I am headed for the rocks at the bottom of the falls. But I have hope that I may yet elude those rocks because I am taking the anti-aging vitamins, and as this book demonstrates, this makes healing of my Aging 2 disease possible.
Now we are ready for a more complete version of my (mostly) non-technical mental motion picture. (While this motion picture is focused on Aging 2, it is not meant to exclude Aging 0 and Aging 1 diseases, which obviously provide their own additional sources of motivation.)
At the start of the motion picture, I am not taking the anti-aging vitamins. As previously, Aging 2 is a tall waterfall, and my physiological boat is headed down the Aging 2 falls towards the rocks. If I hit the rocks, my life is over. My body's amazing natural repair mechanisms are represented by a powerful but very slow winch anchored at the top of the falls. The rope from the winch is tied to my falling boat. The power the winch needs to wind in the rope and pull my boat back up the falls is transferred from an electric motor to the winch via a belt and pulley system. When it was in relatively new condition, the winch successfully slowed my boat down when I first went over the falls. But the falling water has been so forceful that the belt is now worn and noticeably beginning to slip in the pulleys. The rope is being pulled out of the winch, allowing my physiological boat to keep falling toward the rocks faster and faster. All too soon I will be close enough to the rocks to realize that this is no amusement park ride and that I am in dire peril.
Enter MePiA. It turns out that there is a tensioner for the belt which may be tightened with a wrench. MePiA is the wrench.
Every day, when I supply my body with the MePiA it needs, the wrench adjusts the belt tensioner another tiny amount. Each tiny adjustment reduces the slipping of the belt, which slows down the rate at which the rushing water is able to pull the rope out of the winch. Eventually, as long as I don't hit the rocks first, the belt will actually be tight enough for the winch to start slowly pulling me back up out of the falls.
With all the spray and thunderous roar coming up from the rushing water hitting the rocks below, I don't know exactly how close I am to those rocks. I don't know exactly how long it will take for the belt to get tight enough to stop my fall towards the rocks and begin to pull my boat back up. I also don't know whether at any time a very negative random event might happen, like the rope or the belt breaking. What I do know is that I need to get out of the falls and back onto the placid lake above as quickly as I possibly can. Each day is a new opportunity to better my position—to enable that wrench to make another tiny adjustment and thus improve my chances of survival—by taking my anti-aging vitamins.
One of the challenges my wife Esther and I have had to face is how to use the information we have about the anti-aging vitamins with our kids. Often, for us, there was no precedent set by others to follow. We want what is best for our kids. The only question is, what is truly best for them?
Unfortunately, most children are not even able to make healthy breakfast cereal choices for themselves, and so we, as parents, end up making important nutritional choices for our kids every day. The anti-aging vitamins are no different, but they are relatively new and unfamiliar, and unfamiliar things are often intimidating.
As part of my 8-to-5 job, I have delved more deeply into questions regarding the safety of MePA and MePiA than probably any other person. As a research scientist with a background in physiology and nutrition, I conduct experiments supplementing the diets of laboratory animals with MePiA and MePA and personally evaluate the response over time. My job puts me in a better position than probably anyone else to know the truth about the safety of dietary supplementation with the anti-aging vitamins. My conclusion is that the evidence for safety is very, very strong. Meanwhile, the evidence for the large risks to health of not supplementing with the anti-aging vitamins has only grown over the last several years.
Eventually, out of a desire to do what is best for our kids, and out of concerns about the potential negative effects of our kids not receiving the anti-aging vitamins, my wife and I decided to begin supplementing the diets of our children with the anti-aging vitamins at low levels. We began with just MePA, before the discovery of MePiA. As my confidence in the safety data grew from strong to overwhelming, we gradually increased their supplementation rate to be on par with the current recommended daily intakes. When Aging 2 disease was discovered last year, my wife and I were very thankful that we had done this. When it comes to aging, a year of prevention is worth many years of healing.
Our experience has been what we fully expected; our kids are all profoundly normal. There has been no dramatic change in growth, development, or anything else. But, as they race around the house and yard with their mitochondria producing energy (and therefore free radicals) at full steam, we are content, knowing that they got their anti-aging vitamins at supper, and the free radicals are venting their fury on the sacrificial antioxidant MePiA, rather than on our children's mitochondrial DNA.
My wife, Esther, has had her own unique experience with the anti-aging vitamins. She had a history of more health challenges than I had, some going back to her teen years. She is also a loving mom who wants what is best for her kids. She has grappled with questions about supplementation with the anti-aging vitamins during pregnancy, birth, and lactation. She carefully sorted through the available evidence to make the best decisions she could for herself and her children at each stage, and her testimonial is key reading for any woman in the childbearing years of life who is taking the anti-aging vitamins.
I am in my late 20's, and I would describe my health as "very good," although I have a history of fatigue, low body weight, preterm labor, lower back trouble, and (currently self-diagnosed) asthma. I began supplementing with vitamin MePA during my fourth pregnancy and began to supplement with both anti-aging vitamins when MePiA became available between my fourth and fifth pregnancies. I believe the vitamins have contributed to improvement in my back as well as significant improvement in my energy levels, especially throughout pregnancy and postpartum periods.
I had three pregnancies and deliveries prior to beginning vitamin MePA. I will include the details of those in brief, as context of what my "typical" pregnancy experience was before starting the anti-aging vitamins.
Baby 1:
I was underweight before, during, and after my first pregnancy. I did not keep good notes during pregnancy and do not remember many specifics of symptoms I experienced. Baby 1 arrived at 36.5 weeks after a spontaneously occurring 20-hour labor. Baby was 5 lb 10 oz. There were no complications for baby or me during pregnancy, labor, delivery, or postpartum.
Baby 2:
Pregnancy started 9.5 months after Baby 1 was born. I was still underweight but not as much as before. I experienced clear SPD (symphysis pubis dysfunction) symptoms (symphysis pubis pain, nerve pain in inner legs, pelvic instability) as well as some backache throughout the later months of pregnancy. Baby 2 arrived at 39.5 weeks after a six-hour labor and weighed 7 lb 2 oz. There were no complications for baby or me during pregnancy, labor, delivery, or postpartum, although I struggled with low body weight in the months immediately following delivery.
Baby 3:
Pregnancy started 16 months after Baby 2 was born. I began pregnancy at a healthy weight and managed to stay at a healthy weight throughout pregnancy. I experienced onset of SPD symptoms at about 20 weeks, with symptoms flaring up to extreme pain toward the end of pregnancy. I also had more back pain and seemed to have slipped a disk (chiropractor diagnosed) at 30 weeks pregnant. My water spontaneously broke at 36 weeks and 6 days. After a 20-hour labor, Baby 3 arrived at 37 weeks exactly and weighed 6 lb 14 oz. No complications for me or baby. I was concerned that back pain, even severe pain, might be the norm for subsequent pregnancies.
Baby 4:
Pregnancy started 16 months after Baby 3 was born. I was able to keep a healthy weight during pregnancy. I began taking 1 microgram of MePA per day at 13 weeks and 3 days of my fourth pregnancy. I wanted to take MePA because of the evidence suggesting it might offer health benefits to me and my developing baby. I did not intentionally wait to take it until after the first trimester; it just happened that way. I chose this low level of MePA because, as far as I knew, I was the first woman to supplement with MePA during pregnancy, and it seemed wise to start at a low intake. In a nutshell, my pregnancy, birth, and breastfeeding experience was normal. A few small things improved.
After 6 weeks on MePA, I noticed the first signs of changes. I got sick with what I believe was the flu, but I had drastically reduced duration of symptoms and felt better the next day instead of worse. I have never experienced this before. The flu has always wiped me out for a minimum of three days. Even if it was a fluke, it was extremely anomalous for me. My SPD symptoms began around 16 wks of pregnancy.
I sought regular chiropractic care around 20 weeks to try to prevent these symptoms from getting terrible. My SPD never did get so bad in this pregnancy. I am not sure whether the improvement can be attributed to MePA or chiropractic care or other factors. I also did not experience back pain as I had in prior pregnancies.
Ultrasounds at 19 and 31 weeks found everything to be normal both times. I experienced typical pregnancy leg cramps, sciatica, Braxton Hicks contractions, and insomnia.
I discussed fears of preterm labor (due to my prior history) with my midwife at 30 weeks, and I began taking 300 mg magnesium glycinate as well as extra vitamin C at her recommendation. I noticed fewer Braxton Hicks contractions after beginning magnesium, indicating perhaps a latent magnesium deficiency.
Baby 4 arrived at 40 weeks and 3 days after several bouts of prodromal labor. Everything was profoundly normal. My labor was about 3.5 hours long. This was my biggest baby at 8 lb 5 oz, but my labor and delivery were the easiest of the four. Contractions never reached above an 8 out of 10 on a pain scale, and I never reached a point of "I can't do this anymore!" as I had with my three previous labors. There were no delivery or postpartum complications for baby or me. Breastfeeding initiated immediately after birth. I began taking 2 micrograms of MePA per day the night after delivery. Baby 4 has been seen for the normal wellness visits with a pediatrician. Baby 4 has been interactive and active, continues to grow and develop at a normal healthy rate, and is now a busy toddler.
I increased my MePA intake to 4 micrograms per day in early January 2019, about four months postpartum. By April 2019, I had increased my daily MePA intake to 8 micrograms.
In late July 2019, I began taking the combined MePA/MePiA supplement at 5 micrograms of each vitamin per day. I increased my intake to 8 micrograms of each per day in early November 2019. I increased again to 16 micrograms of each per day in late January 2020, shortly before I discovered I was four weeks pregnant.
Baby 5:
Pregnancy began about 16 months after Baby 4 was born. I began pregnancy at a healthy weight. I gained weight more easily this pregnancy and overshot my weight goal by about 15 lb by the end of the pregnancy. Morning sickness lasted longer and was more intense for me this time, beginning at 5 weeks and not going away until 20 weeks, whereas about 7 weeks of nausea was typical of my last three pregnancies. I do not know if this is due to Baby 5's sex being different from the sex of last three babies, but I think it is likely.
During this pregnancy, I experienced an increase in frequency and intensity of coughing attacks. It now seems almost certain that I have diagnosable asthma. My primary care doctor recommended taking diagnostic tests post-pregnancy, and I have yet to do that. Coughing and wheezing symptoms, brought on by specific triggers, date back to at least ten years ago, but they worsened some during this pregnancy. I am not certain why they worsened, but I obtained an inhaler for the first time at my doctor's prescription. Several episodes were alarming, but I have not had to use the inhaler to date. Symptoms have lessened since delivery, possibly from efforts to alleviate potential triggers in our home. My doctor didn't seem to think it was likely that my pregnancy was a big factor in the asthma symptoms, although the increase and decrease of symptoms do seem to line up with the duration of pregnancy.
Other than the increased nausea and asthma, this pregnancy was marvelously smooth. My mid-pregnancy ultrasound results were perfect, my test results all came back great, my typical pregnancy back pain did not bother me at all, and I experienced only relatively mild SPD symptoms even without chiropractic care. Toward the end of pregnancy, I did experience the standard heartburn, sciatica, insomnia, achiness, etc, of the third trimester.
I experienced a labor scare at 35.5 weeks. (Unfortunately, it seems to be "normal" for me to have bouts of consistent pre-labor contractions before term.) I received a steroid injection in case of preterm delivery and went on modified bed rest until term at 37 weeks. Baby held out and arrived safely at 38 weeks, weighing 7 lb 4 oz. After several days of irregular come-and-go prodromal labor contractions, my active labor was an intense 1 hour 50 minutes. There were no delivery or postpartum complications for baby or me.
My postpartum physical recovery proceeded phenomenally. Baby has been seen for normal wellness visits and is growing and developing very well. I increased my anti-aging vitamins daily intake to 20 micrograms a couple weeks after delivery to be in line with the current recommended daily intake for a breastfeeding woman.
I have chronically struggled with exhaustion and fatigue since about age 13. Even as an adult, I dragged through every day. If I did anything "big," like grocery shopping or scrubbing the kitchen floor, it would take a day or more of extra rest and sleep to recover my energy. I would feel physically drained, a bone-deep weariness that I could not shake. These symptoms only worsened when I was pregnant, until basic cooking and laundry were major daily hurdles. In the early months of my first pregnancy, I was sleeping probably 14–15 hours a day, and still I felt wiped out.
After about three months on MePA, while pregnant with Baby 4, I noticed an improvement in my sleep quality. I seemed to sleep much more deeply. I still needed a lot of sleep (ideally 9 hours at night and a 30-minute nap in the afternoon), but I actually felt refreshed when I woke up. I noticed, at the same time, an increase in energy and stamina. I could function on a half-hour nap a day instead of the normal hour plus that I historically had to have to survive. I didn't necessarily feel like I had an excess of energy at any given moment, and I still felt tired all the time, but I was more able to put in a long, hard day of work caring for my family, get up the next day, and do it again. I had enough energy to take it upon myself to do our family's grocery shopping (alone with kids in tow), and amazingly, I did not have to rest the entire next day just to recover my energy. At 35 weeks pregnant, my husband commented that he was very pleasantly surprised that I wasn't "downed" by the pregnancy as in the previous three pregnancies.
I noted a sleep change after about a month on 2 micrograms of MePA (postpartum with Baby 4). I seemed to experience 4–5 hours of unusually deep sleep between 11:00 PM and 4:00 AM. I also was able to go without a nap, or with less of a nap, much sooner than after my older children were born.
When I increased my MePA intake to 4 micrograms per day in early January 2019, about 4 months postpartum with Baby 4, I noticed a return to the deep sleep cycle I had experienced several months earlier.
After I began taking MePiA (in addition to MePA) at about 9 months postpartum with Baby 4, I felt able to get out of bed easily in the morning. Gone was the historical 10- to 60-minute-long struggle to talk myself into getting up and starting into my day. When I woke up, I felt renewed and refreshed instead of feeling as though I had run marathons all night long. My husband can corroborate that I have never been a morning person, but suddenly I was up before him, making him coffee and enjoying some quiet before the family awoke! And loving it! One morning I rose early and thought for the first time in over ten years, "Maybe I am a morning person after all."
That initial energetic exuberance faded some, but I have continued to experience much better energy levels and sleep quality than I had pre-vitamins. During my fifth pregnancy, my energy levels were, in general, surprisingly high. I felt energized most of the pregnancy and, especially in the second and early third trimesters, I felt very motivated to accomplish tasks, many of them large and demanding. My husband had to remind me to slow down at times, afraid that I would overexert myself.
During this most recent pregnancy and postpartum, I have been busier than ever before. I believe the vitamins likely have contributed significantly to my energy levels and ability to handle the many and varied needs in a given day. I sleep deeply when the baby is asleep at night. I do not usually find it difficult to get up in the morning after I wake up, and it does not typically take an extended period of time to feel fully awake in the morning. I was able to drop my midday nap within a matter of weeks after delivery. The bone-deep fatigue that characterized my earlier pregnancies did not characterize this one. Instead, I amazed myself and my husband by the many projects and tasks I accomplished while preparing for Baby 5.
The vitamins haven't been a total sleep/energy miracle cure for me. I have experienced a phase or two of an overabundance of energy. I have also experienced a phase or two of feeling as though I needed to sleep more than normal. I still get tired. I still sleep poorly at times. Energy and sleep are very complicated and affected by many factors. But overall, compared to my historical chronic state before supplementation with the anti-aging vitamins, I have seen dramatic, encouraging improvement in my ability to function in day-to-day life. Words don't do justice to the difference it has made to my personal life experience; life is so much more enjoyable when it doesn't take everything I have to do just the small things.
Lower back pain was chronic for me in my late teens. It recurred frequently after marriage and worsened during my early pregnancies. As mentioned previously, I slipped a disc in my lower back in late pregnancy with Baby 3 in mid 2016.
Before my fourth pregnancy began in late 2017, possibly dating back as far as slipping the disc, I began to feel a "clicking" in my lower back as two or three of my lumbar vertebrae shifted every time I rocked my back (i.e., bending over and standing up). This clicking continued throughout my fourth pregnancy and afterward, lessening in the spring of 2019, about 9 months postpartum with Baby 4, but returning in full force that summer. This was extremely discouraging, especially with busy small children to keep up with, as well as concerns about potential added stress of pregnancy on my spine. Surgery for lumbar instability is a slippery slope with a low rate of long-term success, as methods to reduce stress on one vertebra tend to increase stress on adjacent vertebrae, causing pain and instability again. The possibility of needing to pursue medical intervention was frightening.
Thankfully, this clicking seems to be healed today. I cannot remember when it went away—it is one of those things where one day you realize it has been that way for awhile—but I have not experienced the same shifting/clicking for at least a year (and a pregnancy). I cannot say with certainty that it will not return again, but I am very encouraged that it is not a daily concern anymore. I do not know if the vitamins had a role in this healing, but I think it is possible.
I did not experience any back pain while pregnant with Baby 4 and Baby 5, contrary to my earlier pregnancies.
While my experience is just that, an experience of one woman, I wanted to share it in the event that it may be useful to others. I believe that supplementation with the anti-aging vitamins has improved my sleep quality, energy, stamina, and skeletal health.
I (GEA) began digging into the cause of pre-Flood longevity in my spare time in my early twenties, in the late 1970's. By 2000, I had laid sufficient groundwork to mandate that I make finding vitamin X my full-time occupation.
I first identified MePA as a vitamin X candidate, after many false starts, in September of 2014. I began computer modeling work on MePA in June of the following year. By late November 2015, tests with mice had revealed no signs of toxicity, and the theoretical case for MePA had become sufficiently strong to warrant testing it on myself.
It seemed clear that the risks of not beginning to take this compound were at least as high as any risks which might possibly be associated with taking it. I was reasonably sure by that time that MePA had been naturally present in the environment and everybody's diets for time out of mind prior to its loss in the Flood five and a half thousand years ago. This meant that I did not need to worry about it being like a new synthetic compound, totally foreign to the body, with potentially disastrous consequences.
My daily intake—1 microgram/day—would be miniscule. It seemed unlikely that many even synthetic compounds, chosen at random, would be able to do a body much harm at that low intake rate. To get a feeling for the low risk associated with such a small intake, compare, for example, to the deadly synthetic compound sarin, "used as a chemical weapon due to its extreme potency as a nerve agent."[122] A single dose of 500 micrograms—still tiny, but 500 times larger than the intake of MePA I would be taking—administered to a healthy male volunteer caused only "mild symptoms of intoxication."[123]
Meanwhile, I was approaching 61 years of age. My body was making it increasingly clear that I had passed the plateau phase of the human survivorship curve and that I was rapidly on my way down its precipitous slope.
My own mortality had first begun to register with me when I had been forced to get prescription glasses early in my forties. I had always kept myself in reasonably good shape, without getting fanatical about it. I had never smoked, and I had stayed clear of alcohol. My wife and I had walked two miles or more together each day for decades, and I had found little trouble maintaining a healthy weight. But declining eyesight was just the beginning.
Subsequent years brought significant loss of hearing, hypothyroidism, worsening migraine headaches, and more. I had spent a great deal of time studying "old age." Now I was getting to experience first hand what I had studied.
The biggest age-related health problem struck just after I turned 50. It began with random pains, short in duration (one or two seconds) but intense, like being stabbed with a needle. I had these in a variety of locations, mostly extremities: thumb, big toe, etc. They seemed not to hit the same spot twice. I shrugged them off. Life was too short and its mysteries too absorbing to be distracted by petty annoyances.
Then, while raking gravel in the driveway, I found I was unable to maintain my grip on the rake handle. Peripheral weakness continued to spread over the following weeks, and then months. It would come and go, but each time it returned, it would be worse than before and stay longer.
After a couple of years of following false leads, I was finally diagnosed with CIDP (chronic inflammatory demyelinating polyneuropathy) by a talented neurologist. CIDP is a rare disease (a few per hundred thousand), most common in men over age 50.
CIDP is an autoimmune disease. The immune system attacks the myelin sheath surrounding nerves. The result is loss of nerve impulses to peripheral muscles, with ensuing weakness.
By the time I got the diagnosis, I was in very bad shape. I was having difficulty lifting my fork to feed myself, I could not button my shirt, I couldn't walk up or down stairs unassisted, and I was worrying at night about smothering under the blankets because I lacked the strength to move them off of my face.
While CIDP cannot be cured by modern medicine, its symptoms can generally be treated and controlled. Over the better part of the next decade, I went through several treatment regimens, from high prednisone to IV-Ig to Hizentra® home infusions, 60 ml twice per week. Relief of symptoms provided by treatment was not total, but it was nonetheless substantial, allowing me to live a somewhat normal life once again.
Eventually, I was able to resume walking two miles each day with my wife, Helen. But my leg muscles were no longer what they had been. They would rapidly grow weary and pretty much give out after the first mile. No amount of conditioning or exercise helped, so I began taking a bike with me. I would wheel it along beside me as I walked the first mile. It functioned as somewhat of a welcome "walker," keeping me more stable on my feet. Then I would ride it beside Helen as she walked the second mile.
I started taking MePA at 1 microgram/day on November 26, 2015. Three and a half weeks later, I was noticing positive health effects. Most significantly, I began to feel that I could do without the bike on our daily walks. I tried it and, sure enough, I could walk the entire two miles! CIDP had clearly begun to let up. This was eleven years after I had felt those first stabbing pains and subsequent peripheral weakness.
Another two weeks later, I was able to stop the biweekly infusions for CIDP that I had been obliged to be on for years. I had tried coming off the treatments in the past, only to be forced back on them if I wanted to stay out of a wheelchair. Had MePA healed my CIDP?
There were other possibilities. Spontaneous remittance is a characteristic of CIDP in the early years. But I was no longer in the early years. I had not experienced any spontaneous remittance since the relapse that nearly put me in a wheelchair prior to diagnosis. That had been nine years ago. One research study, aimed at assessing the long term prognosis of CIDP, concluded that prognosis "may be determined by the course and response to treatment in the first five years."[124] Based on the normal progression of this disease, remission seemed most unlikely. And the timing seemed just too coincidental. Remission had begun three and a half weeks following start of MePA dosing, after nothing but relentless disease for years.
Another possibility was the well-known placebo effect. But this, too, seemed pretty unlikely to me. Following eleven years of disease, I had expected to have CIDP for the rest of my life. I had always regarded it as possible that vitamin X might reverse CIDP, but I had not regarded this as probable. CIDP is an age-related disease. Age-related diseases are results of "aging." It was clear from the start that, in the process of rolling back "aging," vitamin X might also roll back some age-related diseases. In my case, that meant that it might roll back CIDP. But through the years, when Helen and I had discussed the possible impact on CIDP of the yet-to-be-discovered vitamin X, I had expressed my sincerely held doubts that it could impact diseases of the immune system. I had started on MePA in this frame of mind. My purpose had been to halt "aging." Remission of CIDP had come as a bit of a (pleasant) surprise.
What was needed, to know for sure what was going on, was a proper clinical trial involving both treated and control groups. But all I had was myself. So we waited. Neither placebo effect nor spontaneous remittance would be expected to last more than a few weeks.
It has now been over five years, and there has been no looking back.
I don't often think about CIDP since its cure. A few years back, while rummaging around on my computer, searching for a file from long ago, I bumped into a checklist I had drafted for Helen and myself for administering home infusions for my CIDP. Suddenly, it all came flooding back. The checklist was a full page long. The needed infusions took Helen and me several hours to carry out, twice each week.
The infusions were a net blessing, of course. They enabled me to have the strength I needed to button my shirt, feed myself, get out of a chair unassisted, navigate the two porch steps without peril of winding up in a broken heap at the bottom, and walk up to a mile unassisted.
But what a great blessing now, to have all this and more—I can even jog or run whenever I feel like it—with no infusions. It is wonderful not to have to plug those four hypodermic needles into my flesh twice each week. It is wonderful to do as I now please with the hours the infusions previously claimed each week. I am tremendously grateful that my CIDP has been cured, restoring me to a level of strength normal to healthy males in their sixties.
At the same time as CIDP was beginning to let up, I noticed that my skin was becoming more moist and supple. I'd had pretty severe eczema as a child, and my skin had tended to be dry and scaly into adulthood. The evident change for the better with my skin, together with the letting up of CIDP, got me really excited, because I knew that one of the symptoms of hyperthyroidism is oilier skin. I had been obliged to start taking levothyroxine for hypothyroidism early in my fifties. It seemed possible that MePA had begun to heal my thyroid, so that normal supplementation with levothyroxine was now causing hyperthyroidism.
I expected the next regularly scheduled blood test to come back showing high TSA. It came back normal and has remained normal. So MePA has made no discernible difference, so far, to my "worn-out" thyroid gland. Nonetheless, my skin has definitely improved.
I had always regarded a hot bath or shower as one of modern life's few affordable, worthwhile luxuries. I did some of my best thinking in the shower. But after age 50, I had found it increasingly necessary to stay out of the bath and out of the shower. The hot water would make my skin dry, rashy, and miserable the following day.
This too has now reversed. I am able to enjoy the luxury of a hot bath or shower pretty much as I please once again.
For a few weeks during the summer of 2018, my eczema flared up again, following many trouble-free months. This was confusing. Up until then, the vitamin seemed to have made eczema a thing of the past for me. Why had it returned, why in the summer when winters were always previously the worst months, and why had it returned with such stubborn ferocity?
Eventually, I was able to figure out that consumption of an abundance of fresh garden tomatoes was the culprit. When I eliminated tomatoes and citrus (both contain salicylates and amines, which trigger eczema in some individuals) the problem rapidly cleared up.
In hindsight, it seems clear that MePA allowed my skin to be sufficiently normal, for the first time in my life, for me to be able to figure out that I am allergic to tomatoes and possibly also to citrus.
Once I had begun to take MePA, I also found that I was sleeping better. It is easiest to describe the change which took place as simply a return to a more youthful sleep experience. Sleep was deeper and less interrupted. I needed less sleep, and I felt more rested.
Sleep still continues to be of higher quality than it was before the vitamin. I continue to need less sleep, giving me more usable hours in a day. I rise feeling ready to get to work. For the first few years after beginning to take MePA, I would sleep soundly for seven to seven and a half hours, waking once to go to the bathroom. Then the frequency of early morning insomnia began to increase. This seemed to result from sleep having been so sound and refreshing in the first half of the night that the need for sleep was substantially diminished in the second half of the night. Being doubtful that a mere four or five hours of sleep per night could be sufficient, I deliberately kept myself as somnolent as possible when I got up at night, by, for example, using a dim night light in the bathroom, and refusing to let my thoughts focus on anything disturbing or absorbing.
More recently, I have found that a routine of office work in the mornings (when my mental faculties are at their best) and physical activity in the afternoons greatly reduces early morning insomnia. A final helpful discovery has been that early morning insomnia is markedly reduced by a personal ban on the news media in general. In hindsight, this makes sense. The news industry is profit driven. Profits are highest whenever listeners are kept anxious over what might happen next so they keep coming back to hear more. Anxiety fuels insomnia. I tune the industry out and am rewarded with full nights of deep sleep.
In sharp contrast to pre-vitamin days, when insomnia now occurs, it does not leave me exhausted and unable to get much done the next day. It is as if the half-night's sleep I did get was enough to see me satisfactorily through the day. And I have found that I frequently sleep the whole next night soundly and without difficulty.
The knowledge that I can now get by for a day on very little sleep, and that I will make up for lost sleep the next night, reduces frustration and apprehension if I find myself awake in the early morning hours when I feel I should normally be asleep.
My experience with headaches and migraines stands in stark contrast to what I was experiencing immediately prior to beginning to supplement MePA.
When I was young, headaches were few, and I could count on a night's sleep to cure a headache completely. As the years passed, I became increasingly susceptible to headaches, and my body became less and less able to cope with them. If I went to sleep with a headache, for example, it would still be with me when I awoke the next morning.
My earliest recollection of a severe headache goes back to my mid thirties. By my mid forties, I was experiencing occasional migraines. All of this was compounded in my mid fifties by treatments for CIDP, of which headaches are a known side effect. Immediately before I started on MePA, when I was nearly 61, I was subject to headaches on a fairly regular basis. These would develop into full-blown, excruciatingly painful migraines if I failed to intervene.
I had learned to take action against headaches as soon as I noticed one beginning. I would take two extra-strength, over-the-counter pain relievers to knock out the headache before it had a chance to get going. The frequency of this corrective/preventive action had increased to an average of roughly once per two weeks. I kept a supply of pain pills in my office, in my vehicle, and near my bed.
This all changed for the better when I began supplementing my diet with vitamin MePA. The frequency of headaches rapidly declined. At present, the most accurate description of my headache frequency is "almost never."
Common upper respiratory infections rapidly reduced in frequency and severity once supplementation with MePA began. The same is true of the stomach flu. I had been living in increasing dread of these common ailments prior to MePA. I was beginning to understand how elderly persons might die of them. I am now back to taking upper respiratory ailments pretty much in stride. I can still catch a full-blown cold or flu and have it take a week or ten days to clear out, but this is relatively rare. More common is to get just a touch of the cold or flu, with symptoms totally resolving in under 24 hours. In the several instances of exposure to stomach flu which I experienced after starting on MePA, only brief, slight nausea resulted.
Coming off of twice-per-week 60 ml infusions of Hizentra® for CIDP complicated the question of what changes were due to MePA.For this reason, in the early years, before we had testimonials from any other users of MePA, the experience of my wife, Helen, was especially helpful.
Helen began taking 1 microgram MePA/day after I had been taking it for a year with no adverse side effects. She had lived close up with my CIDP for enough years to know that something real had happened. And she was concerned for her own future health, being aware from my research that MePA would not suddenly erase her own rapidly accumulating "old age" symptoms and debilitations.
Three weeks later, she began reporting greatly improved sleep. Previously, she had been experiencing chronic sleep trouble. A typical morning had begun with, "I slept so terribly again last night, I don't know how I can keep up, feeling like this…" Now she was saying, "I haven't slept like this in years." Our grown children began to comment on her remarkably improved ability to cope with life when they came home to visit. She would reply, "Yes, what a difference a good night's sleep makes!"
Helen also corroborated the increased skin oils. In her case, the change was not so welcome as it has been for me. Her skin has always been pretty normal—definitely youthful compared to mine. The practical impact for her was that she had to begin washing her hair three times per week instead of two.
Another improvement is that we seem to heal more quickly. For example, I developed very sore muscles in my back between my shoulder blades, and opposite that on my front side, from a bunch of heavy lifting I had to do, on top of some unusual hand pulling of plants I had been obliged to tackle in the garden. The pain was of the sort which takes your breath away. I took nothing for the pain, and I did not treat the sore muscles in any way, which is pretty standard practice for me. I went to bed expecting to lose a lot of sleep due to pain whenever I rolled over. From years of previous experience, I expected at least three days of pain, followed by a week or two of slow healing of residual soreness. What happened is that I slept better than expected, and I woke up with only slight twinges of pain left. By that afternoon, even those were gone.
As another example, I accidentally mashed the end of a finger between two steel pipes. The right half of the fingernail rapidly turned dark purple. It was pretty painful all that day. I wondered whether the purple half of the nail might separate from the finger. But the next day, only a thin purple line remained under the nail, and there was only slight residual pain. The following day, it was difficult to distinguish the finger I had injured from neighboring fingers.
The blessing of rapid healing also shows up repeatedly after a day of strenuous physical work or other unaccustomed physical activity. It was common, prior to the vitamin, to wake up the next morning with aches and pains. These would typically take several days to resolve. Now, when I wake up the next morning, there are no aches and pains.
I do not want to give a wrong impression. It is still possible for some injuries to take time to heal. As an example, about half a year ago, I had a bout of pain in my right hip, causing me to walk with a limp. This was caused by dragging a number of heavy objects across the ground a considerable distance. I seemed to have injured some tissue in my hip joint in the process. The pain would be aggravated by climbing stairs or by peddling a bike. A deep squat would set things right for a while, relieving the pain. This particular injury took a full six weeks to heal.
It had no sooner gotten better than my left hip started acting up. This resulted from repeated trips up and down a long flight of stairs carrying some heavy loads. I thought I was in for another six weeks of pain and limping, but it resolved in a little over a week.
The norm has shifted from slow healing all my life before the vitamin to rapid healing since starting to take the vitamin, making the six-week hip episode to be noteworthy as a rare exception.
I made my weight a personal priority a year or so after starting on MePA. Proper weight pays back well for all the discipline it costs—health risks due to excess weight are well known.
I went to the Internet to learn my healthy weight range and my "ideal" weight. This gave me a target of 150 to 155 pounds, at the center of my healthy weight range. I was in the low 170's when I started. I used a calorie counter on my computer to count calories every meal. I adjusted my daily calorie allotment to lose just a half pound per week—I did not want to feel hungry (it is too distracting when I am working), and the anti-aging vitamins promised a lengthy future, so there was no rush. I soon settled comfortably into my 150- to 155-pound target range and shortly thereafter stopped using the calorie counter. This has resulted in staying around 157 to 158, which I am content with.
I had tried, unsuccessfully, to lose weight prior to starting on vitamin MePA. I credit the vitamin, not with making it easier to lose weight, but with providing the mental energy, resolve, and fortitude needed to make the slow transition to a more healthy way of eating.
Helen had suffered from arthritis for years. At present, she is pretty much free of this disease. We tend to forget what an increasingly nagging nuisance it once was.
Arthritis resolves relatively slowly with MePA. In mid 2017, after having been on vitamin MePA at 1 microgram per day for half a year, Helen had noticed no relief from arthritis. But by late 2018, two years and a month after beginning MePA, and having increased her daily intake to 8 micrograms per day, she was reporting significant improvement:[125]
I used to have constant hip pain [due to arthritis] on my left side, with it being worse at night. The pain is now completely gone, even though I'm on my feet much of the day.The swollen arthritis in my hands has gone down quite a bit, and I no longer have pain in my hands. Several of my fingers have straightened out now that the swelling has gone down.
The experience of my older sister, Valorie, another early user of MePA, confirmed that healing of arthritis is relatively slow.[126]
The arthritis in my joints cleared up after ten months of 2 micrograms per day of MePA.
Also, my knees were starting to have problems, which I figured would lead to knee surgery eventually. That has also entirely cleared up.
Helen struggled on and off for several years with neck pain which seemed to be due to arthritis, and also with bursitis for many years. The bursitis took the longest time to resolve after beginning to take MePA. It was still acting up two years later. But at present, another two years later, neither arthritis nor bursitis has been a problem for a very long time.
Helen is now supplementing both MePA and MePiA at the current recommended daily intake for adult females of 15 micrograms each per day. It seems likely that her arthritis and bursitis symptoms would have resolved sooner had she been supplementing at this higher intake level, but, in the early days, we were just beginning to learn about the optimum daily intake level of MePA.
Here is more of Helen's experience, shared in late 2018:[127]
The most noticeable change for me has been my energy level. I have always had high energy, but now I seem to have super high energy. I work typically 12 to 14 hours each day in our home businesses, going at a pretty high speed all day until late evening, when I drop into bed exhausted and sleep soundly.
I notice that I can multitask at a much higher level than before the vitamin. I am busier than I have ever been in my life, which is saying something for a woman who has raised and home schooled ten children. I am wearing many hats, yet I seem to be able to handle the stress of piles of work on my desk just fine. Before the vitamin, I frequently exclaimed (mostly to myself), "I can't DO all of this!" Now, I am able to realize that what doesn't get done today will get done tomorrow. I was never like that; I had to get my desk cleared off every day, or I was stressed—which meant I was often stressed!
The vitamin reduces work-related anxiety. I have much less anxiety now even when in high stress situations. Before the vitamin, when there was a large stress load, I would stay awake for hours at night, sometimes for several nights, "playing the tapes" over and over in my mind. Now I may lose a few hours of sleep, but I am able eventually to relax and go to sleep even though the stress level hasn't reduced. Even when the workload is extremely heavy, I am able to handle it just fine.
It is hard to describe the feeling I have of psychological wellness—the feeling of being able to handle whatever life throws at you. As you can imagine, with ten children and 26 grands plus several growing businesses, I feel like I get thrown the occasional hard and fast curve ball. I am now able to somehow put things in proper perspective and move on much quicker than I had been able to before the vitamin.
I am able to handle company much more now, and even welcome it! I enjoyed having company when I was younger, but in the years just prior to the vitamin, I was dreading having company more and more, even family, and I tried to get out of having company as much as I could.
Thanksgiving 2018 had me decorating, cooking, cleaning, organizing a large children's treasure hunt, door prizes, and the like with great enthusiasm despite the heavy workload due to growth in our various home businesses. I'm very excited about Christmas 2018 and am already decorating. (In the past I would normally not decorate until a few days before Christmas, and it would usually be minimal.) This year, I am having a family open house in our home for several events over the holidays.
I can get away with less sleep, feeling ready to get up after about 5–6 hours of heavy sleep. I find that I'm dreaming a lot more, especially in the early hours of the morning. Even if I have a bad night's sleep, for whatever reason, I'm still able to work hard and generally have no need for an afternoon nap.
The age spots on the left side of my face are almost gone, and the other aging spots on my face are fading slowly.
I am still having to wash my hair about every 2 to 3 days as it is much oiler than before the vitamin.
My eyesight seems the same; I still need glasses, sadly.
My eyebrows and hair have thickened. I was always losing hair before taking the vitamin. It is now thicker, like it was when I was a much younger woman. My gray hair is still there. My hair doesn't seem to be getting grayer, but the amount of gray is a hard thing to measure.
I notice that if I get injured in some way, like a cut on my hand, or a bruise from falling, then healing is rapid and almost unreal. The next day, after the injury, it seems totally healed.
The truth is, I have always felt younger than my age. But now, at 64, I feel like I'm in my 20's.
For example, I have always wanted to have a straight (pencil) jean skirt. I had been watching for one in the thrift store for a year or so and finally found one. I was so excited and could hardly wait to wear it. I put it on one morning, and got to work in the shipping room packing boxes for mail orders. But it was just not practical—one cannot run in a straight skirt!
Even though I am a grandmother many times over, I don't spend much time on the porch in a rocking chair!
From time to time, Gerald comments, "You're a new woman." He's right!
On the "no improvement" side, my thyroid dysfunction persists unchanged, and, like Helen, I have noticed no regression of gray hairs and no improvement in my eyesight. But then, based on the slow healing rate of Aging 2, Helen and I are not expecting to see rapid improvement in everything. We expect to be stuck in our physiological sixties for a long time to come.
But we are happy to have begun the journey back up out of the aging diseases, and we are already feeling much improved. Our resilience is much increased and our quality of life is simply better than it was for both of us before we first began dietary supplementation with MePA. We now have to keep reminding ourselves not to overdo it—that we are still in our physiological sixties.
The foregoing testimonials may make MePA seem like somewhat of a miracle drug. It is not. It is simply a vitamin. In actual practice, the real-world experience with beginning to take the anti-aging vitamins for those 40 years of age and older is likely to be a relatively rapid resolution of a variety of health issues caused by Aging 0, followed by little noticeable health improvement and possibly some slow continuing decline for a few years due to inertia of the mtDNA mutation mess previously induced by Aging 2, followed by a slow trend toward health improvement as cells with mtDNA mutations very slowly get replaced by healthy cells. The anti-aging vitamins are merely vitamins, not miracle drugs. But, to those with advanced scurvy, vitamin C probably seems like a miracle drug too.
The world is a very sick place at present. No doubt others have said this same thing in the past, but none has ever meant it quite as literally as we do. The global population is sick and is dying for want of two previously unknown vitamins. The cure is now in hand. Human health and longevity may be restored globally by the simple expedient of inclusion in the human diet of micrograms per day of the long-lost anti-aging vitamins, methylphosphinic acid (MePiA) and methylphosphonic acid (MePA).[128]

The program listing below shows how the biblical life expectancy data were modeled based on vitamin MePiA produced by atmospheric chemistry from MeP precursor gas generated in anaerobic sediments and delivered to the atmosphere via the oceans.
The code is in free-format Fortran 95. It was compiled as a 64-bit executable using GNU Fortran 6.1.0 via Simply Fortran by Approximatrix. The executable was run in a Windows 10 Command Prompt window on a PC having an Intel Pentium G3258 CPU.
program MeP_20170105
!
! MeP_20170105 seeks to model the biblical life expectancy data based on the
!precursor gas MeP (methylphosphine).
!It uses a grid search to find a minimum of the chisqr hypersurface.
! It allows the width of the Spike interval to be easily adjusted, to optimize
!this discrete integer parameter.
! The results are saved in the output file MeP.txt for plotting in the
!spreadsheet MeP.xls.
!
! Units are everywhere mka (meters, kilograms, years).
!
include "MeP_20170105.inc"
!
! Input biblical life span data; see MeP.xls for the source of these data.
! The life spans of the first 7 pre-Flood patriarchs are averaged separately.
! They are not part of the data being fit by the program and they are not
!included in the following data statements.
!
DATA jb /-4120, -3617, -3517, -3482, -3452, -3418, -3388, -3356, -3297, -3167, -3081, -3067, -3007, -2943, -2916, -2806, -2668, -2530, -2527/
DATA LEb /950, 600, 438, 433, 464, 239, 239, 230, 205, 175, 137, 180, 147, 137, 110, 133, 137, 123, 120/
i_pts = 19
!
! Compute standard deviations of life expectancy at birth.
! The mean and standard deviation from pre-Flood data points (excluding Noah)
!is 926 +/- 28.9.
! The mean and standard deviation from modern data for U.S. males
![https://www.ssa.gov/oact/STATS/table4c6.html] is 76.8 +/- 16.8.
!Interpolate between these two points.
do 5 i=1, i_pts
sigma(i) = ( sigma_LE_0 - sigma_LE_now ) / ( LE_0 - LE_now ) * ( LEb(i) - LE_now ) + sigma_LE_now
5continue
!
! * * * FIRST, WORK OUT MePiA IN DRINKING WATER * * *
!
! Model SC(i) from before the Flood to the present by breaking the timescale into
!four time intervals.
!The first is the pre-Flood interval. The second is the Spike interval. The third
!is the Decline interval. The fourth is the Modern interval.
! _ _ _ PRE-FLOOD _ _ _
! The pre-Flood extends from the distant past to the Flood in 3520 B.C.
!C_0 is the pre-Flood concentration of MePiA in drinking water.
!It is assumed to be constant.
!All MePiA concentrations in drinking water are normalized with respect to
!it, yielding the specific concentration: SC(t) = C(t)/C_0
!C(t) is the time-varying concentration, equal to C_0 pre-Flood.
!
! Set SC to 1 pre-Flood.
do 10 i = i_start_date, i_Flood_date
SC(i) = 1.0D+0
10continue
!
! _ _ _ SPIKE _ _ _
!The Spike is bounded by the Flood on the left and by the plummeting of life
!spans between Eber (born -3452) and Peleg (born -3418) on the right.
!The Flood happened 3520 B.C., so t_Flood = -3520.
!Thus, the Spike is 3520 - ( 3452 + 3418 ) / 2 = 85 +/- 17 years long.
!SC must exceed 1 at the start of the Spike.
!This results from the southern oceanic floor depressurizing.
! This would have released MeP to the atmosphere from the phosphorus-rich ocean
!floor sediments surrounding Antarctica.
!SC_Spike is the value SC jumped up to following the Flood.
! Theory says it will be very large.
! The model is insensitive to this parameter, as long as it is large, because
!of X_max.
! So it is treated as a constant rather than as a free parameter.
! Excess MeP was steadily removed from the polluted Spike atmosphere by conversion
!of MeP to MePiA.
! At the end of the Spike interval, excess MeP had been entirely removed from the
!atmosphere.
! Thus, the Spike ends 3520 - 85 = 3435 B.C. (+i_delta_Spike)
!_
!_ if (t .gt. i_Flood_date .and. t .le. i_last_Spike_date) SC(t) = SC_Spike
!_
do 11 i = i_Flood_date + 1, i_last_Spike_date + i_delta_Spike
SC(i) = SC_Spike
11continue
! _ _ _ DECLINE _ _ _
! Following the Spike, MeP continued to be sourced to the atmosphere from the
!oceanic reservoir of MeP until the oceanic reservoir was exhausted.
! This defines the Decline time interval.
! It is bounded by the end of the Spike on the left and by a second, smaller
!plummeting of life spans following the birth of Moses.
! Moses observed, in Psalm 90, that life spans had declined to 75 years
!during his life span.
! Moses lived to 120 years.
! A person dying of old age at 75 when Moses was 120 would have been born when
!Moses was 45 years old.
! Thus, this is the latest possible date at which MePiA in drinking water had
!dropped to zero.
! The year following the birth of Moses is the earliest MePiA could have dropped
!to zero.
! Take the midpoint (when Moses was 23) as the best estimate of when MePiA
!actually dropped to zero.
! Moses was born 2527 B.C., so this interval ends 2504 B.C.
! The inventory of MeP in the oceans would probably have been reduced by the
!Flood relative to the pre-Flood inventory.
! So SC would fall to some value less than 1 at the end of the Spike (and start
!of the Decline).
!SC_ocean is the initial value, immediately following the Flood. It is a
!FREE PARAMETER of the model.
! The ocean acts as an elevator, carrying MeP from the deep ocean up
!ultimately to the surface where it vents to the atmosphere.
! MeP is expected to have been lost (due, for example, to having been eaten by
!microbes) on its way to the surface.
! Model this loss as a decaying exponential.
! lambda_ocean is the decay constant for MeP loss within the ocean. It is a
!FREE PARAMETER of the model.
!_
!_ delta_t = t - i_Flood_date
!_ if (t .gt. i_last_Spike_date .and. t .le. i_Moses_Drop_date) SC(t) = SC_ocean *
!exp(-lambda_ocean * delta_t)
!_
! _ _ _ MODERN _ _ _
! The Modern time interval goes from the end of the Decline to the present.
! SC(i) is taken to be zero during this interval.
!_
!_ if (t .gt. i_Moses_Drop_date) SC(t) = 0.0D+0
!_
do 12 i = i_Moses_Drop_date + 1, i_end_date
SC(i) = 0.0D+0
12continue
!
! * * * NOW WORK OUT MePiA IN THE BODY AND CALCULATE AGEDNESS AND LIFE EXPECTANCY
!AT BIRTH * * *
!
! MePiA intake is assumed to be proportional to its concentration in drinking water.
!delta_X is the annual intake of MePiA.
!Thus delta_X = constant_2 * SC(j).
!X is the total pool of MePiA in the body.
!It is unknown, so normalize it with respect to the pre-Flood pool, X_0.
!This means that it starts out pre-Flood with a value of 1.
!
!tau_X is the mean life of MePiA in the body. It is a FREE PARAMETER of
!the model.
!
! In the pre-Flood period, SC(j) = 1.
! So constant_2 = delta_X pre-Flood.
! Meanwhile, X pre-Flood = 1.
! So 1 = exp(-lambda_X) + delta_X, from the general equation
!X = X * exp(-lambda_X) + delta_X.
! Thus, delta_X = 1 - exp(-lambda_X).
!emlx = exp(-lambda_X)
!constant_2 = 1.0D+0 - emlx
!
!Thus X = X_previous_year * exp(-lambda_X) + delta_X, where delta_t has been
!taken to be 1 year.
!
!X_max is the maximum size the MePiA pool in the body can be. It is a FREE
!PARAMETER of the model.
!
!Finally, X_birth, the size of the MePiA pool at birth is needed.
!This will be approximate—the baby will grow and hence deplete
!the initial pool more rapidly than lambda_X would suggest.
!I think the best simple approximation results from taking the initial
!pool size proportional to the concentration in drinking water, SC(t).
!This boils down to setting X_birth = SC.
!For example, in pre-Flood steady state, X_birth should clearly
!be 1, and this corresponds to SC = 1.
!
! * * * NOW SEARCH FOR A MINIMUM REDUCED CHISQR * * *
!Do this by a grid search of the hypersurface centered on the starting point.
!Calculate step sizes for the grid search.
lambda_ocean_start = lambda_ocean_0
tau_X_start = tau_X_0
SC_ocean_start = SC_ocean_0
X_max_start = X_max_0
!
degrees_of_freedom = i_pts - i_free
!
15chisqr_keep = 1000.0D+0
!
delta_lambda_ocean = lambda_ocean_start / sf
delta_tau_X = tau_X_start / sf
delta_SC_ocean = SC_ocean_start / sf
delta_X_max = X_max_start / sf
!
loops_total = ( 2 * i_steps + 1 ) ** i_free
loops = 0.0D+0
!
!Modify tau_X.
!
do 50 i50 = -i_steps, i_steps
tau_X = tau_X_start + i50 * delta_tau_X
lambda_X = 1.0D+0 / tau_X
emlx = exp(-lambda_X)
constant_2 = 1.0D+0 - emlx
!
!Modify lambda_ocean.
!
do 40 i40 = -i_steps, i_steps
lambda_ocean = lambda_ocean_start + i40 * delta_lambda_ocean
!
!Modify SC_ocean.
!
do 30 i30 = -i_steps, i_steps
SC_ocean = SC_ocean_start + i30 * delta_SC_ocean
!
!Modify X_max.
!
do 20 i20 = -i_steps, i_steps
X_max = X_max_start + i20 * delta_X_max
write(*,fmt='(a, f8.2, f12.5,a)', advance='no') 'percent complete, chisqr = ', loops/loops_total*100.0D+0, chisqr_keep, achar(13)
chisqr_1 = chisqr()
loops = loops + 1
if ( chisqr_1 .ge. chisqr_keep ) goto 20
chisqr_keep = chisqr_1
tau_X_keep = tau_X
lambda_ocean_keep = lambda_ocean
SC_ocean_keep = SC_ocean
X_max_keep = X_max
i_tau_X_keep = i50
i_lambda_ocean_keep = i40
i_SC_ocean_keep = i30
i_X_max_keep = i20
itotal = abs(i20) + abs(i30) + abs(i40) + abs(i50)
!
20continue
30continue
40continue
50continue
!
!Set all parameters to the minimum chisqr point found.
!
tau_X = tau_X_keep
lambda_X = 1.0D+0 / tau_X
emlx = exp(-lambda_X)
constant_2 = 1.0D+0 - emlx
lambda_ocean = lambda_ocean_keep
SC_ocean = SC_ocean_keep
X_max = X_max_keep
chisqr_keep = chisqr()
write(*,fmt='(a, f8.2, f12.5)') 'percent complete, chisqr = ', loops/loops_total * 100.0D+0, chisqr_keep
!
!Save results for plotting.
!
open(unit=12, file="MeP.txt")
do 60 i = -5200, -2000
write(12,fmt='(i12,3e12.5)') i, SC(i), LE(i), PA(i)
60continue
close(12)
if (i_steps .eq. 0) goto 70
!
! Repeat if different point found.
! Same point found is a necessary condition to avoid division by zero when
!estimating standard deviations below.
!
if ( itotal .ne. 0 ) then
lambda_ocean_start = lambda_ocean_keep
tau_X_start = tau_X_keep
SC_ocean_start = SC_ocean_keep
X_max_start = X_max_keep
goto 15
endif
!
! Now estimate the standard deviations in the fitted values of the free parameters.
!
!Estimate the standard deviation of lambda_ocean.
!
lambda_ocean = lambda_ocean_keep + delta_lambda_ocean
chisqr_plus = chisqr()
lambda_ocean = lambda_ocean_keep - delta_lambda_ocean
chisqr_minus = chisqr()
lambda_ocean = lambda_ocean_keep
sigma_lambda_ocean = delta_lambda_ocean * sqrt ( 2.0D+0 / (chisqr_plus + chisqr_minus - 2.0D+0 * chisqr_keep) )
print *
print *,'i, lambda_ocean, sigma, pe = ', i_lambda_ocean_keep, lambda_ocean_keep, '+/-', sigma_lambda_ocean, sigma_lambda_ocean/lambda_ocean_keep*100.0D+0
!
!Estimate the standard deviation of SC_ocean.
!
SC_ocean = SC_ocean_keep + delta_SC_ocean
chisqr_plus = chisqr()
SC_ocean = SC_ocean_keep - delta_SC_ocean
chisqr_minus = chisqr()
SC_ocean = SC_ocean_keep
sigma_SC_ocean = delta_SC_ocean * sqrt ( 2.0D+0 / (chisqr_plus + chisqr_minus - 2.0D+0 * chisqr_keep) )
print *
print *,'i, SC_ocean, sigma, pe = ', i_SC_ocean_keep, SC_ocean_keep, '+/-',
sigma_SC_ocean, sigma_SC_ocean/SC_ocean_keep*100.0D+0
!
!Estimate the standard deviation of tau_X.
!
tau_X = tau_X_keep + delta_tau_X
lambda_X = 1.0D+0 / tau_X
emlx = exp(-lambda_X)
constant_2 = 1.0D+0 - emlx
chisqr_plus = chisqr()
tau_X = tau_X_keep - delta_tau_X
lambda_X = 1.0D+0 / tau_X
emlx = exp(-lambda_X)
constant_2 = 1.0D+0 - emlx
chisqr_minus = chisqr()
tau_X = tau_X_keep
lambda_X = 1.0D+0 / tau_X
emlx = exp(-lambda_X)
constant_2 = 1.0D+0 - emlx
sigma_tau_X = delta_tau_X * sqrt ( 2.0D+0 / (chisqr_plus + chisqr_minus - 2.0D+0 * chisqr_keep) )
print *
print *,'i, tau_X, sigma, pe = ', i_tau_X_keep, tau_X_keep, '+/-', sigma_tau_X,
sigma_tau_X/tau_X_keep*100.0D+0
!
!Estimate the standard deviation of X_max.
!
X_max = X_max_keep + delta_X_max
chisqr_plus = chisqr()
X_max = X_max_keep - delta_X_max
chisqr_minus = chisqr()
X_max = X_max_keep
sigma_X_max = delta_X_max * sqrt ( 2.0D+0 / (chisqr_plus + chisqr_minus - 2.0D+0 * chisqr_keep) )
print *
print *,'i, X_max, sigma, pe = ', i_X_max_keep, X_max_keep, '+/-', sigma_X_max,
sigma_X_max/X_max_keep*100.0D+0
!
! All done.
!
! Ring bell to signal completion of run.
70write(*,*)'Ringing bell. Use ctrl-c to end program.'
80write(*,100,advance='no') achar(07)
100format(a)
call sleep(10)
goto 80
end
!
!*********************************************************
! * * * FUNCTION CHISQR * * *
!
function chisqr()
include "MeP_20170105.inc"
PA = 0.0D+0
!_
!_ delta_t = t - (i_Flood_date + 1)
!_ if (t .gt. i_last_Spike_date .and. t .le i_Moses_Drop_date) SC(t) = SC_ocean *
!exp(-lambda_ocean * delta_t)
!_
do 20 i = i_last_Spike_date + i_delta_Spike + 1, i_Moses_Drop_date
delta_t = i - (i_Flood_date + 1)
SC(i) = SC_ocean * exp(-lambda_ocean * delta_t)
20continue
!
! This is the main calculational loop for life expectancy
do 30 i = i_start_date, i_end_date - 500
j = i
!Agedness at birth is zero.
agedness = 0.0D+0
!MePiA body pool at birth.
X = SC(j)
! Time-changing annual intake of MePiA.
25delta_X = constant_2 * SC(j)
! Time-changing body pool size.
X = X * emlx + delta_X! delta_t = 1 year.
if (X .gt. X_max) X = X_max
! Time-changing agedness.
agedness = agedness + m * X + b
if (agedness .lt. 0.0D+0) agedness = 0.0D+0
j = j + 1
! Save physiological agedness of chosen individual; birthdate of individual is
!specified in if statement below
if (i .eq. -4120) PA(j) = agedness * 76.8D+0 ! Noah = -4120, Shem = -3617
if (agedness .lt. 1.0D+0) goto 25
LE(i) = j - i
30continue
!
! Calculate the reduced chisqr
!
chisqr = 0.0D+0
do 40 i = 1, i_pts
! yfit = LE(jb(i))
chisqr = chisqr + ( ( LE(jb(i)) - LEb(i) ) / sigma(i) ) **2
40continue
chisqr = chisqr / degrees_of_freedom
end
!
!*********************************************************
! This is MeP_20170105.inc
!
! real*8 is double precision.
implicit real*8 (a-h,l-z)
!
! User-specified parameters.
!
! Date parameters.
parameter (i_start_date = -5200)
parameter (i_Flood_date = -3520)
parameter (i_last_Spike_date = -3435)
parameter (i_Moses_Drop_date = -2504)
parameter (i_end_date = -1500)
!
! Grid parameters.
parameter (i_steps = 5)! number of grid steps to search out to on either
!side of the starting point; enter zero to do just the starting point
parameter (sf = 1000.0D+0)! scale factor for grid search; divides the start
!point value to give grid spacing for that free parameter
!
! Model parameters.
parameter (SC_Spike = 30.0D+0) ! NOT a free parameter
parameter (i_delta_Spike = -16) ! NOT a free parameter; allows duration of Spike
!to be adjusted; duration = 85 + i_delta_Spike
!
parameter (i_free = 4)! number of free parameters in the model
!starting point for grid search of the free parameter space (followed by
!parameter uncertainties from grid search)
parameter (lambda_ocean_0 = 8.6295194193469055D-4)! +/- 0.072523784375094215
parameter (SC_ocean_0 = 0.83802824227362249D+0)! +/- 0.0061298609207415975
parameter (tau_X_0 = 194.26218274932114D+0)! +/- 4.2427546379676411
parameter (X_max_0 = 1.1831834963273076D+0)! +/- 0.018412002518696499
!
! Life expectancy parameters.
!The mean and standard deviation from pre-Flood data points (excluding Noah)
!is 926 +/- 28.9.
parameter (LE_0 = 926.0D+0)! average pre-Flood life expectancy at birth
parameter (sigma_LE_0 = 28.9D+0)! uncertainty in the average pre-Flood life
!expectancy at birth
! The mean and standard deviation from modern data for U.S. males
![https://www.ssa.gov/oact/STATS/table4c6.html] is 76.8 +/- 16.8.
parameter (LE_now = 76.8D+0)! average modern life expectancy at birth for
!U.S. males
parameter (sigma_LE_now = 16.8D+0)! uncertainty in the average modern life
!expectancy at birth
!
parameter (b = 1.0D+0 / LE_now)! intercept for rate of aging line
parameter (m = 1.0D+0 / LE_0 - b)! slope for rate of aging line
!
! The blank common block is used to define a global variable storage area.
! Program constants and variables (except integers, which are at the very bottom)
common // constant_2
common // degrees_of_freedom
common // emlx
common // lambda_X
common // SC_ocean
common // lambda_ocean
common // tau_X
common // X_max
!
! Program arrays. Integer arrays come last.
! Real arrays
common // LE! fitted life expectancies
real*8 LE(i_start_date:i_end_date)
common // LEb! biblical and modern life expectancy at birth
real*8 LEb(1:19)
common // PA! physiological agedness
real*8 PA(i_start_date:i_end_date)
common // SC! specific concentration of MePiA in the atmosphere
real*8 SC(i_start_date:i_end_date)
common // sigma ! the standard deviations of the biblical life expectancy data
real*8 sigma(1:19)
!
! Integer arrays
common // jb ! jb is the date for biblical life span data
integer jb(1:19)
!
! Integer variables
common // i_pts
^ J. R. R. Tolkien, The Lord of the Rings (Boston: Houghton Mifflin Company, 1987), The Return of the King, 136.
^ Gerald E. Aardsma, A New Approach to the Chronology of Biblical History from Abraham to Samuel, 2nd ed. (Loda, IL: Aardsma Research & Publishing, 1993). www.BiblicalChronologist.org.
^ Gerald E. Aardsma, The Exodus Happened 2450 B.C. (Loda, IL: Aardsma Research & Publishing, 2008). www.BiblicalChronologist.org.
^ Amélie Kuhrt, The Ancient Near East, Volume I (New York: Routledge, 1995), 29.
^ Gerald E. Aardsma, Noah's Flood Happened 3520 B.C. (Loda, IL: Aardsma Research & Publishing, 2015). www.BiblicalChronologist.org.
^ Proverbs 24:11–12 [NASB, 1975].
^ biblicalchronologist.org/products/archives/newsletters.php
^ All of these books are freely available at www.BiblicalChronologist.org.
^ "General Theory of Aging for Biological Organisms: Aging in all organisms of all species is always simply progression of congenital disease." Gerald E. Aardsma, "A General Theory of Aging: Part I," The Biblical Chronologist 10.2 (February 12, 2020): 1–4. www.BiblicalChronologist.org.
^ San Francisco (AP), "Life Expectancy May Be Nearing Its Upper Limit," The News-Gazette (Champaign-Urbana, Illinois), February 19, 2001, pages A-1 and A-6.
^ Gerald E. Aardsma, Noah's Flood Happened 3520 B.C. (Loda, IL: Aardsma Research & Publishing, 2015), 307–313. www.BiblicalChronologist.org.
^ Gerald E. Aardsma, Noah's Flood Happened 3520 B.C. (Loda, IL: Aardsma Research & Publishing, 2015). www.BiblicalChronologist.org.
^ Gerald E. Aardsma, The Exodus Happened 2450 B.C. (Loda, IL: Aardsma Research & Publishing, 2008). www.BiblicalChronologist.org.
^ Gerald E. Aardsma, A New Approach to the Chronology of Biblical History from Abraham to Samuel, 2nd ed. (Loda, IL: Aardsma Research & Publishing, 1993).
^ Gerald E. Aardsma, "A Unification of Pre-Flood Chronology," The Biblical Chronologist 5.2 (March/April 1999): 1–18.
^ Gerald E. Aardsma, "The Cause of Noah's Flood," The Biblical Chronologist 3.5 (September/October 1997): 1–14.
^ Walter H. Eddy and Gilbert Dalldorf, The Avitaminoses: The Chemical, Clinical and Pathological Aspects of Vitamin Deficiency Diseases (Baltimore: The Williams & Wilkins Company, 1937), 175.
^ Walter H. Eddy and Gilbert Dalldorf, The Avitaminoses: The Chemical, Clinical and Pathological Aspects of Vitamin Deficiency Diseases (Baltimore: The Williams & Wilkins Company, 1937), 194.
^ Walter H. Eddy and Gilbert Dalldorf, The Avitaminoses: The Chemical, Clinical and Pathological Aspects of Vitamin Deficiency Diseases (Baltimore: The Williams & Wilkins Company, 1937), 163.
^ Joseph W. Kane and Morton M. Sternheim, Physics, (New York: John Wiley & Sons, 1980), Table 33.1, 577.
^ It does not need to be vitamin X itself which is carried forward in time in the body. One or more metabolites of vitamin X will also do. We are ignoring the distinction between vitamin X and its metabolites in an effort to keep the present discussion as simple as possible.
^ John H. Seinfeld and Spyros N. Pandis, Atmospheric Chemistry and Physics (New York: John Wiley & Sons, Inc., 1998), 315–317.
^ Gerald E. Aardsma, Noah's Flood Happened 3520 B.C. (Loda, IL: Aardsma Research & Publishing, 2015). www.BiblicalChronologist.org.
^ File: WOA09 sea-surf PO4 AYool.png (December 5, 2012, 11:15:13). Wikimedia Commons, the free media repository (accessed from https://en.wikipedia.org/wiki/World_Ocean_Atlas April 5, 2021).
^ File: WOA09 sea-surf NO3 AYool.png (December 5, 2012, 11:11:38). Wikimedia Commons, the free media repository (accessed from https://en.wikipedia.org/wiki/World_Ocean_Atlas April 5, 2021).
^ D. M. Sigman and M. P. Hain,"The Biological Productivity of the Ocean," Nature Education Knowledge 3.10 (2012) 21.
^ John H. Seinfeld and Spyros N. Pandis, Atmospheric Chemistry and Physics (New York: John Wiley & Sons, Inc., 1998), 59.
^ Joris Roels and Willy Verstraete, "Biological Formation of Volatile Phosphorus Compounds," Bioresource Technology 79 (2001): 243–250.
^ P. J. Craig and R. O. Jenkins, "Organometallic Compounds in the Environment: An Overview," Organic Metal and Metalloid Species in the Environment, ed. Alfred V. Hirner and Hendrik Emons (New York: Springer, 2004), 5.
^ William W. Metcalf and Wilfred A. van der Donk, "Biosynthesis of Phosphonic and Phosphinic Acid Natural Products," Annual Review of Biochemistry 78 (2009): 65–94.
^ Xiaomin Yu et al., "Diversity and Abundance of Phosphonate Biosynthetic Genes in Nature," Proceedings of the National Acadeny of Sciences of the United States of America 110.51 (December 17, 2013): 20759–20764.
^ William W. Metcalf and Wilfred A. van der Donk, "Biosynthesis of Phosphonic and Phosphinic Acid Natural Products," Annual Review of Biochemistry 78 (2009): 65–94.
^ William W. Metcalf and Wilfred A. van der Donk, "Biosynthesis of Phosphonic and Phosphinic Acid Natural Products," Annual Review of Biochemistry 78 (2009): 65–94.
^ Denham Harman, "The Biologic Clock: the Mitochondria?," Journal of the American Geriatrics Society 20 (1972): 145–147.
^ Denham Harman, "The Biologic Clock: the Mitochondria?," Journal of the American Geriatrics Society 20 (1972): 145–147.
^ Gerald E. Aardsma, Addendum to Aging: Cause and Cure (Loda, IL: Aardsma Research & Publishing, July 26, 2019), pages 2–5. www.BiblicalChronologist.org.
^ Gerald E. Aardsma, Addendum to Aging: Cause and Cure (Loda, IL: Aardsma Research & Publishing, July 26, 2019), pages 6–9. www.BiblicalChronologist.org.
^ Denham Harman, "The Biologic Clock: the Mitochondria?," Journal of the American Geriatrics Society 20 (1972): 145–147.
^ The average life span of modern males was calculated using data for U.S. males from the United States Social Security Administration's 2013 actuarial table, located at www.ssa.gov/oact/STATS/table4c6.html.
^ John H. Seinfeld and Spyros N. Pandis, Atmospheric Chemistry and Physics (New York: John Wiley & Sons, Inc., 1998), Section 21.4.3, 1098–1100.
^ William W. Metcalf et al., "Synthesis of Methylphosphonic Acid by Marine Microbes: A Source for Methane in the Aerobic Ocean," Science 337 (August 31, 2012): 1104–1107.
^ A second deficiency will become apparent in subsequent chapters dealing with the rate of healing of "aging." The model treats healing as instantaneous. In subsequent chapters it will be shown that, in actual fact, the healing of some aspects of "aging" may take years or even decades. The effect of this on the model is small. Healing comes into play only when the intake of MePiA exceeds the break-even threshold (Figure 11.3). For the model, this condition pertains only during the Spike. The main impact on the model of slowing the rate of healing would be to round off the sharp peak shown for the rise in life expectancies between Adam and Noah for pre-Flood individuals living into the Spike. The model is insensitive to this effect because there are no data points on this peak (i.e., no biblical life expectancy data points between Methuselah and Noah).
^ "Scurvy and Its Prevention and Control in Major Emergencies," World Health Organization, (1999): 17. www.unhcr.org/4cbef0599.pdf
^ For humans, vitamin D and iron are possible exceptions. However, there are reasons that these two nutrients are lower in human milk than their current recommended intakes for infants. Infants exposed to sunlight can make their own vitamin D. Therefore, unlike all other nutrients, milk is not the only source of vitamin D and did not have to supply the entire requirement of the infant before lifestyle changes and skin cancer concerns limited sun exposure of babies. Iron is a limiting nutrient for many pathogenic bacteria. In other words, their growth is limited by how much iron is available. Therefore, lower iron levels in the milk may actually be a protective mechanism against infection in the immunologically naive infant. Thus, even with vitamin D and iron, levels in human milk still appear to be optimized to meet the true needs of the infant.
^ See, for example, Eric S. Saltzman et al., "Glacial/Interglacial Variations in Methanesulfonate (MSA) in the Siple Dome Ice Core, West Antarctica," Geophysical Research Letters 33.11 (June 8, 2006).
^ http://www.fao.org/docrep/004/Y2809E/y2809e0c.htm (accessed April 12, 2021).
^ https://www.ncbi.nlm.nih.gov/books/NBK114302/ (accessed April 12, 2021).
^ Rebecca E. Watson et al., "Toxicity of Binary Chemical Munition Destruction Products: Methylphosphonic Acid, Methylphosphinic Acid, 2-Diisopropylaminoethanol, DF Neutralent, and QL Neutralent," International Journal of Toxicology 26 (2007): 503–512.
^ N. B. Munro et al., "The Sources, Fate, and Toxicity of Chemical Warfare Agent Degradation Products" Environmental Health Perspectives 107.12 (1999): 933–974.
^ https://pubchem.ncbi.nlm.nih.gov/compound/5234#section=Non-Human-Toxicity-Values (accessed June 1, 2020).
^ https://pubchem.ncbi.nlm.nih.gov/compound/2519#section=Acute-Effects (accessed June 1, 2020).
^ Rebecca E. Watson et al., "Toxicity of Binary Chemical Munition Destruction Products: Methylphosphonic Acid, Methylphosphinic Acid, 2-Diisopropylaminoethanol, DF Neutralent, and QL Neutralent," International Journal of Toxicology 26 (2007): 503–512.
^ Gerald E. Aardsma, "ELLM: the Extraordinarily Long-Lived Mouse," The Biblical Chronologist 10.9 (May 19, 2020): Figure 2, page 3. www.BiblicalChronologist.org.
^ Matthew P. Aardsma, "Intake Recommendations for Dr. Aardsma's Anti-Aging Vitamins," The Biblical Chronologist 10.10 (June 10, 2020): 1–8. www.BiblicalChronologist.org.
^ https://draardsmasvitamins.com/
^ https://www.ncbi.nlm.nih.gov/books/NBK56068/table/summarytables.t2/.
^ Genesis 5:23–24.
^ Genesis 5:31.
^ Gerald E. Aardsma, "Readers Write: Did Lamech Die in the Flood?," The Biblical Chronologist 7.5 (September/October 2001): 6–7. www.BiblicalChronologist.org.
^ These data present the difficulty, for survival curve construction purposes, that the birthdates of the individuals involved were not all the same. In fact, the birthdates of these individuals span 687 years (see Table 2.2). This difficulty can be overcome by assuming that conditions were sufficiently uniform in the pre-Flood period in question not to alter life spans. This amounts to the assumption that methylphosphine gas concentrations remained constant in the pre-Flood atmosphere during the time span of interest. Unfortunately, the Table 14.1 data themselves are, if anything, opposed to this assumption. The two longest life spans, for Jared and Methuselah, occur at the end of the list. This suggests the possibility that atmospheric methylphosphine gas concentrations may have been slowly increasing pre-Flood. This possibility is encouraged when the pre-Flood source of methylphosphine gas, from biogenic ocean floor sediments, is considered. It is clearly possible for biogenic sediments to build up with the passage of time, consequently releasing increasing amounts of decomposition gases. Nonetheless, an approximate survival curve can be constructed from these data as long as one is prepared to accept the uncertainty in pre-Flood atmospheric methylphosphine gas concentration. In the present application, this uncertainty turns out to be of relatively minor importance.
^ ssa.gov/oact/STATS/table4c6.html (accessed March 2, 2020).
^ Gerald E. Aardsma, "Modeling Survival Curves in Light of the General Theory of Aging," The Biblical Chronologist 10.7 (April 24, 2020): page 4, Equation 7. www.BiblicalChronologist.org.
^ Gerald E. Aardsma, "Actuarial Data Show That Vitamin MePA Deficiency Disease Is Needlessly Killing a Quarter Million Americans Annually," The Biblical Chronologist 10.15 (October 28, 2020): 8–9. www.BiblicalChronologist.org.
^ ssa.gov/oact/STATS/table4c6.html (accessed March 2, 2020).
^ www.yourgenome.org/facts/what-is-a-telomere (accessed November 7, 2020).
^ This observation leads immediately to the postulate that the phylogenetic development of a self-replicating, mutating machine will be grainy rather than smooth. Even should mutation of the code be a gradual, smooth process, the effect of code mutations on the coded idea is likely to be grainy and has potential even to be discontinuous.
^ Maria Falkenberg, "Mitochondrial DNA Replication in Mammalian Cells: Overview of the Pathway" Essays in Biochemistry 62.3 (July 20, 2018): 287–296.
^ Personal communication October 25, 2020.
^ For an introduction to the current map of human mtDNA, see: Maria Falkenberg, "Mitochondrial DNA Replication in Mammalian Cells: Overview of the Pathway" Essays in Biochemistry 62.3 (July 20, 2018): 287–296, Figure 1.
^ Gerald E. Aardsma, "Human Aging is a Two-Phase Disease," The Biblical Chronologist 10.8 (May 13, 2020): 9–10. www.BiblicalChronologist.org.
^ For 15 degrees of freedom, Χν2 increased from 1.22 for rejuvenation allowed to 1.36 for rejuvenation not allowed.
^ Jason Pellettieri and Alejandro Sánchez Alvarado. "Cell Turnover and Adult Tissue Homeostasis: From Humans to Planarians" Annual Review of Genetics 41 (2007): 83–105.
^ https://en.wikipedia.org/wiki/Mitochondrial_fusion (accessed July 16, 2021).
^ Benedikt Westermann, "Merging Mitochondria Matters," EMBO Reports 3 (2002): 527–531.
^ Rebecca E. Watson et al., "Toxicity of Binary Chemical Munition Destruction Products: Methylphosphonic Acid, Methylphosphinic Acid, 2-Diisopropylaminoethanol, DF Neutralent, and QL Neutralent," International Journal of Toxicology 26 (2007): 503–512.
^ Constantin Lohrer et al., "Methodological Aspects of Methylphosphonic Acid Analysis: Determination in River and Coastal Water Samples," Talanta 211 (May 1, 2020), Article 120724: 1–8.
^ Gerald E. Aardsma, "A General Theory of Aging: Part I," The Biblical Chronologist 10.2 (February 12, 2020): 2. www.BiblicalChronologist.org.
^ A. Piro et al., "Casimir Funk: His Discovery of the Vitamins and Their Deficiency Disorders" Annals of Nutrition & Metabolism 57.2 (2010): 85–88.
^ Walter H. Eddy, Vitaminology: The Chemistry and Function of the Vitamins (Baltimore: The Williams & Wilkins Company, 1949), Foreword.
^ H. Chick, "The Discovery of Vitamins" Progress in Food & Nutrition Science 1.1 (1975): 1–20.
^ Physicians' Vitamin Reference Book, third edition (New York: E.R. Squibb & Sons, January 1940), 46–47.
^ Sareen S. Gropper, Jack L. Smith, and James L. Groff, Advanced Nutrition and Human Metabolism, 5th edition (Belmont, CA: Wadsworth, Cengage Learning, 2009), 309–427.
^ Sareen S. Gropper, Jack L. Smith, and James L. Groff, Advanced Nutrition and Human Metabolism, 5th edition (Belmont, CA: Wadsworth, Cengage Learning, 2009), 309–372.
^ pubchem.ncbi.nlm.nih.gov/compound/5311498#section=Toxicity-Summary (accessed June 9, 2020).
^ Rebecca E. Watson et al., "Toxicity of Binary Chemical Munition Destruction Products: Methylphosphonic Acid, Methylphosphinic Acid, 2-Diisopropylaminoethanol, DF Neutralent, and QL Neutralent," International Journal of Toxicology 26 (2007): 503–512.
^ https://pubchem.ncbi.nlm.nih.gov/compound/62156 (accessed June 6, 2020).
^ https://pubchem.ncbi.nlm.nih.gov/compound/62156#section=Human-Toxicity-Excerpts (accessed June 6, 2020).
^ William W. Metcalf et al., "Synthesis of Methylphosphonic Acid by Marine Microbes: A Source for Methane in the Aerobic Ocean," Science 337 (August 31, 2012): 1104-1107.
^ Amélie Kuhrt, The Ancient Near East, Volume I (New York: Routledge, 1995), 32. The question mark in parentheses at the end is part of the original translation, indicating some uncertainty on the part of the translator.
^ Ecclesiastes 12:9 [NASB, 1975].
^ Proverbs 5:15 [NASB, 1975].
^ https://www.biblicalchronologist.org/products/vitamin_MePA_testimonials.php.
^ See the "Gerald and Helen's Personal Testimonial" section in the epilogue.
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials_Contreras.php
^ Matthew P. Aardsma, "Intake Recommendations for Dr. Aardsma's Anti-Aging Vitamins," The Biblical Chronologist 10.10 (June 10, 2020): 1–8. www.BiblicalChronologist.org.
^ https://draardsmasvitamins.com/
^ Gerald E. Aardsma, "Human Aging is a Two-Phase Disease," The Biblical Chronologist 10.8 (May 13, 2020): 1–10. www.BiblicalChronologist.org.
^ https://draardsmasvitamins.com/
^ www.ssa.gov/oact/NOTES/as120/LifeTables_Body.html (accessed September 24, 2020).
^ ssa.gov/oact/STATS/table4c6.html (accessed March 2, 2020).
^ cdc.gov/nchs/data/databriefs/db293.pdf, page 4, Figure 4, Notes (accessed October 19, 2020).
^ Gerald E. Aardsma, "Modeling Survival Curves in Light of the General Theory of Aging," The Biblical Chronologist 10.7 (April 24, 2020): page 4, Equation 7. www.BiblicalChronologist.org.
^ Gerald E. Aardsma, "Human Aging is a Two-Phase Disease," The Biblical Chronologist 10.8 (May 13, 2020): 1–10. www.BiblicalChronologist.org.
^ cdc.gov/nchs/data/databriefs/db293.pdf, page 4, Figure 4, Notes (accessed October 19, 2020).
^ Gerald E. Aardsma, "Modeling Survival Curves in Light of the General Theory of Aging," The Biblical Chronologist 10.7 (April 24, 2020): page 4. www.BiblicalChronologist.org.
^ www.ssa.gov/oact/NOTES/as120/LifeTables_Body.html (accessed September 24, 2020).
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials.php
^ www.biblicalchronologist.org/RDI.php
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials_immune_system.php; www.biblicalchronologist.org/products/vitamin_MePA_testimonials_arthritis.php.
^ DeLisa Fairweather, Sylvia Frisancho-Kiss, and Noel R. Rose, "Sex Differences in Autoimmune Disease from a Pathological Perspective," The American Journal of Pathology 173.3 (September 2008): 600–609.
^ www.health.harvard.edu/heart-health/the-heart-attack-gender-gap (accessed October 17, 2020); Hae-In Kim et al., "Sex Differences in Cancer: Epidemiology, Genetics and Therapy." Biomolecules & Therapeutics 26.4 (July 1, 2018): 335–342.
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials.php; www.agingcauseandcure.com/testimonials/
^ ssa.gov/oact/STATS/table4c6.html (accessed March 2, 2020).
^ Biological considerations discourage the idea of mutable rates of physical maturation. The uniformly advanced ages (minimum 65 years) of the fathers at the birth of their sons recorded in Genesis 5 may seem to challenge this, especially as the natural sense of this passage suggests that it is a list of male heirs and thus dominated by firstborn sons. But it is possible to ascribe these advanced ages to cultural differences rather than to a slower rate of physical maturation than pertains today. Average marital age varies considerably between different cultures today. Life expectancy appears to be a significant factor influencing marital age, with lower life expectancies correlating with lower marital ages. Thus, the very long pre-Flood life expectancies make it conceivable that a difference between pre-Flood and modern cultural practices is the entire explanation of the advanced ages of fatherhood recorded in Genesis 5. But there may be an additional factor. To see this, first observe that it is probably fallacious to equate sexual maturity with physical maturity. Notice, for example, that sexual fertility is generally achieved prior to physical maturity today. While the age of physical maturity seems likely to be more or less fixed, the age of sexual maturity may very well be mutable. The vital importance of reproduction to survival of the species provides a theoretical basis for the idea that the age of onset of sexual fertility might be influenced by physiological factors affecting probable life span of an organism. Because of the physiological stress and consequent shortened life spans imposed by the absence from modern diets of the anti-aging vitamins, it seems possible that while modern individuals mature physically at much the same rate as did pre-Flood individuals, sexual maturity today may occur significantly earlier than it did prior to the Flood.
^ www.cdc.gov/injury/wisqars/pdf/leading_causes_of_death_by_age_group_2016-508.pdf (accessed January 21, 2021).
^ www.cdc.gov/nchs/fastats/accidental-injury.htm (accessed January 21, 2021).
^ Psalm 90:10 [KJV].
^ Isaiah 65:20b [NASB, 1975].
^ eh.net/eha/wp-content/uploads/2016/08/ClayTroesken.pdf (accessed April 6, 2020).
^ We could not do this to people today—withhold one deficient vitamin while administering another deficient vitamin. The administering of one deficient vitamin is better than the administering of no deficient vitamin, but knowledgeably withholding any deficient vitamin would be unethical. Humans, uniquely made in God's image (Genesis 1:26–27), are not to be treated like lab animals, and "good science" must give way before human ethics.
^ https://www.biblicalchronologist.org/products/vitamin_MePA_testimonials_im-mune_system.php
^ https://en.wikipedia.org/wiki/Sarin (accessed July 5, 2017).
^ https://en.wikipedia.org/wiki/Sarin#Diagnostic_tests (accessed July 5, 2017).
^ S. Kuwabara et al., "Long Term Prognosis of Chronic Inflammatory Demyelinating Polyneuropathy: a Five Year Follow Up of 38 Cases," Journal of Neurology, Neurosurgery and Psychiatry 77.1 (Jan 2006): 66–70.
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials_Helen_and_ Gerald.php
^ Personal communication, January 29, 2019.
^ www.biblicalchronologist.org/products/vitamin_MePA_testimonials_Helen_and_ Gerald.php
^ MePA is patent protected under U.S. patent number 10863764, "Compositions and Methods for Treating Aging and/or Improving Human Health." MePiA is patent pending under U.S. patent application number 16/936,168, "Methylphosphinic Acid Compositions and Methods for Reducing Aging."

